Scalable Microchannel Flow Oxidation for High-Purity Ribonic Acid Lactone Intermediates
Scalable Microchannel Flow Oxidation for High-Purity Ribonic Acid Lactone Intermediates
The pharmaceutical industry is constantly seeking robust manufacturing pathways for critical antiviral intermediates, particularly those required for nucleoside analogues like Remdesivir. Patent CN111548328A introduces a transformative approach to synthesizing 2,3,5-tribenzyloxy-D-ribonic acid-1,4-lactone, a pivotal building block in this therapeutic class. By leveraging continuous flow microchannel technology, this method addresses the longstanding inefficiencies of batch processing, offering a pathway that combines high safety standards with exceptional chemical efficiency. The transition from traditional stirred-tank reactors to micro-reactors represents a significant leap forward in process intensification, allowing for precise control over reaction parameters that were previously difficult to manage in bulk systems.
This technical insight report analyzes the strategic value of adopting this flow chemistry protocol for commercial production. For R&D directors, the promise of improved impurity profiles and higher yields offers a compelling case for process redesign. For procurement and supply chain leaders, the ability to run continuous operations translates to better inventory management and reduced lead times. As a reliable pharmaceutical intermediate supplier, understanding these technological shifts is crucial for maintaining competitiveness in the global API market. The following sections detail the mechanistic advantages and commercial implications of this patented oxidation strategy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5-tribenzyloxy-D-ribonic acid-1,4-lactone has been plagued by significant operational drawbacks that hinder efficient large-scale manufacturing. Traditional methods often rely on stoichiometric oxidants like DMSO and acetic anhydride, which necessitate prolonged reaction times extending up to 48 hours. Such extended durations not only tie up reactor capacity but also increase the risk of thermal degradation and byproduct accumulation. Furthermore, the use of DMSO creates substantial downstream processing challenges due to its high boiling point and water solubility, leading to excessive wastewater generation and complex solvent recovery protocols that inflate operational costs.
Alternative batch methods utilizing sodium hypochlorite have also proven suboptimal due to stability issues and inconsistent product quality. In conventional stirred tanks, maintaining uniform mixing and temperature control during exothermic oxidation is difficult, often resulting in incomplete reactions or over-oxidation if the reaction time exceeds narrow windows. To compensate, operators frequently need to supplement oxidants, which introduces variability and complicates the workup procedure. Additionally, methods employing iodine as an oxidant, while effective chemically, impose a heavy financial burden due to the high cost of raw materials, making them economically unviable for cost-sensitive commercial applications.
The Novel Approach
The innovative method disclosed in the patent utilizes a continuous flow microchannel reactor to overcome these inherent limitations of batch processing. By confining the reaction to micro-scale channels, the system achieves rapid and uniform mixing of the substrate, catalyst, and oxidant streams, ensuring that every molecule experiences identical reaction conditions. This precision allows the oxidation to proceed with remarkable speed, reducing the residence time from hours to merely 15-120 seconds. The drastic reduction in reaction time not only boosts throughput but also minimizes the exposure of the sensitive intermediate to potentially degrading conditions, thereby preserving product integrity.
Moreover, the continuous flow setup facilitates immediate quenching of the reaction mixture upon exiting the reactor, effectively halting the oxidation at the desired lactone stage without further degradation. This capability eliminates the need for excessive oxidant supplementation and stabilizes the product quality, yielding consistent batches with purity exceeding 99%. The simplicity of the post-treatment process, involving straightforward liquid separation and concentration, further streamlines the workflow. This novel approach transforms a cumbersome batch operation into a sleek, continuous manufacturing line that is inherently safer, more economical, and environmentally friendlier.
Mechanistic Insights into TEMPO-Catalyzed Flow Oxidation
The core of this synthetic breakthrough lies in the efficient TEMPO-catalyzed oxidation mechanism facilitated by the unique hydrodynamics of the microchannel reactor. In this system, 2,3,5-tribenzyloxy-D-ribose serves as the substrate, which is oxidized by sodium hypochlorite in the presence of a nitroxyl radical catalyst such as TEMPO or 4-hydroxy TEMPO. The microchannel environment ensures that the mass transfer limitations often seen in biphasic systems are virtually eliminated. The high surface-to-volume ratio of the channels allows for instantaneous heat exchange, keeping the reaction temperature strictly within the optimal 0-20°C range, which is critical for controlling the selectivity of the oxidation and preventing side reactions.
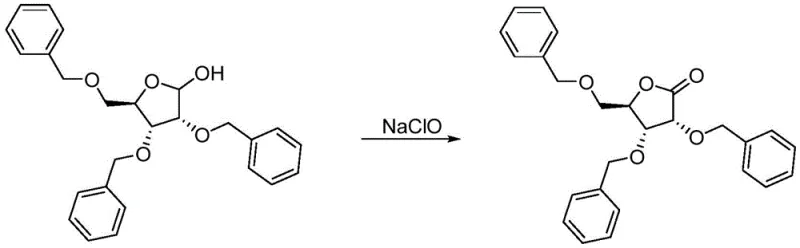
Potassium bromide acts as a crucial co-catalyst in this cycle, accelerating the generation of the active oxoammonium species from the TEMPO radical. The precise metering of the KBr stream alongside the substrate and oxidant streams ensures that the catalytic cycle operates at peak efficiency without excess reagent waste. The rapid consumption of the oxidant within the channel means that hazardous chlorine species do not accumulate, significantly enhancing process safety. This controlled mechanistic pathway results in a clean conversion where the primary alcohol is selectively oxidized to the carboxylic acid, which subsequently cyclizes to form the stable 1,4-lactone ring with minimal formation of chlorinated byproducts or over-oxidized impurities.
How to Synthesize 2,3,5-Tribenzyloxy-D-Ribonic Acid-1,4-Lactone Efficiently
Implementing this continuous flow protocol requires careful preparation of three distinct feed streams to ensure optimal reaction kinetics and safety. The process begins with the conditioning of the oxidant stream, where sodium hypochlorite is diluted and pH-adjusted to a narrow alkaline window to stabilize the active species. Simultaneously, the organic phase containing the ribose derivative and the nitroxyl catalyst is prepared in a compatible solvent such as dichloromethane or toluene. A third aqueous stream containing the bromide co-catalyst is also prepared. These streams are then pumped into the microchannel reactor using high-precision metering pumps to maintain the strict stoichiometric ratios required for high conversion.
- Prepare Material A by adjusting the pH of a 3-12% NaClO solution to 8.2-9.2 using acid or alkali at temperatures below 10°C.
- Prepare Material B by dissolving 2,3,5-tribenzyloxy-D-ribose and a catalytic amount of TEMPO or 4-hydroxy TEMPO in an organic solvent like dichloromethane or toluene.
- Prepare Material C as an aqueous KBr solution and pump all three streams into a microchannel reactor at 0-20°C for a residence time of 15-120 seconds.
- Quench the reaction mixture immediately upon exit using sodium thiosulfate or sodium bisulfite, followed by liquid separation and concentration to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this microchannel flow technology offers profound strategic benefits that extend beyond simple chemical yield improvements. The shift from batch to continuous processing fundamentally alters the cost structure of manufacturing this key intermediate. By eliminating the need for expensive stoichiometric oxidants like iodine and reducing the reliance on difficult-to-remove solvents like DMSO, the raw material costs are significantly lowered. The simplified workup procedure reduces the consumption of auxiliary chemicals and utilities, such as water and energy for distillation, contributing to a leaner and more cost-effective production model.
- Cost Reduction in Manufacturing: The transition to a continuous flow system drastically simplifies the production workflow, removing the need for large reactor vessels and the associated cleaning and validation cycles between batches. The high efficiency of the TEMPO/NaClO system means that raw materials are utilized more effectively, with minimal waste generation. Furthermore, the ability to operate at ambient or slightly cooled temperatures reduces the energy load required for heating or deep cooling, leading to substantial operational expenditure savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently provides a more stable and predictable output compared to batch processing, which is susceptible to variability between runs. The steady-state operation of the microchannel reactor ensures a consistent supply of high-purity intermediate, reducing the risk of stockouts or quality deviations that could disrupt downstream API synthesis. This reliability is crucial for meeting the rigorous demands of pharmaceutical clients who require uninterrupted supply chains for their own production schedules.
- Scalability and Environmental Compliance: Scaling this process does not require building larger, more dangerous reactors; instead, capacity is increased by running the units longer or numbering up the reactor modules. This modular approach minimizes capital investment risks and allows for flexible production adjustments based on market demand. Additionally, the reduced solvent usage and minimized waste generation align with increasingly stringent environmental regulations, positioning the manufacturer as a sustainable partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this flow oxidation technology. These insights are derived directly from the patent data and practical considerations for scaling such processes in a GMP environment. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains.
Q: How does the microchannel reactor improve safety compared to traditional batch oxidation?
A: The microchannel reactor significantly enhances safety by minimizing the hold-up volume of reactive intermediates and providing superior heat transfer capabilities. This precise thermal control prevents local overheating and runaway exotherms often associated with bulk hypochlorite oxidations, ensuring a stable and safe production environment.
Q: What purity levels can be achieved with this continuous flow method?
A: This optimized flow process consistently achieves product purity exceeding 99% with yields greater than 95%. The short residence time and efficient mixing prevent over-oxidation and byproduct formation, resulting in a cleaner crude product that requires less intensive downstream purification.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the continuous nature of the microchannel technology allows for seamless scale-up through numbering-up strategies rather than increasing vessel size. This maintains the same reaction efficiency and safety profile at commercial volumes, making it ideal for the sustained supply of key antiviral intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Tribenzyloxy-D-Ribonic Acid-1,4-Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving antiviral therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,3,5-tribenzyloxy-D-ribonic acid-1,4-lactone meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for this essential intermediate. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced continuous flow capabilities can drive efficiency and reliability in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
