Scalable Green Synthesis of Homoallylic Alcohols via Magnesium Promotion for Global Supply Chains
Introduction to Advanced Green Synthesis Technologies
The pharmaceutical and fine chemical industries are currently undergoing a paradigm shift towards sustainable manufacturing processes that minimize environmental impact while maximizing efficiency. A pivotal development in this domain is documented in patent CN102060659A, which introduces a novel method for preparing homoallylic alcohols, a critical structural motif found in numerous bioactive natural products and pharmaceutical agents. This technology leverages activated magnesium powder as a promoter to facilitate the coupling of aromatic aldehydes with allyl bromide under remarkably mild conditions. By operating at room temperature and eliminating the need for organic solvents during the reaction phase, this approach addresses key pain points in modern chemical synthesis, including energy consumption and hazardous waste generation. For R&D directors and procurement specialists, understanding the mechanistic robustness and economic implications of this patent is essential for optimizing supply chains and reducing the cost of goods sold for complex intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of homoallylic alcohols has relied on a variety of metal-mediated reactions involving elements such as indium, antimony, lead, manganese, iron, zinc, and tin. While effective in laboratory settings, these conventional methods present significant drawbacks when evaluated for industrial scalability and environmental compliance. Many of these protocols necessitate the use of stoichiometric amounts of expensive or toxic metals, which complicates waste disposal and increases raw material costs substantially. Furthermore, traditional approaches often require harsh reaction conditions, including low temperatures or the presence of strong Lewis acids and organic solvents, which escalate energy usage and pose safety hazards. The reliance on such complex conditions frequently leads to side reactions, resulting in lower product purity and necessitating rigorous, cost-intensive purification steps that erode profit margins and extend lead times for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy techniques, the method described in CN102060659A utilizes activated magnesium powder as a non-toxic and inexpensive promoter to drive the reaction between aromatic aldehydes and allyl bromide. This innovative strategy operates effectively at room temperature, completing the transformation in a mere 5 to 6 minutes, which represents a drastic improvement in throughput efficiency. The process is notably solvent-free during the reaction stage, thereby eliminating the environmental burden and cost associated with solvent procurement, recovery, and disposal. By achieving product yields exceeding 80 percent and purity levels greater than 99 percent through simple column chromatography, this method offers a streamlined pathway that aligns perfectly with the principles of green chemistry. 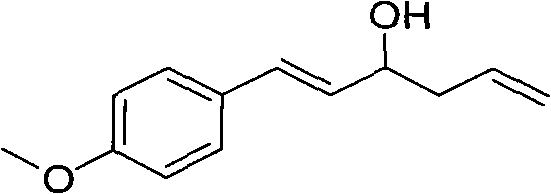
Mechanistic Insights into Magnesium-Promoted Allylation
The core of this technological breakthrough lies in the activation of the magnesium surface, which facilitates the formation of an organomagnesium species in situ, akin to a Barbier-type reaction mechanism. The activation process involves treating magnesium powder with a dilute hydrochloric acid solution, which effectively removes the passivating oxide layer that typically inhibits reactivity. This freshly exposed metallic surface allows for the rapid insertion of magnesium into the carbon-bromine bond of the allyl bromide, generating a reactive nucleophile without the need for pre-formed Grignard reagents or sensitive catalysts. The subsequent nucleophilic attack on the carbonyl carbon of the aromatic aldehyde proceeds with high regioselectivity and stereoselectivity, ensuring the formation of the desired homoallylic alcohol scaffold with minimal byproduct formation. This mechanistic simplicity is a key driver for the method's robustness, as it reduces the sensitivity to moisture and oxygen compared to traditional organometallic protocols.
From an impurity control perspective, the absence of transition metal catalysts such as palladium or nickel eliminates the risk of heavy metal contamination, a critical quality attribute for pharmaceutical intermediates destined for clinical applications. The reaction profile is clean, with the primary impurities being unreacted starting materials which are easily removed during the aqueous workup and extraction phases. The use of saturated ammonium chloride for quenching ensures the complete protonation of the alkoxide intermediate, preventing retro-aldol type decompositions that can occur under basic conditions. Consequently, the final product obtained after solvent evaporation and chromatographic separation exhibits exceptional purity, often surpassing 99 percent, which significantly reduces the burden on downstream quality control laboratories and accelerates the release of batches for further synthetic elaboration.
How to Synthesize Homoallylic Alcohol Efficiently
The synthesis protocol outlined in the patent provides a standardized framework for producing high-purity homoallylic alcohols suitable for both laboratory research and pilot-scale manufacturing. The process begins with the meticulous activation of the magnesium promoter, followed by the direct addition of substrates under ambient conditions, and concludes with a straightforward extractive workup. This sequence minimizes unit operations and equipment requirements, making it an attractive option for facilities looking to optimize their production footprint. The detailed standard operating procedures for substrate ratios, activation times, and purification parameters are critical for ensuring batch-to-batch consistency and maximizing yield.
- Activate magnesium powder by stirring in dilute hydrochloric acid, followed by washing with water, acetone, and ether, and vacuum drying.
- React aromatic aldehyde and allyl bromide with the activated magnesium promoter at room temperature for 5-6 minutes without additional organic solvents.
- Quench the reaction with saturated ammonium chloride, extract with diethyl ether, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this magnesium-promoted synthesis route offers tangible strategic advantages that extend beyond mere technical feasibility. The substitution of expensive and supply-constrained metals like indium or tin with abundant magnesium powder fundamentally alters the cost structure of the manufacturing process, leading to significant raw material savings. Additionally, the elimination of organic solvents during the reaction phase reduces the volume of hazardous waste generated, thereby lowering disposal costs and simplifying regulatory compliance regarding environmental emissions. The mild reaction conditions also translate to reduced energy consumption, as there is no need for cryogenic cooling or prolonged heating, further contributing to a lower carbon footprint and operational expenditure.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of costly metal promoters with inexpensive magnesium powder, which is globally available and price-stable. By removing the requirement for specialized catalysts and reducing solvent usage, the overall variable cost per kilogram of the intermediate is drastically simplified and optimized. This cost efficiency allows manufacturers to offer more competitive pricing to downstream clients while maintaining healthy margins, creating a resilient economic model that can withstand fluctuations in the prices of precious metals or specialty reagents.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like magnesium and allyl bromide mitigates the risk of supply chain disruptions often associated with niche catalysts or rare earth metals. The robustness of the reaction at room temperature ensures that production can continue uninterrupted even in facilities with limited utility infrastructure, enhancing the overall reliability of supply. Furthermore, the short reaction time of approximately 5 minutes allows for rapid turnover of reactor vessels, increasing the effective capacity of existing manufacturing plants without the need for capital-intensive expansion projects.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic extraction and drying, makes this process highly amenable to scale-up from gram to ton quantities. The absence of toxic heavy metals in the final product streamlines the regulatory approval process for pharmaceutical applications, as extensive testing for residual metal impurities is minimized. This alignment with green chemistry principles not only satisfies increasingly stringent environmental regulations but also enhances the brand reputation of the manufacturer as a sustainable partner in the global chemical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and practical application scenarios. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using magnesium powder over traditional metal promoters?
A: Unlike traditional promoters such as indium, tin, or zinc which often require expensive reagents or generate toxic waste, magnesium powder is inexpensive, non-toxic, and abundant. The patented method eliminates the need for organic solvents during the reaction phase, significantly reducing environmental pollution and downstream purification costs while maintaining yields above 80%.
Q: How does this method improve reaction efficiency compared to conventional Lewis acid catalysis?
A: Conventional methods often rely on Lewis acids or metal salts that require low-temperature conditions and long reaction times. This magnesium-promoted protocol operates efficiently at room temperature with a reaction time of merely 5 to 6 minutes, drastically simplifying the thermal management requirements and accelerating the production cycle for high-purity intermediates.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is highly scalable due to its simplicity and mild conditions. The absence of sensitive catalysts and the use of room temperature conditions minimize safety risks associated with exothermic runaway reactions. Furthermore, the straightforward workup procedure involving simple extraction and chromatography facilitates easy adaptation to multi-kilogram or ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homoallylic Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the magnesium-promoted synthesis technology described in CN102060659A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry method are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of homoallylic alcohol meets the exacting standards required by global innovator companies.
We invite you to collaborate with our technical team to explore how this cost-effective and environmentally friendly route can be tailored to your specific project needs. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies for your supply chain. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a reliable supply of high-purity intermediates for your next breakthrough therapy.
