Advanced Purification Technology for Caspofungin Acetate Impurity D: Ensuring Pharmaceutical Quality Control
Advanced Purification Technology for Caspofungin Acetate Impurity D: Ensuring Pharmaceutical Quality Control
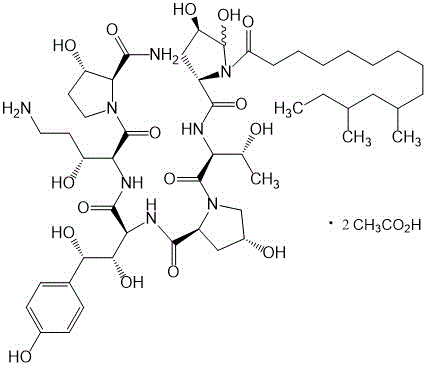
The pharmaceutical industry relies heavily on the availability of high-purity reference standards to ensure the safety and efficacy of antifungal medications. Patent CN113801203A introduces a groundbreaking preparation method for Caspofungin Acetate Impurity D, a critical degradation product used for quality control in echinocandin drug manufacturing. This technology addresses the longstanding challenge of isolating this specific isomer with sufficient purity for analytical validation. By leveraging a specialized preparative chromatography system, the process achieves a purity level exceeding 95%, providing a reliable source for pharmaceutical laboratories. The method utilizes a carefully optimized mobile phase system to maintain the structural stability of the impurity during separation. This innovation not only supports regulatory compliance but also enhances the reliability of impurity profiling in final drug products.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining high-purity Caspofungin Acetate Impurity D has been a significant bottleneck for quality control departments. Conventional isolation techniques often struggle with the inherent instability of the molecule under neutral or alkaline conditions. The electropositive nature of the carbon atom at position 10 makes it highly susceptible to nucleophilic attack by hydroxide ions, leading to the detachment of the ethylenediamine side chain. Furthermore, lone-pair electrons on the nitrogen atom at position 24 can intramolecularly attack position 10, forming an unwanted five-membered ring structure. These degradation pathways result in complex mixtures where the target impurity is contaminated with various isomers and degradation products. Standard purification protocols frequently fail to resolve these closely related species, yielding materials unsuitable for use as quantitative reference standards.
The Novel Approach
The novel approach detailed in the patent overcomes these stability issues through precise control of the chromatographic environment. By employing a mobile phase containing 0.1% acetic acid, the process creates an acidic environment that effectively suppresses the nucleophilic attack responsible for degradation. This strategic adjustment ensures that the impurity remains intact throughout the separation process. Additionally, the use of a specific gradient elution program allows for the sequential washing of early-eluting impurities before the target compound is collected. The integration of SP-120-10-ODS-BP filler provides superior resolution capabilities compared to standard C18 columns. This combination of acidic stabilization and high-resolution chromatography enables the consistent production of solid powder with purity levels surpassing 95%, meeting the rigorous demands of modern pharmaceutical analysis.
Mechanistic Insights into Acidic Stabilization and Chromatographic Separation
The core mechanism behind this successful purification lies in the manipulation of pH to control chemical reactivity. As described in the background art, the formation of Impurity D involves a complex rearrangement where hydroxyl isomerization contributes to intermediate stabilization. In a basic environment, the concentration of hydroxide ions accelerates the hydrolysis of the ethylenediamine linkage. The introduction of 0.1% acetic acid shifts the equilibrium, protonating potential nucleophiles and significantly reducing the rate of degradation. This chemical stabilization is crucial because it allows the molecule to withstand the residence time within the chromatographic column without decomposing. Without this acidic modifier, the target peak would likely broaden or split due to on-column degradation, rendering the separation ineffective. The patent data confirms that under alkaline conditions, product purity drops drastically, validating the necessity of the acidic mobile phase for maintaining molecular integrity.
From a separation science perspective, the choice of SP-120-10-ODS-BP as the stationary phase is equally critical. This specific filler offers a unique balance of surface area and pore size that optimizes the interaction between the analyte and the stationary phase. The gradient elution strategy, moving from 30% to 50% acetonitrile, exploits the differential hydrophobicity of the components in the crude mixture. Early eluting contaminants are removed at lower organic solvent concentrations, while the target Impurity D, which exhibits higher hydrophobicity due to its lipopeptide structure, is retained until the organic phase concentration increases. This stepwise increase in elution strength ensures that the target compound is separated from both early and late-eluting impurities. The result is a sharp, well-defined peak that can be collected with high confidence, minimizing the need for reprocessing and maximizing overall yield.
How to Synthesize Caspofungin Acetate Impurity D Efficiently
The synthesis and purification workflow outlined in the patent provides a clear roadmap for producing this critical reference standard. The process begins with the dissolution of crude material in the stabilized acidic medium, followed by filtration to protect the chromatography column. The subsequent separation relies on a medium-to-low pressure liquid phase system, which is more accessible for scale-up than high-pressure analytical systems. Operators must strictly adhere to the gradient profile to ensure the removal of interfering substances before collecting the target fraction. Finally, the eluent is subjected to freeze-drying to remove solvents without exposing the heat-sensitive product to high temperatures. For a detailed breakdown of the specific operational parameters and step-by-step instructions, please refer to the standardized guide below.
- Dissolve crude Caspofungin Acetate Impurity D in 0.1% acetic acid aqueous solution and filter through a 0.45 μm membrane to remove insoluble particulates.
- Load the filtered solution onto a preparative chromatography column packed with SP-120-10-ODS-BP filler and equilibrate with 30% acetonitrile mobile phase.
- Perform gradient elution increasing acetonitrile from 30% to 50%, collect the target fraction, and freeze-dry to obtain solid powder with >95% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented purification method offers substantial strategic benefits beyond mere technical performance. The reliance on standard chromatographic equipment and commercially available solvents like acetonitrile and acetic acid simplifies the sourcing of raw materials. There is no dependency on exotic reagents or specialized catalysts that might be subject to supply chain disruptions. The robustness of the method means that production batches are highly reproducible, reducing the risk of batch failures that can delay project timelines. Furthermore, the ability to produce high-purity material in-house or through a qualified partner reduces the lead time associated with importing scarce reference standards from overseas suppliers. This localization of supply capability enhances the resilience of the quality control supply chain.
- Cost Reduction in Manufacturing: The elimination of complex synthetic steps required to generate the impurity from scratch represents a significant cost saving. Instead of multi-step organic synthesis, the process utilizes crude intermediates that are concentrated and purified. This reduction in synthetic complexity lowers the consumption of expensive starting materials and reduces labor hours. Additionally, the high recovery yield minimizes waste generation, further driving down the cost per gram of the final product. The use of standard solvents also facilitates solvent recovery and recycling programs, contributing to long-term operational expense reductions.
- Enhanced Supply Chain Reliability: The scalability of the chromatographic process ensures that supply can be ramped up quickly to meet fluctuating demand. Unlike fermentation-based processes that have long cycle times, this purification method can be executed in a matter of hours. This agility allows suppliers to respond rapidly to urgent requests for reference standards from regulatory bodies or internal QC labs. The stability of the product under the defined storage and processing conditions also extends shelf life, reducing inventory write-offs and ensuring that stock remains viable for longer periods.
- Scalability and Environmental Compliance: The process is designed with environmental considerations in mind, utilizing solvents that are easier to manage and dispose of compared to halogenated alternatives often found in older methods. The absence of heavy metal catalysts eliminates the need for costly and complex metal scavenging steps, simplifying waste treatment protocols. As production scales from laboratory to commercial volumes, the linear scalability of column chromatography ensures that environmental impact remains proportional to output. This alignment with green chemistry principles supports corporate sustainability goals and ensures compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Caspofungin Acetate Impurity D. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is essential for quality assurance teams evaluating the suitability of this material for their specific analytical methods. The information covers stability profiles, purity specifications, and process scalability to provide a comprehensive overview for decision-makers.
Q: Why is acidic mobile phase critical for Caspofungin Acetate Impurity D stability?
A: The patent highlights that the carbon atom at position 10 connected to ethylenediamine is electropositive. Under alkaline conditions, hydroxide ions attack this carbon, causing ethylenediamine detachment and ring formation. Acidic conditions (0.1% acetic acid) suppress this nucleophilic attack, ensuring structural integrity during purification.
Q: What is the achievable purity level using this chromatographic method?
A: By utilizing the specific gradient elution profile with SP-120-10-ODS-BP filler, the process consistently yields Caspofungin Acetate Impurity D with a purity exceeding 95%, meeting stringent requirements for analytical reference standards.
Q: Is this preparation method suitable for large-scale manufacturing?
A: Yes, the method employs medium-to-low pressure liquid chromatography with stable operating conditions. The use of common solvents like acetonitrile and acetic acid, combined with robust column packing, facilitates scalability from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caspofungin Acetate Impurity D Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurity standards play in the development and release of life-saving antifungal therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of the project stage. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in chromatographic purification allows us to replicate and optimize processes like the one described in CN113801203A, guaranteeing consistency and reliability in every batch we supply to our global partners.
We invite you to collaborate with us to secure a stable supply of this essential reference standard. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs and quality requirements. Please contact us today to request specific COA data and route feasibility assessments for your upcoming projects. Let us help you streamline your quality control operations with our premium pharmaceutical intermediates.
