Optimizing Phenylsulfanylphenyl Piperidine Production for Commercial Scale
Optimizing Phenylsulfanylphenyl Piperidine Production for Commercial Scale
The pharmaceutical industry continuously seeks robust manufacturing routes for complex heterocyclic scaffolds, particularly those serving as critical intermediates for central nervous system therapeutics. Patent CN101959859B discloses a groundbreaking process for the manufacture of substituted phenylsulfanylphenyl-piperidines, specifically leveraging an N-benzyl protecting group strategy to overcome historical synthesis bottlenecks. This technical advancement addresses the urgent need for methods that are not only chemically efficient but also economically viable for large-scale production. By shifting away from traditional protecting groups that require extreme cryogenic conditions, this innovation facilitates higher overall yields and simplifies the isolation of key intermediates through crystallization. For R&D and supply chain leaders, understanding this methodology is essential for securing a reliable phenylsulfanylphenyl piperidine supplier capable of meeting stringent quality and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-[2-(4-methylphenylsulfanyl)phenyl]-piperidine and its analogs relied heavily on N-tert-butoxycarbonyl (Boc) or N-ethoxycarbonyl protecting groups, which present significant operational challenges. Prior art methods, such as those disclosed in WO 2007/144006, necessitate reaction temperatures as low as -40°C or even -78°C to control reactivity and prevent side reactions. These extreme cryogenic conditions impose a heavy burden on manufacturing infrastructure, requiring specialized cooling equipment and increasing energy consumption substantially. Furthermore, intermediates protected with these groups often exist as oils or amorphous solids that are difficult to purify, frequently requiring chromatographic separation which is impractical and cost-prohibitive at a commercial scale. The cumulative effect of low yields, difficult purification, and harsh conditions renders these conventional routes suboptimal for the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative process detailed in the patent data introduces the use of an optionally substituted benzyl group as the N-piperidine protecting moiety, fundamentally altering the reaction profile. This strategic modification allows the critical lithiation and addition steps to be conducted at significantly higher temperatures, typically around -15°C, which is much more accessible and manageable in standard industrial reactors. The N-benzyl group imparts favorable physical properties to the intermediates, enabling them to form stable crystalline salts, such as hydrochlorides, which can be isolated via simple filtration. This shift from difficult-to-isolate oils to crystalline solids represents a paradigm shift in process chemistry, ensuring higher purity and reduced processing time. By adopting this novel approach, manufacturers can achieve a more streamlined workflow that aligns with the requirements for commercial scale-up of complex pharmaceutical intermediates.
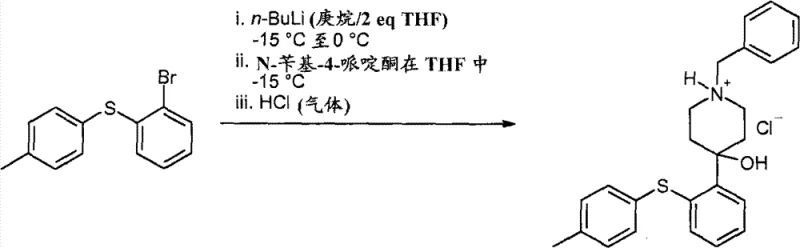
Mechanistic Insights into N-Benzyl Protected Piperidine Synthesis
The core of this synthetic strategy involves the metalation of a substituted phenylsulfanylphenyl bromide followed by nucleophilic addition to an N-benzyl-4-piperidone. The choice of the benzyl group is not arbitrary; it stabilizes the resulting alkoxide intermediate sufficiently to allow warming to room temperature without significant decomposition, a feat not achievable with more labile protecting groups. The reaction mechanism proceeds through the formation of an aryl lithium species at controlled low temperatures, which then attacks the carbonyl carbon of the piperidone. The resulting tertiary alcohol intermediate, protected by the benzyl group, exhibits enhanced stability and crystallinity. This mechanistic robustness is critical for maintaining high-purity phenylsulfanylphenyl piperidine standards, as it minimizes the formation of byproducts that typically arise from thermal instability or competing side reactions during the quench and workup phases.
Impurity control is further enhanced by the specific crystallization behavior of the benzyl-protected intermediates. The patent data highlights that the hydrochloride salt of the intermediate can be precipitated from solution using antisolvents like diethyl ether, effectively trapping the desired compound in a solid lattice while leaving impurities in the mother liquor. This crystallization-driven purification is far superior to extraction or distillation for removing structurally similar byproducts. Additionally, the final deprotection step utilizes triethylsilane and trifluoroacetic acid, a mild yet effective system that cleaves the benzyl group without compromising the integrity of the sensitive sulfanyl linkage. This careful balance of reactivity ensures that the final API intermediate meets the rigorous impurity profiles required by regulatory bodies, thereby reducing the risk of batch rejection.
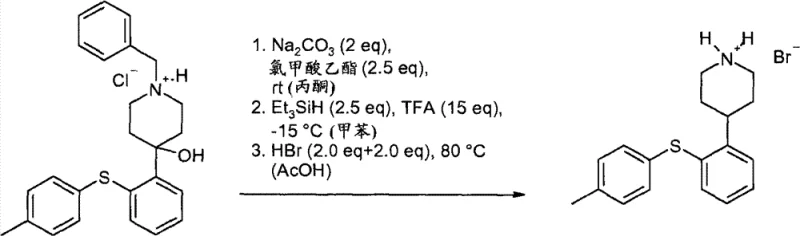
How to Synthesize Phenylsulfanylphenyl Piperidine Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the N-benzyl strategy. The process begins with the preparation of the aryl bromide precursor, followed by the critical lithiation step where temperature management is key to ensuring complete metalation without decomposition. Subsequent addition to the N-benzyl-4-piperidone must be performed with strict stoichiometric control to minimize excess reagents that could complicate downstream purification. The workup procedure leverages pH adjustments and solvent swaps to isolate the intermediate as a crystalline hydrochloride salt, which serves as a pivotal control point for quality assurance. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during technology transfer.
- Prepare 2-(4-methylphenylsulfanyl)-phenyl bromide via nucleophilic substitution.
- Perform lithiation at -15°C and react with N-benzyl-4-piperidone to form the protected intermediate.
- Execute deprotection and reduction using triethylsilane and acid to obtain the final piperidine salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this N-benzyl protection methodology offers substantial cost savings and supply chain resilience. The ability to operate at -15°C rather than -78°C drastically reduces the energy load and capital expenditure associated with cryogenic cooling systems, directly impacting the operational cost structure. Furthermore, the high yields reported in the patent examples, often exceeding 90% for the intermediate formation, mean that less raw material is required to produce the same amount of final product, optimizing the cost of goods sold. The elimination of chromatographic purification in favor of crystallization not only speeds up the cycle time but also reduces the consumption of expensive solvents and silica media. These factors combine to create a manufacturing process that is inherently more economical and sustainable, addressing the critical need for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The transition to N-benzyl protection eliminates the need for expensive and hazardous reagents like tributyltin hydride used in older reduction methods, replacing them with more cost-effective silanes. By avoiding the extreme low temperatures of -78°C, facilities can utilize standard jacketed reactors rather than specialized cryogenic vessels, significantly lowering capital and maintenance costs. The high yield of the crystallization step minimizes material loss, ensuring that the maximum amount of starting material is converted into saleable product. This efficiency translates into a more competitive pricing structure for the final high-purity phenylsulfanylphenyl piperidine, allowing procurement teams to negotiate better terms.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions reduces the likelihood of batch failures due to temperature excursions or reagent instability. Since the intermediates are stable crystalline solids, they can be stored and transported with greater ease compared to unstable oils, providing flexibility in inventory management. This stability ensures a continuous supply of key intermediates, mitigating the risk of production delays that can ripple through the entire pharmaceutical supply chain. Reliable availability of these materials is crucial for maintaining the production schedules of downstream API manufacturers, making this process a strategic asset for supply chain heads.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common solvents like heptane, THF, and toluene which are readily available in bulk quantities. The reduction in solvent usage due to efficient crystallization lowers the volume of chemical waste generated, simplifying waste treatment and disposal compliance. Additionally, the avoidance of heavy metal catalysts or toxic tin reagents aligns with modern green chemistry principles and regulatory expectations for environmental safety. This makes the technology transfer to large-scale production facilities smoother and less burdened by environmental permitting hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific advantages and operational details outlined in the patent documentation to provide clarity for technical decision-makers. Understanding these nuances is vital for evaluating the feasibility of adopting this method for your specific production needs. The answers reflect the consensus on the superior performance of the N-benzyl strategy over conventional alternatives.
Q: Why is the N-benzyl protecting group superior for this synthesis?
A: The N-benzyl group allows the reaction to proceed at significantly higher temperatures (-15°C vs -78°C) and enables crystallization of intermediates, drastically improving yield and purity compared to N-carboethoxy groups.
Q: What are the impurity control advantages of this method?
A: The benzyl-protected intermediates form crystalline hydrochloride salts that are easily isolated by filtration, removing soluble impurities without the need for complex chromatography.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the elimination of cryogenic requirements and the robustness of the crystallization steps make this route highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylsulfanylphenyl Piperidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex pharmaceutical intermediates like phenylsulfanylphenyl piperidines. Our CDMO expertise allows us to adapt advanced patent technologies, such as the N-benzyl protection strategy, to meet the rigorous demands of global pharmaceutical clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest industry standards for quality and safety.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through the adoption of this superior synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can support your long-term production goals. Let us be your partner in transforming innovative chemistry into commercial success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
