Advanced Carboline Compound Synthesis for Commercial Scale-Up of Complex Organic Materials
The landscape of advanced organic materials is constantly evolving, driven by the need for compounds with precise stereochemistry and unique physical properties. Patent CN101157694B introduces a significant breakthrough in this domain by disclosing the synthesis and application of two novel carboline compounds, specifically epimers designated as Compound 1 and Compound 2. These molecules are not merely structural variants; they exhibit distinct nano-scale crystal structures, including surface protrusions and bud-shaped morphologies, which are critical for high-performance applications in fields such as optoelectronics and specialized organic semiconductors. The technical disclosure provides a robust pathway for generating these high-value intermediates from abrine, a naturally derived amino acid derivative, ensuring that the production process is grounded in accessible chemistry while delivering exceptional material properties. For R&D directors and procurement specialists, this patent represents a viable route to acquiring high-purity organic materials that possess the structural complexity required for next-generation device fabrication.
Furthermore, the methodology outlined in the patent emphasizes strict control over reaction conditions, utilizing anhydrous environments and specific catalytic acids to drive the formation of the tetrahydro-beta-carboline core. This level of control is essential for minimizing impurity profiles, a key concern for any manufacturer aiming to integrate these materials into sensitive electronic or pharmaceutical workflows. The ability to produce these compounds with defined optical rotation and specific melting points underscores the reproducibility of the method. As the industry seeks reliable electronic chemical suppliers who can deliver consistent quality, the processes described in CN101157694B offer a blueprint for achieving the stringent specifications demanded by modern technology sectors. The transition from laboratory-scale discovery to commercial viability is facilitated by the use of standard reagents and scalable purification techniques, making this technology highly attractive for industrial adoption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for carboline derivatives often suffer from poor stereoselectivity and the formation of complex impurity mixtures that are difficult to separate. Conventional methods frequently rely on harsh reaction conditions that can degrade sensitive functional groups, leading to lower overall yields and compromised material purity. In many existing processes, the separation of epimers is particularly challenging, often requiring multiple recrystallization steps that result in significant material loss and increased production costs. Additionally, older methodologies may not account for the specific crystal engineering required to achieve the nano-scale surface features that enhance the performance of organic materials in device applications. Without precise control over the stereochemistry at the C-1 and C-3 positions, the resulting materials may exhibit inconsistent physical properties, rendering them unsuitable for high-precision applications. These limitations create bottlenecks in the supply chain, as manufacturers struggle to source materials that meet the rigorous quality standards necessary for commercial deployment.
The Novel Approach
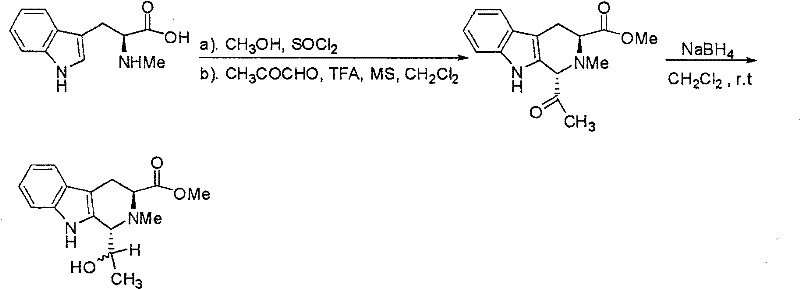
The approach detailed in CN101157694B overcomes these historical challenges by employing a streamlined, acid-catalyzed cyclization followed by a controlled reduction sequence. This novel pathway utilizes abrine methyl ester as a chiral pool starting material, which inherently biases the stereochemical outcome of the reaction, thereby simplifying the downstream purification process. By introducing pyruvic aldehyde and trifluoroacetic acid under anhydrous conditions, the reaction efficiently constructs the rigid carboline scaffold while preserving the integrity of the ester functionality. The subsequent reduction with sodium borohydride is carefully managed to ensure the formation of the desired hydroxyethyl side chain with high fidelity. This method not only improves the overall yield but also facilitates the isolation of the specific epimers required for advanced applications. The result is a process that delivers high-purity carboline compounds with unique nano-structural characteristics, offering a distinct competitive advantage in the market for specialized organic materials.
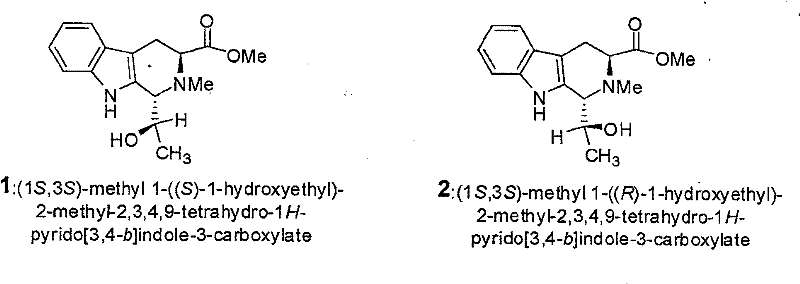
Mechanistic Insights into Acid-Catalyzed Cyclization and Reduction
The core of this synthesis lies in the mechanistic elegance of the Pictet-Spengler-type cyclization coupled with a chemoselective reduction. The reaction begins with the activation of the indole nucleus in the abrine methyl ester by the acidic environment provided by trifluoroacetic acid. This activation promotes the nucleophilic attack on the carbonyl carbon of the pyruvic aldehyde, leading to the formation of an iminium ion intermediate. The presence of molecular sieves in the reaction mixture is crucial, as it sequesters water generated during the imine formation, driving the equilibrium towards the cyclized product. This step is fundamental to establishing the tetrahydro-beta-carboline skeleton, which serves as the rigid core for the final material. The stereochemistry at the newly formed chiral centers is influenced by the existing chirality of the abrine backbone, ensuring that the reaction proceeds with a high degree of diastereoselectivity. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for scale-up, as slight deviations in acidity or water content can significantly impact the ratio of epimers formed.
Following the cyclization, the reduction step utilizes sodium borohydride to convert the ketone functionality into the secondary alcohol, completing the synthesis of the target epimers. The patent specifies a two-stage addition of the reducing agent, which is a critical process parameter for controlling the reaction exotherm and ensuring complete conversion without over-reduction of the ester group. This chemoselectivity is paramount, as preserving the methyl ester functionality is necessary for the subsequent application of these compounds as organic materials. The resulting products, Compound 1 and Compound 2, differ only in the configuration of the hydroxyethyl side chain, yet this subtle difference leads to distinct crystal packing arrangements and surface morphologies. The ability to separate these epimers via normal-phase silica gel chromatography and HPLC demonstrates the robustness of the purification strategy. For R&D teams, this mechanistic clarity provides confidence in the reproducibility of the synthesis, allowing for precise tuning of material properties through controlled stereoisomer production.
How to Synthesize Carboline Compound Efficiently
The synthesis of these high-value carboline compounds requires a disciplined approach to reaction conditions and purification protocols to ensure the highest quality output. The process begins with the preparation of the abrine methyl ester, which serves as the chiral foundation for the entire sequence. Operators must maintain strict nitrogen protection and low-temperature conditions during the esterification step to prevent racemization and degradation of the sensitive indole ring. Following this, the cyclization reaction demands the use of high-quality anhydrous solvents and activated molecular sieves to drive the equilibrium towards the desired product. The reduction phase requires careful monitoring of the addition rate of sodium borohydride to manage heat generation and ensure selectivity. Finally, the separation of the epimeric mixture is achieved through a combination of flash chromatography and preparative HPLC, a standard yet critical step for achieving the purity levels required for electronic applications. Detailed standardized synthesis steps see the guide below.
- Preparation of Abrine Methyl Ester: React abrine with thionyl chloride in anhydrous methanol under nitrogen protection at low temperature (-10°C to -15°C), followed by pH adjustment and extraction.
- Cyclization and Reduction: Dissolve the ester in methylene chloride, add molecular sieves, pyruvic aldehyde, and trifluoric acid. After reaction, reduce with sodium borohydride in two stages.
- Purification and Separation: Separate the resulting epimer mixture using silica gel column chromatography followed by HPLC purification to isolate pure Compound 1 and Compound 2.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthesis route described in CN101157694B offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for advanced organic materials. The reliance on abrine, a derivative of the abundant amino acid tryptophan, ensures that the raw material supply is stable and not subject to the volatility often associated with exotic synthetic precursors. This foundational availability translates directly into reduced supply chain risk and more predictable lead times for production planning. Furthermore, the streamlined nature of the synthesis, which avoids the use of expensive transition metal catalysts or complex protecting group strategies, significantly lowers the cost of goods sold. The elimination of heavy metal catalysts also simplifies the purification process, removing the need for costly metal scavenging steps and reducing the environmental footprint of the manufacturing process. These factors combine to create a highly efficient production model that supports competitive pricing without compromising on material quality.
- Cost Reduction in Manufacturing: The process design inherently minimizes production costs by utilizing readily available starting materials and avoiding expensive catalytic systems. By eliminating the need for precious metal catalysts, the manufacturing process avoids the significant costs associated with metal removal and validation, which are often required in pharmaceutical and electronic grade chemical production. Additionally, the high selectivity of the reaction reduces the formation of by-products, thereby improving the overall yield and reducing the volume of waste that requires disposal. This efficiency allows for a more favorable cost structure, enabling suppliers to offer competitive pricing for high-purity carboline compounds. The simplified workflow also reduces labor and utility costs associated with complex multi-step syntheses, further enhancing the economic viability of large-scale production.
- Enhanced Supply Chain Reliability: The use of abrine as a key starting material provides a robust foundation for supply chain continuity, as tryptophan derivatives are produced globally in large quantities for the food and pharmaceutical industries. This widespread availability mitigates the risk of raw material shortages that can plague the supply of specialized fine chemicals. Moreover, the synthesis does not rely on reagents that are subject to strict regulatory controls or geopolitical supply constraints, ensuring a smoother procurement process. The scalability of the reaction conditions, which utilize standard solvents and reagents, means that production can be easily ramped up to meet increasing demand without requiring specialized equipment or infrastructure. This reliability is crucial for downstream manufacturers who depend on a consistent supply of high-quality organic materials for their own production schedules.
- Scalability and Environmental Compliance: The synthesis route is well-suited for commercial scale-up due to its use of standard unit operations such as filtration, extraction, and chromatography, which are easily adaptable to larger reactor volumes. The absence of toxic heavy metals and the use of relatively benign solvents align with modern environmental, health, and safety (EHS) standards, facilitating regulatory approval and reducing the burden of waste management. The efficient atom economy of the cyclization step minimizes waste generation, contributing to a more sustainable manufacturing process. This environmental compliance is increasingly important for multinational corporations seeking to reduce their carbon footprint and meet sustainability goals. The ability to produce these materials in a green and scalable manner positions them as a preferred choice for industries prioritizing sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carboline compounds, based on the detailed disclosures within the patent literature. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating these materials into their product lines. The answers provided reflect the specific technical advantages and process characteristics outlined in the intellectual property, offering clarity on performance and manufacturability. Stakeholders are encouraged to review these insights to align their technical requirements with the capabilities of the described synthesis method.
Q: What distinguishes the crystal structure of these carboline compounds from conventional organic materials?
A: The patent highlights unique nano-scale surface morphologies, such as protrusions and bud-shaped structures, which are not typically observed in standard carboline derivatives, suggesting enhanced potential for optoelectronic applications.
Q: How is stereochemical purity maintained during the synthesis of the epimers?
A: The process utilizes specific chiral starting materials (abrine) and controlled reduction conditions with sodium borohydride, followed by rigorous HPLC separation on normal-phase silica gel to isolate the distinct (1S,3S) epimers.
Q: Are the raw materials for this synthesis commercially scalable?
A: Yes, the synthesis begins with abrine, a tryptophan derivative that is readily available in bulk quantities, ensuring a stable supply chain for large-scale manufacturing of these organic materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carboline Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-performance organic materials in driving innovation across the electronic and specialty chemical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated synthesis routes described in patents like CN101157694B can be successfully translated into industrial reality. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify stereochemistry and nano-structural integrity. Our capability to handle complex chiral syntheses and precise separations makes us an ideal partner for companies seeking a reliable carboline compound supplier who can guarantee both quality and consistency.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into how our optimized processes can reduce your overall material costs while maintaining the highest standards of quality. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your application requirements. Whether you are developing next-generation OLED materials or advanced pharmaceutical intermediates, our expertise in process chemistry and scale-up ensures that we can be a trusted extension of your R&D and supply chain operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
