Advanced Synthesis of Xylosyl Thiourea Heterocyclic Compounds for Oncology Applications
The pharmaceutical landscape for oncology treatments is continuously evolving, driven by the need for more effective and selective antineoplastic agents. Patent CN101215303B introduces a significant advancement in this field by disclosing a novel group of xylosyl thiourea heterocyclic compounds with demonstrated inhibitory effects on human lung and liver cancer cell lines. This technology represents a critical intersection of carbohydrate chemistry and heterocyclic synthesis, offering a robust pathway for developing next-generation therapeutic intermediates. For R&D directors and procurement specialists, understanding the technical nuances of this six-step synthetic route is essential for evaluating its potential in drug discovery pipelines. The patent details a method that transforms peracetylated xylose into complex bioactive molecules using specific reagents like lead thiocyanate and various heterocyclic bases. This report provides a deep technical and commercial analysis of this synthesis, highlighting its implications for cost reduction in API manufacturing and supply chain stability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing glycosyl thiourea derivatives often suffer from significant drawbacks that hinder their commercial viability and scalability in a GMP environment. Many conventional routes rely on harsh reaction conditions that can degrade sensitive sugar moieties, leading to complex impurity profiles that are difficult to purify. Furthermore, standard glycosylation techniques frequently struggle with stereocontrol, resulting in mixtures of alpha and beta anomers that require extensive and costly chromatographic separation. The use of unstable isothiocyanate intermediates in older protocols often necessitates cryogenic conditions, which drastically increases energy consumption and operational complexity. Additionally, the solubility of final products in aqueous media is often poor, limiting their biological efficacy and requiring additional formulation steps. These technical bottlenecks create substantial risks for supply chain heads who require consistent quality and reliable lead times for clinical trial materials. The cumulative effect of these limitations is a high cost of goods sold and a prolonged time-to-market for potential antineoplastic drugs.
The Novel Approach
The methodology outlined in patent CN101215303B offers a transformative solution to these historical challenges through a carefully engineered six-step synthetic sequence. This novel approach utilizes peracetylated xylose as a stable starting material, which is systematically converted into a reactive bromide and subsequently into an isothiocyanate using lead thiocyanate in refluxing xylene. 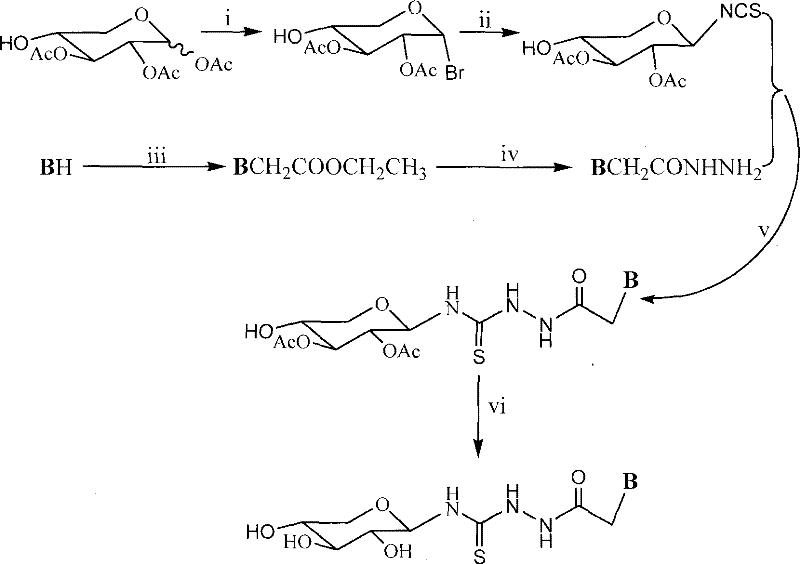 . This specific pathway avoids the need for extreme cryogenic temperatures, operating instead within manageable ranges of 0 to 140 degrees Celsius depending on the step. The strategy employs common industrial solvents such as DMF, methanol, and toluene, which simplifies solvent recovery and waste management processes. By coupling the sugar isothiocyanate with heterocyclic base hydrazides, the method ensures high regioselectivity and yield, as evidenced by the specific examples provided in the patent data. This streamlined process significantly reduces the number of purification steps required, thereby enhancing the overall efficiency and economic feasibility of producing these complex intermediates.
. This specific pathway avoids the need for extreme cryogenic temperatures, operating instead within manageable ranges of 0 to 140 degrees Celsius depending on the step. The strategy employs common industrial solvents such as DMF, methanol, and toluene, which simplifies solvent recovery and waste management processes. By coupling the sugar isothiocyanate with heterocyclic base hydrazides, the method ensures high regioselectivity and yield, as evidenced by the specific examples provided in the patent data. This streamlined process significantly reduces the number of purification steps required, thereby enhancing the overall efficiency and economic feasibility of producing these complex intermediates.
Mechanistic Insights into Glycosyl Thiourea Formation
The core innovation of this technology lies in the precise construction of the thiourea linkage attached to the xylosyl scaffold, which is critical for the compound's biological activity. The reaction mechanism involves the nucleophilic attack of the heterocyclic hydrazide on the electrophilic carbon of the xylosyl isothiocyanate intermediate. This step is facilitated by the use of anhydrous alkaline inert solvents like DMF, which stabilize the transition state and promote the formation of the desired thiosemicarbazide bond. The subsequent deacetylation step using sodium methoxide in methanol is equally crucial, as it reveals the free hydroxyl groups on the sugar ring. 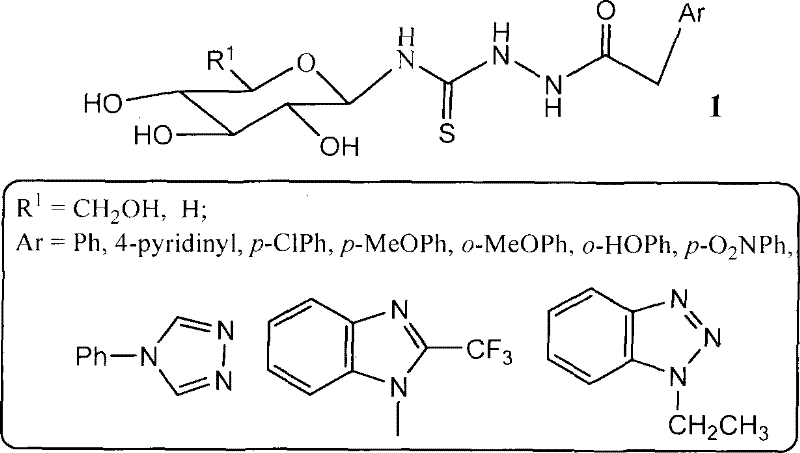 . These hydroxyl groups are responsible for the enhanced water solubility mentioned in the patent, a key pharmacokinetic property for antineoplastic agents. The structural diversity is achieved by varying the heterocyclic base B, which can range from adenine and guanine to benzimidazole and triazole derivatives. This modularity allows medicinal chemists to fine-tune the electronic and steric properties of the final molecule to optimize binding affinity with biological targets. Understanding this mechanistic flexibility is vital for R&D teams aiming to generate analog libraries for structure-activity relationship studies.
. These hydroxyl groups are responsible for the enhanced water solubility mentioned in the patent, a key pharmacokinetic property for antineoplastic agents. The structural diversity is achieved by varying the heterocyclic base B, which can range from adenine and guanine to benzimidazole and triazole derivatives. This modularity allows medicinal chemists to fine-tune the electronic and steric properties of the final molecule to optimize binding affinity with biological targets. Understanding this mechanistic flexibility is vital for R&D teams aiming to generate analog libraries for structure-activity relationship studies.
Impurity control is another critical aspect of this synthesis that directly impacts the feasibility of commercial manufacturing. The use of lead thiocyanate introduces a heavy metal into the process, which necessitates rigorous purification strategies to meet ICH Q3D guidelines for elemental impurities. The patent describes specific workup procedures, including filtration to remove excess lead salts and recrystallization from mixed solvents like toluene and petroleum ether. The final deacetylation step is monitored by thin-layer chromatography to ensure complete conversion, preventing the carryover of acetylated intermediates which could act as genotoxic impurities. The ability to control these impurities through standard unit operations like filtration, extraction, and crystallization is a major advantage for process chemists. It ensures that the final high-purity pharmaceutical intermediates meet the stringent quality specifications required by regulatory agencies. This level of control reduces the risk of batch failures and ensures consistent supply for downstream drug product manufacturing.
How to Synthesize Xylosyl Thiourea Efficiently
Implementing this synthetic route requires a detailed understanding of the reaction parameters and safety considerations associated with the reagents involved. The process begins with the bromination of peracetylated xylose, followed by the formation of the isothiocyanate, and concludes with the coupling and deprotection steps. Each stage requires precise temperature control and stoichiometry to maximize yield and minimize byproduct formation. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety protocols. This structured approach ensures reproducibility and safety, which are paramount when scaling up from laboratory to pilot plant operations.
- Bromination of peracetylated xylose using red phosphorus and bromine water to form 1-bromo-2,3,4-tri-O-acetyl-alpha-D-xylose.
- Conversion to isothiocyanate derivative via reaction with lead thiocyanate in refluxing xylene.
- Coupling with heterocyclic base hydrazides followed by deacetylation using sodium methoxide to yield the final xylosyl thiourea compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic technology offers substantial benefits for procurement managers and supply chain heads focused on cost optimization and reliability. The use of readily available starting materials like peracetylated xylose and common heterocyclic bases reduces dependency on exotic or single-source reagents. This availability mitigates supply chain risks and ensures continuity of supply even during market fluctuations. The process avoids the use of expensive transition metal catalysts that often require complex removal steps, thereby simplifying the downstream processing workflow. Furthermore, the reaction conditions are compatible with standard stainless steel reactors, eliminating the need for specialized glass-lined or Hastelloy equipment. These factors collectively contribute to significant cost savings in API manufacturing without compromising on the quality or purity of the final intermediate. The robustness of the route also implies a lower risk of batch-to-batch variability, which is a key metric for supply chain performance.
- Cost Reduction in Manufacturing: The elimination of cryogenic conditions and the use of common solvents drastically reduce energy consumption and utility costs associated with the synthesis. By avoiding expensive chiral catalysts and relying on the inherent chirality of the xylose starting material, the process achieves high stereochemical purity at a fraction of the cost of asymmetric synthesis methods. The efficient workup procedures minimize solvent usage and waste generation, leading to lower disposal costs and a reduced environmental footprint. These operational efficiencies translate directly into a more competitive cost structure for the final pharmaceutical intermediate. Procurement teams can leverage these efficiencies to negotiate better pricing or reinvest savings into further R&D activities.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the majority of the reagents ensures that the supply chain is resilient to disruptions. Unlike processes that depend on custom-synthesized catalysts or rare earth metals, this route can be sourced from multiple global suppliers. The scalability of the reaction steps allows for flexible production scheduling, enabling manufacturers to respond quickly to changes in demand from clinical or commercial partners. This flexibility is crucial for maintaining just-in-time inventory levels and reducing working capital requirements. Supply chain heads can confidently plan long-term procurement strategies knowing that the raw material base is stable and secure.
- Scalability and Environmental Compliance: The synthetic route is designed with scale-up in mind, utilizing unit operations that are easily transferred from laboratory to commercial scale. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through standard treatment protocols. The absence of persistent organic pollutants or highly toxic byproducts simplifies regulatory compliance and environmental permitting. This alignment with green chemistry principles enhances the sustainability profile of the manufacturing process. Companies prioritizing ESG goals will find this technology particularly attractive as it supports responsible production practices while maintaining high output levels.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these xylosyl thiourea compounds. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They serve to clarify the feasibility and advantages of adopting this synthetic route for your specific project needs. Understanding these details is essential for making informed decisions about technology transfer and process validation.
Q: What are the key raw materials for synthesizing xylosyl thiourea derivatives?
A: The process utilizes peracetylated xylose, bromine water, lead thiocyanate, heterocyclic bases, ethyl chloroacetate, and hydrazine hydrate as primary starting materials.
Q: How does this synthesis method improve solubility for biological applications?
A: The introduction of the glycosyl moiety significantly enhances the water solubility of the thiourea derivative, facilitating better bioavailability in antineoplastic applications.
Q: Is this process scalable for commercial API intermediate production?
A: Yes, the six-step route uses common solvents like DMF and methanol and avoids ultra-low temperature conditions, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Xylosyl Thiourea Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of glycosylation and heterocyclic chemistry, ensuring that your project benefits from our deep process knowledge. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest industry standards. Our commitment to quality and reliability makes us the ideal partner for developing and supplying these critical antineoplastic intermediates. We understand the pressures of drug development timelines and are dedicated to supporting your success through every stage of the product lifecycle.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for these specialized compounds. Request a Customized Cost-Saving Analysis to understand the specific economic benefits of partnering with us for your xylosyl thiourea needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to meet your exact requirements. Let us help you accelerate your drug development program with our reliable and high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
