Scalable Synthesis of Chiral Beta-Butyrolactone Using Novel Cinchona-Derived Bifunctional Catalysts
Scalable Synthesis of Chiral Beta-Butyrolactone Using Novel Cinchona-Derived Bifunctional Catalysts
The landscape of asymmetric synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more sustainable and cost-effective catalytic systems. A significant breakthrough in this domain is detailed in patent CN115073399A, which introduces a novel method for preparing chiral beta-butyrolactone compounds using a uniquely designed cinchona alkaloid catalyst. This technology addresses long-standing challenges in the [2+2] cycloaddition of aldehydes and ketenes, a critical transformation for generating high-value synthetic building blocks. The core innovation lies in the creation of a bifunctional catalyst that possesses both Lewis base (nitrogen) and Lewis acid (boron) active centers, enabling superior stereocontrol and reaction efficiency compared to historical precedents. For procurement and R&D teams seeking a reliable pharmaceutical intermediate supplier, this methodology represents a paradigm shift towards greener, high-performance manufacturing protocols that do not compromise on enantiomeric purity.
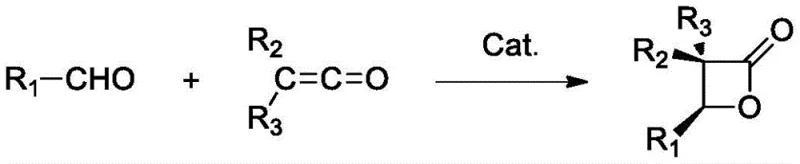
The strategic value of this patent extends beyond mere academic interest; it offers a tangible pathway for cost reduction in fine chemical manufacturing by utilizing abundant natural alkaloids as starting materials. The resulting chiral beta-butyrolactone structures are ubiquitous in bioactive natural products and serve as versatile precursors for complex drug molecules. By leveraging the cooperative catalysis mechanism described in the patent, manufacturers can achieve yields approaching 99 percent with enantiomeric excess values up to 97 percent, all under remarkably mild reaction conditions. This level of performance is critical for maintaining stringent purity specifications required by global regulatory bodies, ensuring that the final API intermediates meet the rigorous quality standards demanded by the international pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric [2+2] cycloaddition of ketenes and carbonyl compounds has been plagued by significant operational and economic hurdles that hinder widespread industrial adoption. Early pioneering work by Wynberg and Staring in 1982 utilized quinidine to achieve asymmetric induction, but this system was severely restricted to activated aldehydes containing multiple chlorine atoms, limiting its substrate scope considerably. Subsequent improvements by Nelson et al. in 2004 introduced a trimethylsilyl-protected quinine complex with lithium perchlorate, which expanded the substrate range but introduced new problems regarding cost and waste management. The requirement for large amounts of expensive lithium perchlorate (15-300 mol%) not only drives up raw material costs but also creates substantial issues with salt waste disposal and product purification. Furthermore, other methods reported by Fu et al. necessitated extremely low reaction temperatures of -78°C and specific solvents like tetrahydrofuran, imposing heavy energy burdens and cryogenic infrastructure requirements on the manufacturing process.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach disclosed in patent CN115073399A utilizes an in-situ generated bifunctional catalyst that operates efficiently under ambient to mildly cooled conditions. By modifying natural cinchona alkaloids such as quinine or quinidine through a simple hydroboration reaction, the inventors have created a catalyst system that activates both reaction partners simultaneously without the need for exogenous salt additives. This new methodology allows the reaction to proceed at temperatures ranging from 0°C to 50°C, drastically reducing energy consumption compared to the cryogenic conditions of previous art. The elimination of lithium perchlorate and the use of low catalyst loadings (0.1-1.0 mol%) streamline the workup procedure, removing the need for extensive aqueous washes to remove metal salts. This results in a cleaner reaction profile and a more robust process that is inherently safer and more scalable for commercial production environments.
Mechanistic Insights into Cinchona-Derived Bifunctional Catalysis
The exceptional performance of this catalytic system stems from its sophisticated dual-activation mechanism, which orchestrates the spatial arrangement of reactants to maximize stereoselectivity. The catalyst features a boron atom that acts as a Lewis acid to activate the carbonyl group of the aldehyde substrate, increasing its electrophilicity. Simultaneously, the tertiary amine nitrogen within the cinchona scaffold functions as a Lewis base to activate the ketene species. This cooperative interaction brings the two reactants into close proximity within a rigid chiral environment defined by the alkaloid backbone. Such precise molecular recognition ensures that the [2+2] cycloaddition occurs with high facial selectivity, leading to the formation of the desired enantiomer with minimal formation of the opposite isomer. This mechanistic elegance allows the system to tolerate a wide variety of substrates, including both electron-rich and electron-deficient aromatic aldehydes as well as aliphatic variants, without sacrificing optical purity.
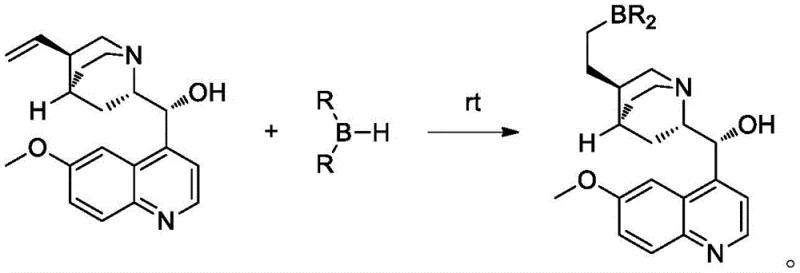
From an impurity control perspective, this mechanism offers distinct advantages by minimizing side reactions that typically plague ketene chemistry. The rapid and selective activation of the aldehyde prevents polymerization of the ketene or non-selective background reactions that could degrade the ee value. The patent data indicates that even with challenging substrates like acetaldehyde or propionaldehyde, the system maintains robust stereocontrol, achieving ee values consistently above 70 percent and often exceeding 90 percent for aromatic substrates. For R&D directors focused on impurity profiles, this means a simpler crystallization strategy to upgrade the optical purity of the final product. The ability to tune the catalyst by selecting different cinchona alkaloids (quinine vs. quinidine) or borane reagents provides an additional layer of process optimization, allowing chemists to invert chirality or enhance selectivity based on specific project requirements without redesigning the entire synthetic route.
How to Synthesize Chiral Beta-Butyrolactone Efficiently
The implementation of this technology in a pilot or production setting follows a straightforward protocol that leverages standard chemical engineering equipment. The process begins with the preparation of the catalyst solution, where the cinchona alkaloid and borane reagent are mixed in an anhydrous solvent such as dichloromethane or toluene at room temperature. This step is rapid, typically completing within 20 minutes, and does not require specialized handling beyond standard inert atmosphere techniques. Once the catalyst is formed, the aldehyde substrate is introduced, and the mixture is cooled to the optimal reaction temperature, often 0°C, to maximize selectivity. The ketene gas is then fed slowly into the reactor, ensuring that its concentration remains low to prevent dimerization, while the catalyst drives the cycloaddition to completion. Detailed standardized synthesis steps see the guide below.
- Prepare the bifunctional catalyst by reacting cinchona alkaloid (e.g., quinine) with disubstituted borane in an anhydrous solvent at room temperature for 20 minutes.
- Add the aldehyde substrate and solvent to the reaction vessel, cooling the mixture to 0°C to 50°C depending on reactivity requirements.
- Slowly feed ketene gas into the reaction mixture while maintaining inert atmosphere, allowing the [2+2] cycloaddition to proceed for 1 to 6 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates directly into enhanced operational resilience and significant cost optimization opportunities. The shift away from expensive and hazardous additives like lithium perchlorate removes a major cost driver from the bill of materials, while the use of naturally derived cinchona alkaloids ensures a stable and renewable supply of the chiral source. The mild reaction conditions eliminate the need for energy-intensive cryogenic cooling systems, allowing facilities to utilize standard chilled water loops instead of liquid nitrogen or dry ice-acetone baths. This reduction in utility consumption contributes to a lower carbon footprint and aligns with increasingly strict environmental regulations governing chemical manufacturing. Furthermore, the simplified workup procedure reduces solvent usage and waste generation, lowering the overall cost of goods sold and minimizing the environmental impact associated with waste disposal.
- Cost Reduction in Manufacturing: The elimination of stoichiometric salt additives and the ability to use low catalyst loadings drastically reduce raw material expenses. By avoiding the purchase and disposal of large quantities of lithium perchlorate, manufacturers can achieve substantial cost savings per kilogram of product. Additionally, the higher yields and selectivity reduce the loss of valuable starting materials, further improving the overall process economics and making the production of high-purity pharmaceutical intermediates more financially viable.
- Enhanced Supply Chain Reliability: Utilizing commercially available and abundant raw materials such as quinine and common boranes mitigates the risk of supply disruptions associated with specialized reagents. The robustness of the reaction conditions means that production can be maintained consistently without the volatility often introduced by sensitive low-temperature processes. This stability ensures reliable delivery schedules for downstream customers, fostering stronger long-term partnerships and reducing the risk of production delays due to equipment failure or reagent shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-ton commercial production without significant re-engineering. The absence of heavy metals and the use of benign solvents facilitate easier regulatory approval and compliance with green chemistry principles. This ease of scale-up allows manufacturers to respond quickly to market demand fluctuations, ensuring a continuous supply of critical intermediates while maintaining a strong environmental stewardship profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel catalytic system. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: What are the primary advantages of this cinchona-derived catalyst over traditional lithium perchlorate systems?
A: Unlike traditional systems requiring stoichiometric amounts of expensive lithium perchlorate, this novel bifunctional catalyst operates at extremely low loadings (0.1-1.0 mol%) and eliminates the need for hazardous salt waste disposal, significantly simplifying downstream purification.
Q: Can this method accommodate both aromatic and aliphatic aldehydes effectively?
A: Yes, the patent data demonstrates high versatility, successfully catalyzing reactions with benzaldehyde derivatives as well as aliphatic aldehydes like acetaldehyde and butyraldehyde, achieving yields up to 99% and ee values reaching 97%.
Q: Is the catalyst preparation suitable for large-scale industrial manufacturing?
A: Absolutely. The catalyst is generated in situ at room temperature within 20 minutes using commercially available raw materials, and the subsequent reaction proceeds under mild temperatures (0-50°C) and normal pressure, making it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN115073399A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. We understand that in the pharmaceutical industry, consistency is key, and our dedicated technical team works tirelessly to optimize every parameter of the synthesis to meet the exacting standards of global regulatory agencies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact us today to request specific COA data and route feasibility assessments that demonstrate how we can help you achieve your production goals efficiently and economically. Let us be your trusted partner in navigating the complexities of chiral synthesis and bringing your innovative therapies to market faster.
