Advanced Synthesis of Oxaagolide Intermediates: Enhancing Purity and Scalability for Global API Production
The pharmaceutical landscape for treating endometriosis has evolved significantly with the advent of Gonadotropin-Releasing Hormone (GnRH) antagonists, among which oxaagolide sodium salt represents a critical therapeutic advancement. However, the commercial viability of such potent active pharmaceutical ingredients (APIs) is intrinsically linked to the efficiency and robustness of their synthetic routes. Patent CN110835302A discloses a groundbreaking preparation method for the key intermediate of oxaagolide, specifically targeting the synthesis of Compound 5 and its derivatives. This intellectual property addresses long-standing bottlenecks in the manufacturing of this high-value pharmaceutical intermediate, offering a pathway that diverges sharply from traditional, hazard-prone methodologies. By leveraging a continuous reaction strategy that eliminates the isolation of volatile intermediates, this technology not only enhances operational safety but also drastically improves the overall yield and purity profile. For global supply chain stakeholders, understanding the nuances of this patent is essential, as it signals a shift towards more sustainable and cost-effective production capabilities for complex gynecological therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
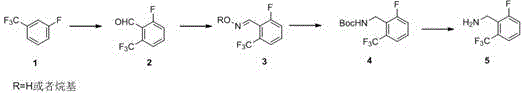
Historically, the synthesis of oxaagolide intermediates has been plagued by significant technical and safety challenges that hinder efficient commercial scale-up. Prior art methods, such as those disclosed in earlier patents, often rely on the use of borane (BH3) as a reducing agent or zinc powder for reduction steps, which introduces substantial safety risks due to the explosive nature and hazardous waste generation associated with these reagents. Furthermore, conventional routes typically involve a stepwise synthesis where the aldehyde intermediate (Compound 2) must be isolated; however, this compound possesses a very low boiling point, making the separation process technically difficult and leading to considerable material loss during distillation or purification. Additionally, direct catalytic hydrogenation of the oxime intermediate in traditional processes often results in the uncontrolled formation of Impurity A, a dimeric byproduct that compromises the quality of the final API and necessitates costly downstream purification steps to meet regulatory standards.
The Novel Approach
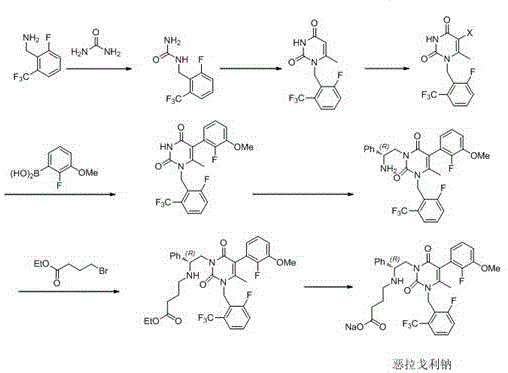
In stark contrast to these legacy methods, the novel approach detailed in CN110835302A introduces a telescoped continuous reaction sequence that fundamentally reengineers the production workflow. This innovative strategy allows for the efficient synthesis of Compound 3 directly from the reaction solution of Compound 2, completely bypassing the need for isolating the volatile aldehyde intermediate. By maintaining the reaction mixture in a continuous flow, the process minimizes exposure to air and moisture, thereby reducing degradation and loss. Moreover, the integration of Boc anhydride during the hydrogenation step serves as a critical process control measure, effectively capping the amine functionality as it forms and preventing the dimerization that leads to Impurity A. This methodological shift not only simplifies the operational complexity but also aligns with green chemistry principles by reducing solvent usage and waste generation, presenting a compelling value proposition for manufacturers seeking to optimize their production lines.
Mechanistic Insights into Boc-Protected Catalytic Hydrogenation
The core chemical innovation of this patent lies in the precise manipulation of reaction conditions to control selectivity during the reduction phase. The synthesis begins with the regioselective lithiation of 3-fluoro-trifluoromethylbenzene using n-butyllithium at cryogenic temperatures ranging from -60 to -80°C, ensuring that the lithiation occurs specifically at the ortho-position relative to the fluorine atom. This intermediate is then quenched with DMF to generate the aldehyde in situ, which is immediately reacted with hydroxylamine hydrochloride in the presence of a mild base like sodium bicarbonate to form the oxime (Compound 3). The critical mechanistic breakthrough occurs in the subsequent hydrogenation step, where Raney nickel is employed as the catalyst under a hydrogen pressure of 0.2 to 0.5 MPa. Unlike standard reductions, the presence of di-tert-butyl dicarbonate (Boc anhydride) in the reaction mixture ensures that the nascent amine is protected instantaneously as a carbamate (Compound 4), which sterically and electronically hinders the nucleophilic attack required to form the dimeric Impurity A.
Beyond the control of Impurity A, the process also demonstrates an unexpected ability to suppress the formation of Impurity B, a structural analog that shares similar physicochemical properties with the target product and is notoriously difficult to remove via crystallization. The specific solvent system, utilizing methanol or ethanol, combined with the controlled temperature range of 20 to 40°C during the hydrogenation, creates an environment that favors the formation of the desired mono-amine product over side reactions. The resulting Compound 4 can then be deprotected under acidic conditions to yield Compound 5 with exceptional purity, often exceeding 99.5% as confirmed by HPLC analysis. This high level of impurity control is paramount for R&D directors, as it reduces the burden on analytical quality control and ensures that the final API meets the stringent impurity profiles required by regulatory bodies like the FDA and EMA for human use.
How to Synthesize Oxaagolide Intermediate Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent stoichiometry to maximize the benefits of the continuous process. The procedure begins with the preparation of the organolithium species, followed by the telescoped formylation and oximation steps which should be monitored via HPLC to ensure complete conversion before proceeding to the hydrogenation stage. The hydrogenation step is the critical control point where the Boc protection strategy must be executed precisely to avoid impurity formation. For detailed operational parameters, including specific stirring rates, addition times, and workup procedures, please refer to the standardized synthesis steps provided below which outline the exact protocol for achieving the reported yields and purity levels.
- Perform regioselective lithiation of 3-fluoro-trifluoromethylbenzene at -60 to -80°C followed by formylation with DMF to generate the aldehyde intermediate in situ.
- React the crude aldehyde solution directly with hydroxylamine hydrochloride and sodium bicarbonate to form the oxime derivative without isolation.
- Execute catalytic hydrogenation using Raney nickel in the presence of Boc anhydride to simultaneously reduce the oxime and protect the amine, yielding the target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial strategic advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for pharmaceutical intermediates. The elimination of hazardous reducing agents like borane not only reduces the cost associated with specialized safety equipment and waste disposal but also mitigates the risk of production stoppages due to safety incidents. Furthermore, the continuous nature of the reaction significantly enhances throughput capabilities, allowing manufacturers to respond more agilely to market demand fluctuations without the need for extensive batch processing times. This efficiency translates into a more resilient supply chain, where the risk of delays caused by complex purification steps or low-yield isolations is drastically minimized, ensuring a steady flow of high-quality intermediates to downstream API production facilities.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by replacing expensive and hazardous reagents with more economical alternatives such as Raney nickel and Boc anhydride. By eliminating the isolation step for the volatile aldehyde intermediate, the method reduces solvent consumption and energy costs associated with distillation, leading to substantial overall savings in the cost of goods sold. Additionally, the high yield and purity reduce the need for reprocessing or discarding off-spec material, further enhancing the economic viability of the manufacturing process.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to a more reliable supply chain by minimizing the variables that typically cause production delays. The use of commercially available starting materials and standard catalytic hydrogenation equipment ensures that the process can be easily replicated across different manufacturing sites without the need for specialized or hard-to-source technology. This standardization reduces the lead time for high-purity pharmaceutical intermediates, allowing procurement teams to secure inventory with greater confidence and shorter notification periods.
- Scalability and Environmental Compliance: The method is inherently designed for commercial scale-up, with reaction conditions that are easily manageable in large-scale reactors. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the compliance burden on manufacturing facilities. This environmental compatibility ensures long-term operational sustainability, protecting the supply chain from potential regulatory shutdowns or fines associated with non-compliant waste management practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a reliable foundation for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new process control Impurity A formation during hydrogenation?
A: The process introduces Boc anhydride during the catalytic hydrogenation step. This immediately protects the newly formed amine group, preventing it from reacting with unreacted intermediates to form the dimeric Impurity A, thereby significantly enhancing product purity.
Q: What are the safety advantages of this method over prior art?
A: Unlike conventional methods that rely on hazardous borane reducing agents or zinc powder, this novel route utilizes catalytic hydrogenation with Raney nickel. This eliminates the need for explosive reagents and reduces the generation of hazardous waste, ensuring a safer operational environment.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method is designed for continuous reaction without the need to isolate the volatile aldehyde intermediate (Compound 2). This telescoped approach minimizes material loss, simplifies operations, and is highly conducive to industrial amplification from kilogram to metric ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxaagolide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the production of high-value pharmaceutical intermediates like those used in oxaagolide synthesis. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110835302A are realized in practical, large-scale operations. Our facility is equipped with state-of-the-art rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for global API manufacturing. We are committed to translating complex chemical innovations into reliable commercial supply solutions.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can optimize your supply chain. Whether you require pilot-scale validation or full commercial production, our team is ready to support your project with the technical depth and operational excellence necessary to bring next-generation therapeutics to market efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
