Advanced Oxalohydrazide Ligands: Revolutionizing C-N Bond Coupling for Industrial Pharmaceutical Manufacturing
Advanced Oxalohydrazide Ligands: Revolutionizing C-N Bond Coupling for Industrial Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving, driven by the need for more sustainable, cost-effective, and efficient methodologies. A significant breakthrough in this domain is documented in patent CN111807986B, which introduces a novel class of oxalohydrazide ligands designed specifically to enhance copper-catalyzed C-N bond coupling reactions. This technology addresses a critical bottleneck in the production of aryl hydrazines, which are vital scaffolds in the development of active pharmaceutical ingredients (APIs) and agrochemicals. By shifting the paradigm from expensive palladium-based systems to robust copper-ligand complexes, this innovation offers a compelling value proposition for global supply chains seeking reliability and economic efficiency without compromising on chemical purity or yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of C-N bonds, particularly for synthesizing aryl hydrazines, has relied heavily on palladium-catalyzed cross-coupling reactions. While effective, these methods suffer from severe economic and logistical drawbacks. Palladium is a precious metal with volatile pricing and limited global availability, making it a significant cost driver in large-scale manufacturing. Furthermore, the removal of trace palladium residues from the final product is a rigorous and expensive process, often requiring specialized scavengers to meet the stringent regulatory limits imposed by health authorities for pharmaceutical products. Alternative copper-catalyzed systems have existed but often lacked the necessary ligand support to ensure high solubility and catalytic turnover, leading to inconsistent yields and the need for harsh reaction conditions that complicate safety protocols.
The Novel Approach
The methodology outlined in the patent presents a transformative solution by utilizing specifically engineered oxalohydrazide ligands. These ligands are synthesized through a straightforward acylation reaction between hydrazine derivatives and oxalyl chloride, a process that is both operationally simple and scalable. 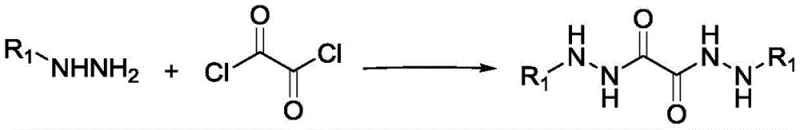 . When employed in conjunction with inexpensive copper salts, these ligands dramatically improve the solubility of the catalyst in the reaction medium. This enhancement allows the coupling of aryl iodides with hydrazine hydrate to proceed efficiently at room temperature, eliminating the need for energy-intensive heating and reducing the formation of thermal degradation byproducts. The result is a streamlined process that delivers high-purity aryl hydrazines with exceptional operational simplicity.
. When employed in conjunction with inexpensive copper salts, these ligands dramatically improve the solubility of the catalyst in the reaction medium. This enhancement allows the coupling of aryl iodides with hydrazine hydrate to proceed efficiently at room temperature, eliminating the need for energy-intensive heating and reducing the formation of thermal degradation byproducts. The result is a streamlined process that delivers high-purity aryl hydrazines with exceptional operational simplicity.
Mechanistic Insights into Copper-Catalyzed C-N Coupling with Oxalohydrazide Ligands
The efficacy of this system lies in the unique coordination chemistry between the oxalohydrazide ligand and the copper center. The ligand acts as a bidentate chelator, stabilizing the copper species and preventing the formation of inactive copper aggregates that typically plague uncatalyzed or poorly ligated copper reactions. This stabilization ensures a consistent concentration of the active catalytic species throughout the reaction cycle, facilitating the oxidative addition of the aryl iodide and the subsequent reductive elimination to form the C-N bond. The mild reaction conditions, typically ranging from 0°C during ligand synthesis to room temperature during coupling, are crucial for maintaining the integrity of sensitive functional groups on the aromatic ring, thereby expanding the scope of compatible substrates for complex molecule synthesis.
From an impurity control perspective, the mechanism offers distinct advantages. The high selectivity of the ligand-copper complex minimizes side reactions such as homocoupling of the aryl halide or over-alkylation of the hydrazine nitrogen. 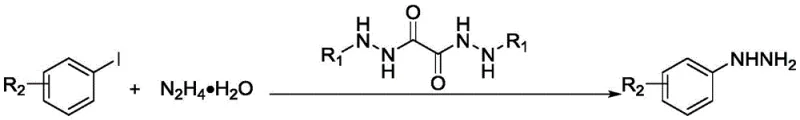 . For instance, the patent data demonstrates successful conversion of iodobenzene to phenylhydrazine with yields exceeding 80%, alongside successful coupling of substituted aryl iodides like 4-iodotoluene and 4-iodobenzonitrile. The ability to tolerate both electron-rich and electron-deficient substituents indicates a robust catalytic cycle that is less susceptible to electronic deactivation, ensuring consistent quality across different batches of pharmaceutical intermediates.
. For instance, the patent data demonstrates successful conversion of iodobenzene to phenylhydrazine with yields exceeding 80%, alongside successful coupling of substituted aryl iodides like 4-iodotoluene and 4-iodobenzonitrile. The ability to tolerate both electron-rich and electron-deficient substituents indicates a robust catalytic cycle that is less susceptible to electronic deactivation, ensuring consistent quality across different batches of pharmaceutical intermediates.
How to Synthesize Oxalohydrazide Ligands Efficiently
The synthesis protocol described in the patent is designed for practical implementation in both laboratory and pilot plant settings. It begins with the preparation of the ligand precursor, such as 1,2-dioxalobenzohydrazide, by reacting benzoylhydrazine with oxalyl chloride in dichloromethane under controlled low-temperature conditions. This step is critical for managing the exothermic nature of the acylation and ensuring high purity of the ligand before it is introduced to the coupling reaction. Once the ligand is isolated, it is combined with copper iodide, a phosphate base, and the aryl iodide substrate in a polar aprotic solvent like DMSO. The detailed standardized synthetic steps for optimizing yield and purity are provided in the guide below.
- Synthesize the oxalohydrazide ligand by reacting hydrazine derivatives with oxalyl chloride in dichloromethane at 0°C, followed by stirring at room temperature.
- Prepare the coupling reaction mixture by combining aryl iodide, hydrazine hydrate, copper catalyst (e.g., CuI), base (e.g., K3PO4), and the synthesized ligand in a polar solvent like DMSO.
- Stir the reaction at room temperature for approximately 12 hours, followed by extraction, drying, and purification via silica gel chromatography to isolate the aryl hydrazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible strategic benefits beyond mere chemical curiosity. The shift from palladium to copper represents a fundamental restructuring of the cost base for C-N coupling processes. By eliminating the dependency on precious metals, manufacturers can insulate their production costs from the volatility of the commodities market. Furthermore, the simplified workup procedures—often requiring only standard extraction and filtration rather than complex metal scavenging—reduce the consumption of auxiliary materials and shorten the overall production cycle time. This efficiency gain directly impacts the throughput capacity of existing manufacturing facilities, allowing for greater output without significant capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with abundant copper salts results in substantial raw material cost savings. Additionally, the ligand itself is derived from commodity chemicals like oxalyl chloride and hydrazine, which are widely available and inexpensive. The mild reaction conditions also lead to lower energy consumption, as there is no need for prolonged heating or cryogenic cooling beyond the initial ligand synthesis step. These factors combine to drastically lower the cost of goods sold (COGS) for aryl hydrazine derivatives, enhancing profit margins for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Reliance on a single source for specialized catalysts can create supply chain vulnerabilities. In contrast, the reagents required for this oxalohydrazide-mediated coupling are bulk chemicals produced by numerous global suppliers. This diversification of the supply base mitigates the risk of shortages and ensures business continuity. The robustness of the reaction also means that slight variations in raw material quality are less likely to cause batch failures, further stabilizing the supply of critical intermediates to pharmaceutical clients who demand just-in-time delivery.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The use of common solvents like DMSO and dichloromethane, coupled with simple aqueous workups, facilitates easy integration into existing multi-purpose chemical plants. From an environmental standpoint, the reduction in heavy metal waste aligns with increasingly stringent green chemistry regulations. Lower copper loading and the absence of palladium simplify wastewater treatment protocols, reducing the environmental footprint and associated compliance costs for the manufacturing facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxalohydrazide ligand technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what potential adopters can expect regarding performance and operational requirements.
Q: What are the primary advantages of using oxalohydrazide ligands over traditional palladium catalysts?
A: The primary advantage is significant cost reduction. Palladium catalysts are notoriously expensive and often require complex removal steps to meet pharmaceutical purity standards. The oxalohydrazide ligand system enables the use of abundant and inexpensive copper salts while maintaining high catalytic efficiency under mild conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights suitability for industrial scale-up. The ligand synthesis uses readily available raw materials like oxalyl chloride and operates under safe, mild conditions (0°C to room temperature). The subsequent coupling reaction avoids harsh conditions, simplifying post-treatment and waste management.
Q: What types of substrates are compatible with this C-N coupling protocol?
A: The method demonstrates broad substrate scope, successfully coupling various aryl iodides including those with electron-donating groups (like methyl) and electron-withdrawing groups (like cyano). This versatility makes it highly valuable for synthesizing diverse pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxalohydrazide Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced ligand technologies like the oxalohydrazide system described in CN111807986B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or intermediate we produce adheres to the highest international standards required by the global pharmaceutical industry.
We invite you to leverage our technical expertise to optimize your synthesis routes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our supply of high-quality oxalohydrazide ligands can drive efficiency and profitability in your aryl hydrazine manufacturing operations.
