Advanced Synthesis of Galactosyl Thiourea Heterocycles for Commercial Oncology Applications
The pharmaceutical landscape for antineoplastic agents is continuously evolving, with a specific emphasis on novel glycosylated heterocyclic compounds that exhibit enhanced bioavailability and targeted biological activity. Patent CN101307078B introduces a groundbreaking methodology for the synthesis of galactosyl thiourea heterocyclic compounds, a class of molecules that has demonstrated significant inhibitory effects against human lung cancer and liver cancer cell lines. This technical disclosure provides a robust six-step synthetic pathway that transforms readily available peracetylated galactose into complex thiourea derivatives through a strategic isothiocyanate intermediate. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a critical asset for developing next-generation oncology therapeutics. The structural versatility of these compounds, as illustrated by the general formula, allows for extensive modification of the heterocyclic base, enabling the fine-tuning of pharmacological properties to meet specific therapeutic requirements. 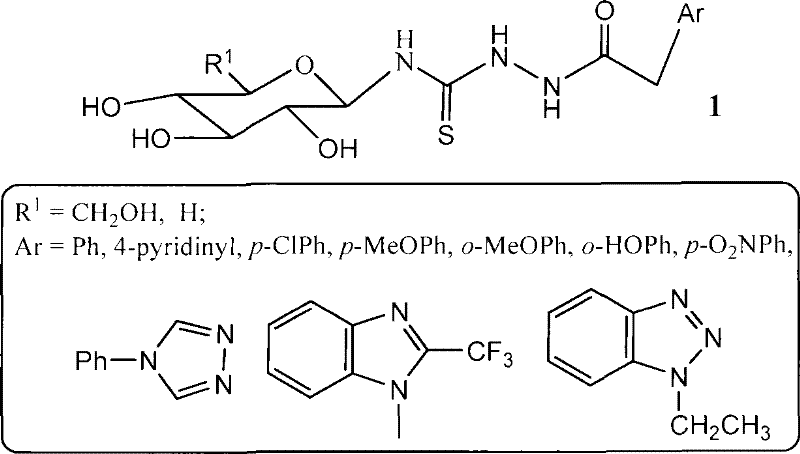
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing glycosyl thiourea derivatives often suffer from significant drawbacks related to reaction selectivity and overall yield efficiency. Conventional glycosylation methods frequently require harsh acidic conditions or expensive transition metal catalysts that can lead to the degradation of sensitive heterocyclic bases or the formation of complex impurity profiles that are difficult to purge. Furthermore, direct coupling strategies often result in poor stereocontrol at the anomeric center, leading to mixtures of alpha and beta isomers that complicate downstream purification and regulatory approval processes. The reliance on unstable intermediates in older methodologies also poses substantial safety risks and supply chain vulnerabilities, as these materials often have short shelf lives and require specialized storage conditions. These technical bottlenecks historically increased the cost reduction in API manufacturing challenges, making it difficult to produce these high-value intermediates at a commercially viable scale without compromising on quality or safety standards.
The Novel Approach
The methodology disclosed in CN101307078B overcomes these historical limitations by employing a modular six-step synthesis that prioritizes stability and selectivity at every stage. By converting peracetylated galactose into a stable galactosyl isothiocyanate intermediate, the process decouples the glycosylation step from the heterocyclic coupling, allowing each reaction to be optimized independently for maximum yield. This strategic separation ensures that the sensitive thiourea linkage is formed under mild conditions, preserving the integrity of the heterocyclic base whether it is adenine, benzimidazole, or other complex nitrogenous structures. The use of standard solvents such as dimethylformamide and methanol, combined with common reagents like sodium hydride and lead thiocyanate, simplifies the procurement of raw materials and reduces dependency on exotic catalysts. This novel approach not only enhances the reproducibility of the synthesis but also facilitates the commercial scale-up of complex heterocyclic compounds by utilizing equipment and unit operations that are standard in modern fine chemical manufacturing facilities.
Mechanistic Insights into Galactosyl Isothiocyanate Coupling
The core mechanistic innovation of this patent lies in the generation and utilization of the galactosyl isothiocyanate intermediate, which serves as a highly electrophilic species for the subsequent nucleophilic attack by the heterocyclic acetylhydrazide. The formation of this isothiocyanate from the corresponding glycosyl bromide using lead thiocyanate proceeds with high stereochemical fidelity, ensuring that the beta-configuration is maintained throughout the synthesis. This is critical for the biological activity of the final molecule, as the spatial orientation of the sugar moiety often dictates its recognition by cellular transporters and enzymes. The coupling reaction between the isothiocyanate and the hydrazide is driven by the formation of a stable thiourea bond, a process that is thermodynamically favorable and kinetically controlled to minimize side reactions. 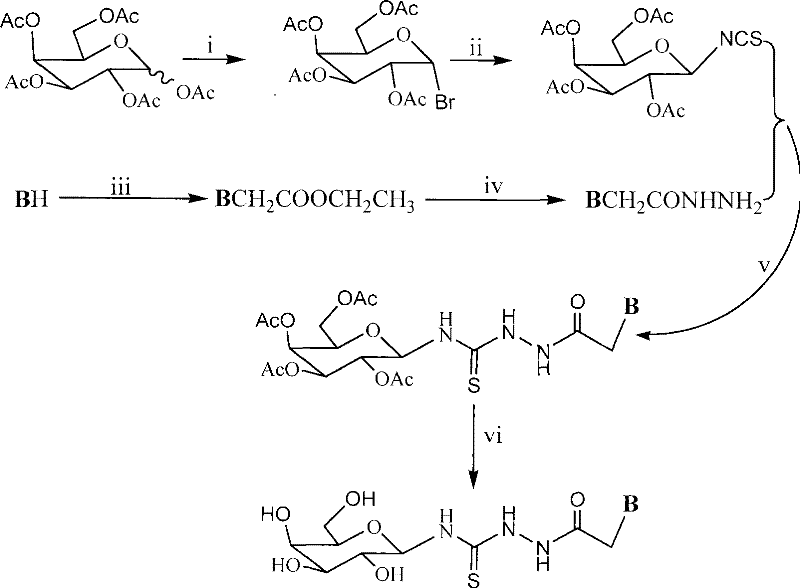
Impurity control is meticulously managed through the final deprotection step, which utilizes sodium methoxide in methanol to remove the acetyl protecting groups from the galactose ring. This Zemplén deacetylation is highly selective for ester bonds and leaves the thiourea and heterocyclic functionalities intact, preventing the formation of degradation products that could compromise the safety profile of the drug substance. The process includes rigorous workup procedures involving cation exchange resin treatment to neutralize the reaction mixture, which effectively removes metal ions and basic residues that could catalyze decomposition during storage. This attention to mechanistic detail ensures that the final high-purity galactosyl thiourea derivatives meet the stringent purity specifications required for clinical development, providing a solid foundation for regulatory filings and commercial production.
How to Synthesize Galactosyl Thiourea Efficiently
The synthesis of these valuable anticancer intermediates requires precise control over reaction parameters to ensure consistent quality and yield across multiple batches. The process begins with the bromination of peracetylated galactose, followed by conversion to the isothiocyanate, which is then coupled with a pre-synthesized heterocyclic acetylhydrazide. The final deprotection step is critical for revealing the active hydroxyl groups on the sugar moiety, which are essential for water solubility and biological interaction. Detailed standard operating procedures for each of the six steps, including specific temperature ranges, reaction times, and workup protocols, are essential for transferring this technology from the laboratory to the pilot plant.
- Bromination of peracetylated galactose followed by conversion to galactosyl isothiocyanate using lead thiocyanate.
- Alkylation of heterocyclic bases with ethyl chloroacetate and subsequent hydrazinolysis to form acetylhydrazides.
- Coupling of isothiocyanate and acetylhydrazide followed by Zemplén deacetylation to yield the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for oncology intermediates. The reliance on commodity chemicals such as galactose, bromine, and common heterocyclic bases ensures a stable and diversified supply chain that is less susceptible to market volatility or single-source bottlenecks. The elimination of expensive transition metal catalysts and the use of recyclable solvents contribute to a drastic simplification of the waste management profile, aligning with modern environmental compliance standards and reducing disposal costs. Furthermore, the high yields reported in the patent examples indicate a material-efficient process that maximizes the output from raw materials, directly translating to substantial cost savings in the overall manufacturing budget.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for precious metal catalysts and specialized reagents, relying instead on cost-effective inorganic salts and organic bases that are readily available in bulk quantities. This shift in reagent profile significantly lowers the direct material costs associated with production, allowing for more competitive pricing structures in the final API market. Additionally, the high conversion rates observed in the coupling and deprotection steps minimize the loss of valuable intermediates, further enhancing the economic viability of the process for large-scale operations.
- Enhanced Supply Chain Reliability: By utilizing raw materials that are produced by multiple global suppliers, this manufacturing route reduces the risk of supply disruptions caused by geopolitical issues or vendor-specific production failures. The robustness of the chemical steps means that the process can be easily replicated across different manufacturing sites, providing redundancy and flexibility in the supply network. This reliability is crucial for reducing lead time for high-purity anticancer intermediates, ensuring that clinical and commercial demands can be met without delay.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, using standard reaction vessels and separation techniques that can be easily expanded from kilogram to multi-ton scales without requiring specialized engineering modifications. The waste streams generated are primarily organic solvents and inorganic salts, which can be treated using conventional wastewater treatment protocols, ensuring full compliance with environmental regulations. This ease of scale-up and environmental compatibility makes the technology an attractive option for sustainable pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these galactosyl thiourea derivatives. The answers are derived directly from the experimental data and technical disclosures within the patent, providing accurate information for decision-makers.
Q: What are the key advantages of this glycosylation method over conventional approaches?
A: This method utilizes a stable isothiocyanate intermediate which allows for milder coupling conditions compared to direct glycosylation, significantly reducing side reactions and improving overall yield consistency.
Q: How is the purity of the final galactosyl thiourea derivative ensured?
A: The process employs a final Zemplén deacetylation step using sodium methoxide, which effectively removes protecting groups without degrading the sensitive thiourea linkage, ensuring high chemical purity.
Q: Is this synthetic route scalable for industrial API production?
A: Yes, the six-step sequence relies on standard unit operations such as reflux, filtration, and crystallization, making it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Galactosyl Thiourea Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the synthetic route described in CN101307078B to meet your specific volume and quality requirements, ensuring that every batch adheres to stringent purity specifications. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of every intermediate and final product, guaranteeing consistency and reliability for your drug development pipeline. Our commitment to quality and technical excellence makes us the ideal partner for bringing these complex oncology intermediates to the global market.
We invite you to engage with our technical procurement team to discuss your specific project needs and explore how we can support your supply chain goals. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing efficiencies can translate into tangible economic benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a smooth and successful transition from development to commercial supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
