Advanced Photocatalytic Synthesis of Ponatinib for Commercial API Production
Advanced Photocatalytic Synthesis of Ponatinib for Commercial API Production
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for the synthesis of complex oncology therapeutics. Patent CN108047230B introduces a groundbreaking preparation method for Ponatinib, a critical tyrosine kinase inhibitor used in treating Chronic Myelogenous Leukemia. This technical disclosure outlines a novel synthetic route that leverages acridine salt photocatalysts in conjunction with copper salts within an alkaline organic solvent system. By shifting away from traditional thermal activation, this process achieves mild reaction conditions and superior yield profiles. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the supply chain for high-purity API intermediates. The following analysis details the mechanistic advantages and commercial viability of this photocatalytic approach, demonstrating how it addresses key pain points in modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ponatinib has relied heavily on palladium-catalyzed cross-coupling reactions, which present substantial challenges for large-scale manufacturing. Traditional routes often require the use of expensive palladium catalysts such as Pd(PPh3)2Cl2, coupled with rigorous heating conditions that can exceed 80°C. These harsh thermal requirements not only increase energy consumption but also elevate the risk of thermal degradation for sensitive heterocyclic intermediates. Furthermore, the reliance on precious metals introduces significant cost volatility and necessitates complex downstream purification steps to remove trace metal impurities to meet stringent regulatory standards. The cumulative effect of these factors is a higher cost of goods sold (COGS) and potential supply chain bottlenecks, making the conventional synthesis less attractive for high-volume commercial production of this critical anticancer agent.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a visible-light-driven photocatalytic system that fundamentally alters the reaction landscape. By employing specific acridine salt photocatalysts alongside copper salts, the reaction proceeds efficiently at ambient or mild temperatures, eliminating the need for energy-intensive heating. This shift not only drastically reduces the operational expenditure associated with energy but also preserves the structural integrity of the reactants, leading to a cleaner reaction profile. The use of abundant copper salts instead of scarce palladium further decouples the process from precious metal market fluctuations. This innovative approach ensures a more stable and predictable manufacturing process, aligning perfectly with the needs of a reliable agrochemical intermediate supplier or pharmaceutical partner seeking long-term process stability.
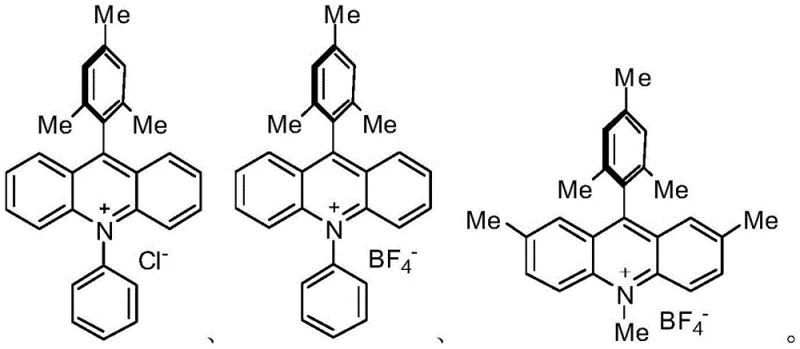
Mechanistic Insights into Acridine Salt Photocatalytic Coupling
The core of this technological advancement lies in the sophisticated interplay between the acridine salt photocatalyst and the copper co-catalyst under visible light irradiation. Upon exposure to light wavelengths ranging from 300nm to 590nm, typically provided by blue LED sources, the acridine salt enters an excited state that facilitates single-electron transfer processes. This photo-excitation activates the copper species, enabling the oxidative addition and reductive elimination cycles necessary for the C-N or C-C bond formation without the high activation energy barriers of thermal methods. The alkaline environment, maintained at a pH between 9 and 12 using bases like diisopropylamine, is crucial for stabilizing the intermediate species and ensuring the forward progress of the substitution reaction. This precise control over the electronic environment allows for high conversion rates while minimizing side reactions that typically plague thermal coupling methods.
Furthermore, the mechanism inherently supports superior impurity control, a critical factor for R&D directors focused on purity and杂质谱 (impurity profiles). The mild conditions prevent the formation of thermal byproducts and decomposition species that are common in high-temperature palladium catalysis. The specific structural configuration of the acridine salts, as illustrated in the patent, ensures high catalytic efficiency even at low molar concentrations, typically ranging from 1:100 to 5:100 relative to the substrate. This efficiency reduces the chemical load in the reaction mixture, simplifying the workup and purification stages. By avoiding the use of transition metals that are difficult to remove, the process naturally yields a product with lower metal contamination, thereby reducing the burden on downstream purification technologies and ensuring compliance with strict ICH guidelines for elemental impurities in pharmaceutical substances.
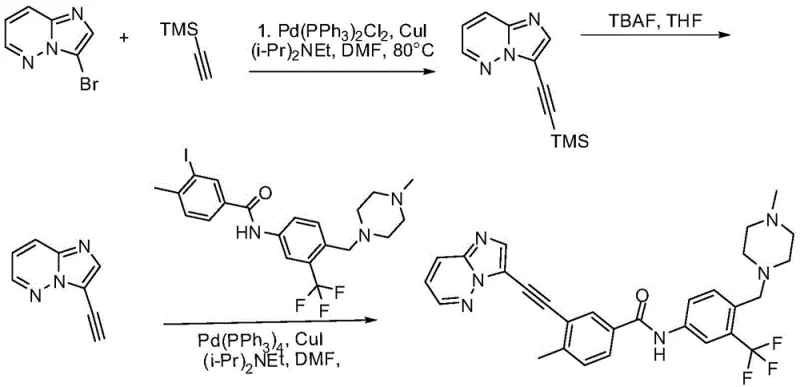
How to Synthesize Ponatinib Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and the maintenance of specific atmospheric conditions. The process begins with the preparation of the key starting materials, including the 3-halo-pyrazolo[1,5-a]pyrimidine derivative and trimethylsilylacetylene, which must be handled under a protective nitrogen atmosphere to prevent oxidation. The reaction system is constructed by dissolving these components in a suitable organic solvent such as acetonitrile or DMF, followed by the addition of the acridine photocatalyst and copper salt. The mixture is then subjected to controlled visible light irradiation for a period ranging from 12 to 48 hours, depending on the specific scale and light intensity. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction system by combining 3-halo-pyrazolo[1,5-a]pyrimidine and trimethylsilylacetylene in an alkaline organic solvent with acridine salt and copper salt.
- Execute visible light irradiation (300nm-590nm) under a protective nitrogen atmosphere to generate the alkynyl intermediate compound.
- Perform desilylation using tetrabutylammonium fluoride, followed by a second photocatalytic coupling with the iodo-aniline derivative to yield Ponatinib.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this photocatalytic method offers tangible strategic benefits beyond mere technical novelty. The primary advantage lies in the significant cost reduction in API manufacturing driven by the elimination of expensive palladium catalysts. By substituting precious metals with abundant copper salts and organic photocatalysts, the raw material costs are substantially lowered, providing a buffer against market volatility. Additionally, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to lower utility costs and a smaller carbon footprint. These factors combine to create a more economically resilient supply chain, ensuring that the production of high-purity oncology intermediates remains financially sustainable even in fluctuating market conditions.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with acridine salts and copper effectively removes one of the most significant cost drivers in traditional cross-coupling reactions. This substitution not only lowers the direct material cost but also reduces the expense associated with metal scavenging and waste treatment. The process operates efficiently with lower catalyst loading, meaning less chemical waste is generated per kilogram of product. Consequently, the overall cost of production is optimized, allowing for more competitive pricing strategies without compromising on the quality or purity of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on palladium often exposes manufacturers to supply risks due to the geopolitical concentration of precious metal mining. By shifting to a copper-based photocatalytic system, the dependency on scarce resources is minimized, enhancing the robustness of the supply chain. The reagents required, such as acridine salts and common copper halides, are readily available from multiple global suppliers, reducing the risk of single-source bottlenecks. This diversification of raw material sources ensures continuous production capability and reduces lead time for high-purity pharmaceutical intermediates, safeguarding against unexpected market disruptions.
- Scalability and Environmental Compliance: The mild conditions of this photocatalytic process make it inherently safer and easier to scale from laboratory to commercial production. The absence of high-temperature and high-pressure requirements simplifies reactor design and reduces safety hazards associated with thermal runaways. Furthermore, the reduced use of heavy metals aligns with increasingly stringent environmental regulations regarding industrial waste discharge. The process generates less hazardous waste, simplifying compliance with environmental standards and reducing the costs associated with waste disposal. This makes the commercial scale-up of complex polymer additives or pharmaceutical intermediates more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent data, providing clarity on process feasibility and advantages. Understanding these details is essential for stakeholders evaluating the integration of this technology into their existing manufacturing portfolios. The insights provided here reflect the consensus on the operational benefits and chemical principles governing this novel approach.
Q: How does the acridine salt photocatalyst improve Ponatinib yield compared to palladium methods?
A: The acridine salt photocatalyst operates under mild visible light conditions, eliminating the need for high-temperature heating required by traditional palladium catalysts. This reduces thermal degradation of sensitive intermediates and significantly improves the overall yield of the target product while lowering catalyst costs.
Q: What are the specific reaction conditions for the photocatalytic coupling step?
A: The reaction requires a protective nitrogen atmosphere and visible light irradiation with a wavelength between 300nm and 590nm, typically using a blue LED source. The system must maintain an alkaline environment (pH 9-12) using bases like diisopropylamine or potassium carbonate to facilitate the copper-assisted photocatalytic cycle.
Q: Is this synthesis method suitable for large-scale commercial manufacturing?
A: Yes, the method is designed for industrial scalability. It uses readily available copper salts instead of expensive precious metals and operates at mild temperatures, which simplifies reactor requirements and safety protocols. The stable yield and controlled impurity profile make it highly viable for commercial scale-up of complex oncology APIs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ponatinib Supplier
The technical potential of this photocatalytic route underscores the importance of partnering with a CDMO expert capable of translating complex patent data into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative synthesis methods are successfully implemented at an industrial level. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Ponatinib or related intermediate meets the highest global standards. We understand the critical nature of oncology APIs and are committed to delivering consistent quality and reliability.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced chemical engineering. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us help you navigate the complexities of modern API synthesis with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
