Advanced Manufacturing of High-Purity Carvone via Manganese Dioxide Oxidation for Global Flavor Markets
The global demand for high-purity terpenes, particularly carvone, continues to surge across the flavor, fragrance, and pharmaceutical sectors, driving the need for more efficient and environmentally sustainable synthesis routes. Patent CN101613263B introduces a transformative methodology that addresses critical bottlenecks in traditional carvone manufacturing by utilizing activated or freshly prepared manganese dioxide (MnO2) as a robust oxidizing agent. This technical breakthrough shifts the paradigm from complex, hazardous hydrolysis or hydrogenation processes to a streamlined oxidation protocol that delivers exceptional purity levels exceeding 99% with impressive yields. For R&D directors and process chemists, this patent represents a significant opportunity to optimize synthetic pathways, offering a route that minimizes toxic byproducts while maximizing atom economy through the direct conversion of carvone oxime. The strategic implementation of this technology allows manufacturers to align with increasingly stringent environmental regulations while maintaining competitive pricing structures in the volatile fine chemicals market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of carvone has been plagued by significant technical and environmental challenges inherent to legacy synthesis strategies. One predominant method involves the acid-catalyzed hydrolysis of carvone oxime using acetone, a process that is notoriously inefficient due to low single-step yields and the generation of substantial hazardous waste streams. This conventional approach produces large quantities of waste acid salts that require costly neutralization and disposal, alongside the formation of acetone oxime, a compound suspected of having carcinogenic properties, which poses severe safety and regulatory risks for production facilities. Alternatively, another established route employs the hydrogenation of carvone oxime using poisoned catalysts; however, this method suffers from extreme complexity in product purification and relies on expensive catalytic systems that degrade over time, leading to inconsistent batch quality and inflated operational expenditures. These traditional pathways create substantial barriers to entry for new manufacturers and impose heavy compliance burdens on existing supply chains, necessitating a urgent transition toward greener, more reliable chemical technologies.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel approach detailed in the patent utilizes a heterogeneous oxidation strategy driven by activated manganese dioxide, offering a remarkably simple yet powerful solution for carvone synthesis. By heating and refluxing carvone oxime with activated or freshly prepared MnO2, the reaction proceeds smoothly to yield carvone directly, bypassing the formation of dangerous nitrogenous byproducts and eliminating the need for corrosive acidic conditions. This method leverages the unique oxidative properties of activated MnO2 to selectively transform the oxime functionality into the desired ketone without affecting other sensitive groups within the terpene skeleton, ensuring high chemical fidelity. The operational simplicity of this route, which typically operates within a moderate temperature range of 0°C to 100°C, allows for straightforward scale-up using standard reactor configurations without the need for specialized high-pressure hydrogenation equipment. Furthermore, the solid nature of the oxidant facilitates easy separation via simple filtration, drastically reducing the complexity of the workup procedure and enabling a continuous flow potential that modern manufacturing demands.
Mechanistic Insights into MnO2-Mediated Oxidation of Oximes
The core of this technological advancement lies in the specific mechanistic interaction between the activated manganese dioxide surface and the carvone oxime substrate, which facilitates a clean dehydrogenation process. Activated MnO2 acts as a mild yet effective oxidizing agent that abstracts hydrogen atoms from the oxime group, promoting the elimination of water or hydroxylamine equivalents to form the carbonyl double bond characteristic of the ketone product. The activation of the manganese dioxide, often achieved through treatment with dilute organic or inorganic acids followed by thermal drying, increases the surface area and reactivity of the reagent, ensuring rapid kinetics even at reflux temperatures in solvents like cyclohexane or carbon tetrachloride. This surface-mediated reaction mechanism minimizes side reactions such as over-oxidation or skeletal rearrangement, which are common pitfalls in liquid-phase oxidations using harsher reagents like chromic acid or permanganate. The heterogeneity of the system further aids in controlling the reaction exotherm and prevents the accumulation of reactive intermediates that could lead to polymerization or degradation of the valuable terpene backbone.
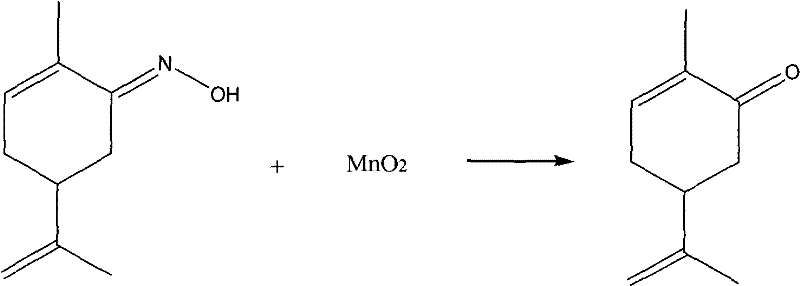
From an impurity control perspective, this mechanism offers distinct advantages by inherently avoiding the generation of acetone oxime, a persistent contaminant in acid hydrolysis routes that is difficult to separate and poses toxicological concerns. The selectivity of the MnO2 oxidation ensures that the isopropenyl double bond remains intact, preserving the stereochemical integrity required for high-value applications in flavor and fragrance formulations where specific enantiomers like L-carvone are prized. By operating under neutral to slightly acidic conditions generated in situ or using non-protic solvents, the process mitigates the risk of acid-catalyzed hydration of the alkene moiety, which would otherwise lead to diol impurities that compromise the sensory profile of the final product. This high level of chemoselectivity translates directly into reduced downstream purification costs, as the crude reaction mixture requires fewer distillation cuts to achieve pharmaceutical or food-grade specifications, thereby enhancing the overall process mass intensity.
How to Synthesize Carvone Efficiently
To implement this superior synthesis route effectively, manufacturers must adhere to precise protocols regarding the preparation of the oxidant and the management of reaction parameters to ensure consistent reproducibility. The process begins with the critical activation of commercial manganese dioxide, which may involve washing with dilute sulfuric or acetic acid followed by rigorous drying at temperatures between 120°C and 125°C to generate the active species necessary for efficient oxidation. Once the activated reagent is prepared, it is suspended in a suitable organic solvent such as cyclohexane, and the carvone oxime substrate is introduced under stirring to ensure homogeneous contact before heating the mixture to reflux for a duration of approximately 3 to 4 hours. Monitoring the reaction progress is essential, typically achieved through chromatographic analysis to confirm the complete disappearance of the starting oxime, after which the mixture is cooled and filtered to remove the spent manganese residues. The detailed standardized synthesis steps, including specific molar ratios and safety precautions for handling solvents and reagents, are outlined in the guide below.
- Preparation of Activated Manganese Dioxide: Treat commercial manganese dioxide with dilute acid (such as sulfuric or acetic acid), wash to neutrality, dry at 120-125°C, and grind to a fine powder.
- Oxidation Reaction: Suspend the activated manganese dioxide in a suitable solvent (e.g., cyclohexane), add carvone oxime, and heat the mixture to reflux for 3-4 hours until the starting material is consumed.
- Workup and Purification: Cool the reaction mixture, filter off the solid manganese residues, and subject the filtrate to vacuum rectification to isolate carvone with purity exceeding 99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this MnO2-based oxidation technology presents a compelling value proposition centered on cost stability, raw material availability, and operational efficiency. Unlike processes reliant on precious metal catalysts or specialized high-pressure infrastructure, this method utilizes manganese dioxide, a commodity chemical that is abundant, inexpensive, and widely available from multiple global suppliers, thereby insulating the supply chain from geopolitical volatility and price spikes associated with rare earth elements. The elimination of complex catalyst recovery systems and the reduction in hazardous waste generation significantly lower the total cost of ownership for the manufacturing facility, allowing for more competitive pricing models in the final market. Additionally, the robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment maintenance or stringent safety shutdowns, ensuring a more reliable delivery timeline for downstream customers in the flavor and pharmaceutical industries.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive, single-use catalysts with low-cost manganese dioxide, which drastically reduces the direct material cost per kilogram of product. Since the oxidant is heterogeneous, it can be easily separated by filtration, eliminating the need for costly and time-consuming extraction or chromatography steps often required to remove soluble metal contaminants from the product stream. Furthermore, the waste manganese residues generated can potentially be sold or regenerated for other industrial applications, creating an additional revenue stream or offsetting disposal costs, which contributes to a leaner and more profitable production model. The simplification of the workflow also reduces labor hours and energy consumption associated with extended reaction times or complex purification sequences, compounding the financial advantages over traditional methods.
- Enhanced Supply Chain Reliability: By relying on a chemistry that uses universally available reagents and standard glass-lined or stainless steel reactors, manufacturers can diversify their supplier base and reduce dependency on single-source vendors for specialized catalysts. The operational simplicity of the reflux and filtration steps minimizes the risk of batch failures due to equipment malfunction or operator error, leading to higher first-pass yields and more predictable inventory levels. This reliability is crucial for maintaining just-in-time delivery commitments to major multinational clients who require consistent quality and volume, thereby strengthening long-term contractual relationships and market share. The ability to rapidly scale production up or down in response to market demand without retooling expensive infrastructure provides a strategic agility that is highly valued in the fast-moving consumer goods sector.
- Scalability and Environmental Compliance: The environmental profile of this synthesis route aligns perfectly with modern sustainability goals, as it avoids the discharge of heavy metal effluents and reduces the volume of acidic wastewater that requires neutralization. The solid waste generated is chemically stable and easier to handle than liquid sludge, simplifying compliance with increasingly strict environmental protection regulations and reducing the liability associated with hazardous waste storage and transport. From a scalability standpoint, the exothermic nature of the oxidation is manageable under reflux conditions, allowing for safe operation in large-volume reactors without the risk of thermal runaway that can plague other oxidation chemistries. This combination of environmental stewardship and engineering safety makes the process highly attractive for expansion into new geographic markets where regulatory hurdles for chemical manufacturing are becoming progressively more stringent.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese dioxide oxidation technology for carvone production. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios and assess the potential return on investment. The answers reflect the consensus on the operational benefits and chemical advantages observed during the development and validation of this specific synthetic pathway.
Q: What are the primary advantages of using activated MnO2 over acid hydrolysis for carvone production?
A: The activated MnO2 method avoids the formation of suspected carcinogenic byproducts like acetone oxime and eliminates the generation of large quantities of waste acid salts, resulting in a cleaner process with easier downstream purification compared to traditional acid hydrolysis.
Q: How does this oxidation method impact the overall production cost?
A: This method significantly reduces costs because manganese dioxide is an inexpensive and readily available reagent. Furthermore, the solid waste generated can often be sold or regenerated, minimizing disposal costs and enhancing the economic viability of large-scale manufacturing.
Q: Is this process suitable for industrial scale-up?
A: Yes, the process is highly scalable due to its operational simplicity, involving standard reflux and filtration steps without the need for complex high-pressure equipment or expensive poisoned catalysts, making it ideal for commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carvone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies like the MnO2 oxidation route to meet the evolving demands of the global flavor and fragrance industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of this oxidation chemistry, including the safe handling of activated reagents and the rigorous purification steps necessary to achieve stringent purity specifications. With our dedicated rigorous QC labs, we guarantee that every batch of carvone meets the highest international standards for identity, potency, and impurity profiles, providing our partners with the confidence they need to launch successful products.
We invite you to collaborate with us to leverage this innovative technology for your specific application needs, whether for natural-identical flavorings or pharmaceutical intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient synthesis route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-quality carvone for your business growth.
