Advanced Electrochemical Oxidation for High-Purity Alpha,alpha-Dibromoketone Manufacturing
Advanced Electrochemical Oxidation for High-Purity Alpha,alpha-Dibromoketone Manufacturing
The landscape of fine chemical synthesis is undergoing a transformative shift towards greener, more efficient methodologies, as exemplified by the groundbreaking technology disclosed in patent CN111118529B. This patent introduces a novel diaphragm-free electrochemical oxidation method for synthesizing alpha,alpha-dibromoketone compounds, a class of versatile intermediates crucial for the development of complex pharmaceutical agents. Unlike traditional routes that rely on harsh oxidants and toxic metal catalysts, this innovation utilizes a streamlined electrolytic process operating at room temperature, achieving exceptional purity levels exceeding 98 percent. For R&D directors and procurement strategists alike, this represents a pivotal opportunity to enhance supply chain resilience while adhering to increasingly stringent environmental and safety regulations. The method's ability to generate carbon-bromine bonds efficiently without transition metals addresses a long-standing pain point in the production of high-value drug precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,alpha-dihalogenoketone compounds has been plagued by significant operational and safety challenges that hinder large-scale commercial viability. Conventional protocols typically necessitate the use of strong chemical oxidants such as hydrogen peroxide or molecular oxygen, often in conjunction with electrophilic halogen sources like N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS). These reactions frequently demand rigorous conditions, including elevated temperatures and the presence of expensive transition metal catalysts to drive the transformation forward. Furthermore, the reliance on metal salts and photocatalytic systems introduces the risk of heavy metal contamination, necessitating costly and time-consuming purification steps to meet pharmaceutical grade specifications. The atom economy of these traditional halogenation processes is often poor, generating substantial waste streams that complicate disposal and increase the overall environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast, the electrochemical strategy outlined in CN111118529B offers a paradigm shift by replacing chemical oxidants with electrons as the primary driving force for the reaction. This diaphragm-free electrolytic system operates under remarkably mild conditions, specifically at room temperature, thereby eliminating the energy costs associated with heating or cooling large reactor vessels. By utilizing a simple mixture of lithium perchlorate (LiClO4) and tetrabutylammonium iodide (nBu4NI) as electrolytes in an acetonitrile solvent system, the method effectively activates dihalogenated alkynes to form the desired ketone structures. The absence of a diaphragm simplifies the reactor design significantly, reducing capital expenditure for equipment and facilitating easier maintenance. This approach not only streamlines the workflow but also ensures that the final product is free from metal catalyst residues, a critical quality attribute for any reliable pharmaceutical intermediate supplier aiming to serve the global API market.
Mechanistic Insights into Diaphragm-Free Electrochemical Oxidation
The core of this technological breakthrough lies in the unique interplay between the electrochemical potential and the specific electrolyte system employed. During the electrolysis process, the anodic oxidation generates reactive halogen species in situ from the halogen solvent, such as dibromomethane, which then react with the alkyne substrate. The presence of water and the specific ratio of electrolytes, particularly the 1:0.5 to 1.5 molar ratio of alkyne to LiClO4, creates an optimal environment for the formation of C-C and C-Br bonds. This mechanism avoids the formation of aggressive radical species that often lead to over-oxidation or decomposition of sensitive functional groups on the aromatic ring. Consequently, the reaction exhibits excellent chemoselectivity, tolerating various substituents including electron-donating groups like methyl and tert-butyl, as well as electron-withdrawing moieties, although the latter may slightly influence reaction kinetics.
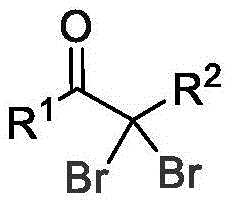
Furthermore, the control of impurities is inherently superior in this electrochemical setup compared to batch chemical oxidation. Since the reaction rate is governed by the applied current (optimized at 10 mA in the examples), the generation of reactive intermediates is steady and controlled, preventing the localized hotspots of reactivity that often cause side reactions in traditional batch processes. The use of platinum electrodes ensures stability and longevity, minimizing the introduction of particulate matter into the reaction mixture. Post-reaction workup is equally straightforward, involving simple rotary evaporation followed by column chromatography, which efficiently separates the target alpha,alpha-dibromoketone from minor byproducts. This robustness in impurity control is vital for ensuring that the final material meets the stringent purity specifications required for downstream coupling reactions in drug synthesis.
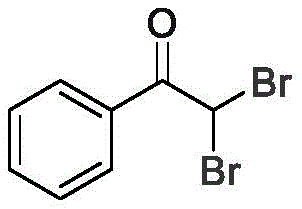
How to Synthesize Alpha,alpha-Dibromoketones Efficiently
To implement this cutting-edge synthesis route effectively, precise adherence to the optimized reaction parameters is essential for maximizing yield and reproducibility. The process begins with the careful preparation of the electrolyte solution, ensuring that the molar ratios of the alkyne substrate, LiClO4, and nBu4NI are maintained within the specified ranges to facilitate efficient charge transfer. The reaction mixture, containing the alkyne, electrolytes, water, and halogen solvent in acetonitrile, is subjected to constant current electrolysis for a duration of approximately 6 hours. Detailed standard operating procedures regarding electrode spacing, stirring rates, and workup protocols are critical for scaling this technology from the benchtop to pilot plant operations. For a comprehensive guide on the exact experimental steps and safety precautions, please refer to the standardized synthesis instructions below.
- Mix the alkyne compound with LiClO4, nBu4NI, H2O, a halogen solvent (such as dibromomethane), and acetonitrile in a reaction vessel.
- Equip the vessel with platinum electrodes and perform constant current electrolysis at 10 mA for 6 hours at room temperature without a diaphragm.
- Remove solvents via rotary evaporation and purify the crude product using column chromatography with petroleum ether and ethyl acetate to obtain the final alpha,alpha-dibromoketone.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this electrochemical methodology offers profound advantages that directly impact the bottom line and supply chain security. By eliminating the need for expensive transition metal catalysts and hazardous chemical oxidants, manufacturers can achieve significant cost reduction in fine chemical manufacturing. The simplified reagent profile reduces the complexity of raw material sourcing, mitigating the risks associated with supply disruptions of specialized catalysts. Moreover, the mild reaction conditions translate to lower energy consumption and reduced wear and tear on processing equipment, further enhancing the economic viability of the process. These factors combined create a more resilient supply chain capable of delivering high-quality intermediates with greater consistency and reliability.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes a major cost driver from the bill of materials, while also obviating the need for expensive metal scavenging resins or complex filtration steps during purification. This streamlined workflow reduces both material costs and labor hours, leading to substantial overall savings in the production budget. Additionally, the high atom efficiency of the electrochemical process minimizes waste generation, lowering disposal costs and environmental compliance fees. The ability to operate at room temperature further contributes to energy savings, making the process economically attractive for large-scale production runs.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as lithium perchlorate and common organic solvents, are widely available commodities with stable global supply chains. This contrasts sharply with proprietary catalysts that may be sourced from single suppliers, creating bottlenecks. By relying on readily accessible materials, procurement teams can secure long-term contracts with better pricing and ensure uninterrupted production schedules. The robustness of the method against variations in substrate electronics also means that a wider range of starting materials can be utilized without compromising yield, providing flexibility in raw material selection.
- Scalability and Environmental Compliance: The diaphragm-free design of the electrochemical cell simplifies the engineering requirements for scaling up, allowing for easier transition from laboratory to commercial scale production. The absence of toxic heavy metals in the process effluent significantly simplifies wastewater treatment and aligns with green chemistry principles, reducing the regulatory burden on the manufacturing facility. This environmental compatibility is increasingly important for maintaining social license to operate and meeting the sustainability goals of multinational corporate partners. The process is inherently safer due to the avoidance of strong oxidants, reducing the risk of thermal runaway incidents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on its practical application. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production lines. The answers reflect the specific advantages of the diaphragm-free system and its impact on product quality and process efficiency.
Q: Does this electrochemical method leave metal residues in the final product?
A: No, the entire reaction process does not require metal catalysis, fundamentally eliminating the problem of metal residues which is critical for pharmaceutical intermediate synthesis.
Q: What are the typical yields and purity levels achieved with this protocol?
A: The method achieves high yields, with specific examples reaching up to 88%, and consistently delivers product purity exceeding 98% as confirmed by HPLC analysis.
Q: Is this process scalable for industrial production of drug intermediates?
A: Yes, the process operates under mild conditions (room temperature) and uses simple reagents, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha,alpha-Dibromoketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of critical pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN111118529B can be seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical capabilities to guarantee stringent purity specifications for every batch of alpha,alpha-dibromoketones we produce. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry.
We invite you to collaborate with us to leverage this advanced synthetic route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free approach can optimize your supply chain economics. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you secure a sustainable and cost-effective supply of these vital building blocks for your next-generation therapeutics.
