Advanced Synthesis Of Glucosyl Thiourea Heterocycles For Commercial Antitumor Drug Development
Advanced Synthesis Of Glucosyl Thiourea Heterocycles For Commercial Antitumor Drug Development
The pharmaceutical industry is constantly seeking novel chemical scaffolds that offer improved therapeutic indices and manageable synthesis pathways for oncology applications. Patent CN101215305B discloses a significant advancement in this domain by presenting a series of glucosyl thiourea heterocyclic compounds and their robust six-step synthesis method. This technology addresses the critical need for water-soluble antitumor agents by conjugating biologically active heterocyclic bases with a glucose moiety through a thiourea linkage. The strategic combination of carbohydrate chemistry and heterocyclic synthesis creates a unique chemical space that enhances molecular recognition processes within biological systems. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for evaluating new pipeline candidates. The disclosed method utilizes readily available starting materials such as glucose, bromine water, and various heterocyclic bases, establishing a foundation for scalable manufacturing. By leveraging this intellectual property, stakeholders can explore new avenues for developing next-generation anticancer therapeutics with optimized pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing thiourea derivatives often struggle with poor aqueous solubility, which severely limits their bioavailability and clinical utility. Many conventional heterocyclic compounds exhibit high lipophilicity, leading to aggregation in physiological fluids and inconsistent absorption rates in vivo. Furthermore, standard coupling reactions frequently require harsh conditions or expensive transition metal catalysts that complicate downstream purification and increase the risk of toxic metal residues in the final active pharmaceutical ingredient. The lack of a polar carrier group in typical thiourea synthesis means that additional formulation steps are required to solubilize the drug, adding significant cost and complexity to the manufacturing process. These limitations create bottlenecks in the supply chain, as producing high-purity material that meets regulatory standards becomes increasingly difficult and costly. Consequently, there is a persistent demand for synthetic routes that inherently improve solubility without compromising the structural integrity of the pharmacophore.
The Novel Approach
The methodology outlined in CN101215305B offers a transformative solution by integrating a glucosyl unit directly into the thiourea heterocyclic framework. This innovative route employs a six-step sequence that begins with the acetylation and bromination of glucose, followed by conversion to an isothiocyanate intermediate. This reactive species is then coupled with heterocyclic acethydrazides to form the protected thiourea linkage, which is subsequently deprotected to yield the final bioactive compound. 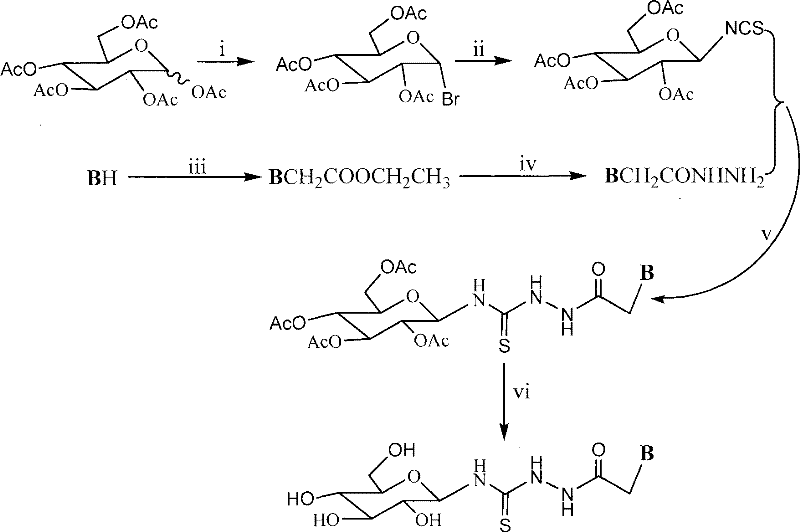 This process avoids the need for complex protecting group strategies often seen in carbohydrate chemistry, streamlining the overall production timeline. The use of common solvents like dimethylformamide and methanol ensures that the process is compatible with existing industrial infrastructure. By embedding the sugar moiety early in the synthesis, the resulting molecules possess intrinsic water solubility, reducing the need for excipients and simplifying formulation development. This approach not only enhances the therapeutic potential of the compounds but also offers a more economically viable pathway for large-scale production of antitumor intermediates.
This process avoids the need for complex protecting group strategies often seen in carbohydrate chemistry, streamlining the overall production timeline. The use of common solvents like dimethylformamide and methanol ensures that the process is compatible with existing industrial infrastructure. By embedding the sugar moiety early in the synthesis, the resulting molecules possess intrinsic water solubility, reducing the need for excipients and simplifying formulation development. This approach not only enhances the therapeutic potential of the compounds but also offers a more economically viable pathway for large-scale production of antitumor intermediates.
Mechanistic Insights into Glycosyl Thiourea Formation
The core of this technology lies in the precise construction of the thiourea bridge between the sugar and the heterocyclic base. The reaction mechanism involves the nucleophilic attack of the hydrazide nitrogen on the electrophilic carbon of the glucosyl isothiocyanate. This step is critical as it determines the regioselectivity and yield of the coupling reaction. The presence of the acetyl protecting groups on the glucose ring during this stage prevents unwanted side reactions at the hydroxyl positions, ensuring that the thiourea bond forms exclusively at the anomeric center. Following the coupling, the deprotection step using sodium methoxide in methanol cleanly removes the acetyl groups without disrupting the sensitive thiourea linkage. This chemoselectivity is vital for maintaining the structural fidelity of the final product. 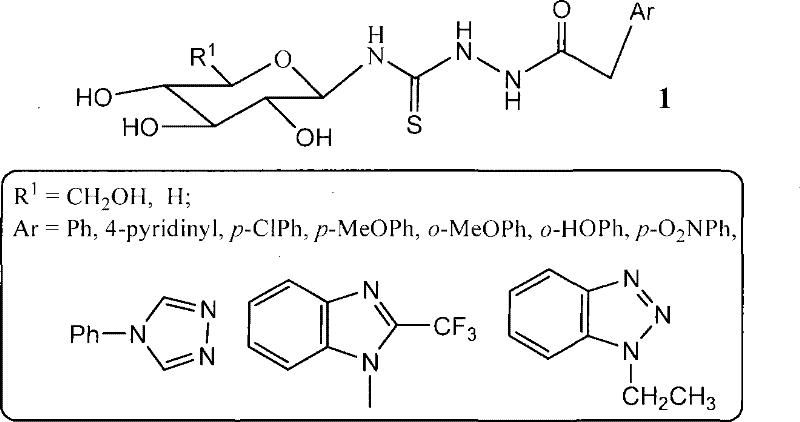 The resulting structure features a flexible linker that allows the heterocyclic base to interact effectively with biological targets while the glucose unit facilitates transport through aqueous environments. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction conditions or explore analog variations.
The resulting structure features a flexible linker that allows the heterocyclic base to interact effectively with biological targets while the glucose unit facilitates transport through aqueous environments. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction conditions or explore analog variations.
Furthermore, the impurity profile of this synthesis is well-defined, primarily consisting of unreacted starting materials or partially deprotected intermediates which are easily removed via crystallization or chromatography. The patent specifies the use of zeolite or ion-exchange resins during the workup to neutralize the reaction mixture, which helps in controlling the pH and minimizing degradation products. This level of control over the impurity spectrum is essential for meeting the stringent purity requirements of pharmaceutical regulators. The ability to produce high-purity glucosyl thiourea derivatives consistently reduces the risk of batch failures and ensures a reliable supply of quality material for preclinical and clinical studies. For supply chain managers, this predictability translates to reduced inventory buffers and more efficient resource allocation.
How to Synthesize Glucosyl Thiourea Efficiently
The synthesis of these high-value intermediates requires strict adherence to the patented six-step protocol to ensure optimal yield and purity. The process begins with the activation of glucose, followed by the sequential construction of the thiourea backbone and the attachment of the heterocyclic warhead. Detailed operational parameters regarding temperature control, stoichiometry, and solvent selection are critical for success. While the general pathway is robust, minor adjustments may be needed depending on the specific heterocyclic base employed. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Preparation of 1-bromo-2,3,4,6-tetra-O-acetyl-alpha-D-glucose using red phosphorus and bromine.
- Conversion to glucosyl isothiocyanate via reaction with lead thiocyanate in xylene.
- Coupling of heterocyclic acethydrazide with glucosyl isothiocyanate followed by deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement and supply chain operations within the pharmaceutical sector. The reliance on commodity chemicals such as glucose, bromine, and common heterocycles ensures that raw material costs remain stable and predictable. Unlike processes dependent on rare earth metals or proprietary reagents, this method leverages a supply chain that is resilient to market fluctuations. The elimination of complex purification steps typically associated with lipophilic drugs further reduces processing time and solvent consumption. This efficiency gain directly contributes to cost reduction in antitumor drug manufacturing, allowing companies to allocate resources more effectively towards R&D and clinical trials. Additionally, the improved solubility of the final product minimizes the need for expensive solubilizing agents, lowering the overall bill of materials for the finished dosage form.
- Cost Reduction in Manufacturing: The streamlined six-step synthesis avoids the use of expensive transition metal catalysts, which significantly lowers the cost of goods sold. By utilizing standard organic bases and common solvents, the process reduces the financial burden associated with specialized reagent procurement and hazardous waste disposal. The high yields reported in the patent examples indicate a material-efficient process that maximizes output from input raw materials. This efficiency is crucial for maintaining competitive pricing in the generic and specialty pharmaceutical markets. Furthermore, the simplified workup procedures reduce labor hours and energy consumption, contributing to a leaner manufacturing footprint.
- Enhanced Supply Chain Reliability: The use of widely available starting materials mitigates the risk of supply disruptions caused by geopolitical issues or single-source dependencies. Glucose and basic heterocyclic bases are produced globally in large volumes, ensuring a continuous flow of inputs for production. This availability supports the commercial scale-up of complex pharmaceutical intermediates without the fear of raw material shortages. For supply chain heads, this reliability means shorter lead times and the ability to respond quickly to changes in market demand. The robustness of the chemistry also implies fewer batch failures, leading to a more consistent and dependable supply of critical antitumor intermediates.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory to pilot and commercial scales. The avoidance of highly toxic reagents where possible aligns with modern green chemistry principles, reducing the environmental impact of manufacturing operations. Waste streams generated during the process are manageable using standard treatment protocols, facilitating compliance with increasingly strict environmental regulations. This alignment with sustainability goals enhances the corporate social responsibility profile of the manufacturing entity. Scalability ensures that as the drug candidate progresses through clinical phases, the supply can be ramped up seamlessly to meet commercial launch requirements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of glucosyl thiourea heterocyclic compounds. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of incorporating these intermediates into their drug development pipelines. The answers reflect the current state of the art as described in the intellectual property.
Q: What is the primary advantage of introducing a glycosyl group to thiourea derivatives?
A: The introduction of a glycosyl moiety significantly enhances the water solubility of the lipophilic thiourea and heterocyclic backbone, facilitating better bioavailability and formulation stability in physiological environments.
Q: Does this synthesis route avoid the use of heavy metal catalysts?
A: While lead thiocyanate is utilized in the isothiocyanate formation step, the process relies primarily on standard organic bases like sodium hydride and sodium methoxide, allowing for established purification protocols to ensure low residual metal content in the final API intermediate.
Q: What is the reported antitumor activity of these compounds?
A: Experimental data indicates inhibitory effects against human lung cancer cell lines (PG) and human liver cancer cell lines (BEL-7402), with specific derivatives demonstrating EC50 values in the micromolar range, suggesting potential for further oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glucosyl Thiourea Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the race to develop effective antitumor therapies. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from bench to market. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of glucosyl thiourea derivative meets the highest international standards. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains for critical oncology ingredients.
We invite you to collaborate with us to optimize your sourcing strategy and accelerate your development timelines. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your commercial goals. Let us help you navigate the complexities of fine chemical sourcing with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
