Advanced Manufacturing Protocol for High-Purity Dansyl Acid and Commercial Scalability
Advanced Manufacturing Protocol for High-Purity Dansyl Acid and Commercial Scalability
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for higher purity intermediates that serve as the backbone for advanced diagnostic and pharmaceutical applications. A pivotal development in this sector is documented in patent CN102875426A, which outlines a robust and scientifically rational method for the synthesis and purification of dansyl acid (5-dimethylaminonaphthalene-1-sulfonic acid). This compound is a critical precursor in the production of fluorescence labels, widely utilized in bioconjugation and protein analysis. The technical breakthrough described in this patent addresses long-standing challenges in yield optimization and impurity control, offering a pathway that is not only chemically efficient but also commercially viable for large-scale production. By leveraging a specific molar ratio of reagents and a controlled temperature profile, this method ensures the consistent delivery of high-quality tabular crystals, meeting the rigorous demands of global supply chains for reliable dansyl acid supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfonic acid derivatives like dansyl acid has been plagued by inconsistent reaction kinetics and difficult purification processes. Traditional methods often rely on harsh alkaline conditions that can lead to the hydrolysis of the sulfonic acid group or the formation of complex polymeric byproducts, significantly reducing the overall yield. Furthermore, conventional purification techniques frequently involve extensive column chromatography or multiple solvent extractions, which are not only time-consuming but also generate substantial chemical waste, posing environmental and cost burdens. The lack of precise temperature control in older protocols often results in the over-methylation of the amine group or incomplete reaction, leading to an impurity profile that is unacceptable for high-sensitivity fluorescence applications. These inefficiencies create bottlenecks in the supply chain, causing delays and increased costs for procurement managers seeking cost reduction in fluorescence label manufacturing.
The Novel Approach
The methodology presented in patent CN102875426A introduces a refined approach that mitigates these issues through precise stoichiometric control and a staged temperature regimen. By utilizing sodium bicarbonate as a buffering agent, the reaction environment is maintained at a pH that favors mono-methylation while suppressing side reactions. The process involves a low-temperature initiation phase followed by a controlled heating period, which ensures complete conversion of the starting material without degrading the product. This novel approach simplifies the downstream processing significantly; instead of complex chromatographic separations, the product is isolated through acidification and recrystallization. This shift from complex separation to crystallization-based purification drastically reduces the operational complexity and solvent usage, aligning with modern green chemistry principles and offering substantial cost savings in dansyl acid manufacturing.
Mechanistic Insights into Bicarbonate-Buffered N-Methylation
The core of this synthesis lies in the nucleophilic substitution reaction where the amino group of 1-naphthylamine-5-sulfonic acid attacks the methyl group of dimethyl sulfate. The presence of sodium bicarbonate is mechanistically crucial; it serves to neutralize the methyl sulfuric acid formed as a byproduct, thereby driving the equilibrium forward according to Le Chatelier's principle. Without this buffering capacity, the accumulation of acid would protonate the amine, rendering it non-nucleophilic and stalling the reaction. The specific molar ratio of 4:1:2 (sodium bicarbonate:amine:dimethyl sulfate) ensures that there is a sufficient excess of base to maintain the reaction pH throughout the 5-hour low-temperature phase and the subsequent overnight heating at 60 to 70°C. This careful balance prevents the formation of quaternary ammonium salts, which are common impurities in methylation reactions, thus ensuring the structural integrity of the final dansyl acid molecule.
Impurity control is further enhanced by the specific workup procedure involving hydrochloric acid. After the reaction is complete, the mixture is acidified to precipitate the crude product, which effectively separates the organic sulfonic acid from inorganic salts and water-soluble impurities. The subsequent step of boiling the filter cake with dilute hydrochloric acid is a critical purification mechanism; it dissolves the product while leaving behind insoluble mechanical impurities or polymeric tars. Upon cooling and recrystallization, the dansyl acid forms distinct tabular crystals, a physical characteristic indicative of high crystallinity and purity. This mechanism ensures that the final product meets the stringent purity specifications required for use as a high-purity OLED material or pharmaceutical intermediate, minimizing the risk of fluorescence quenching in downstream applications.
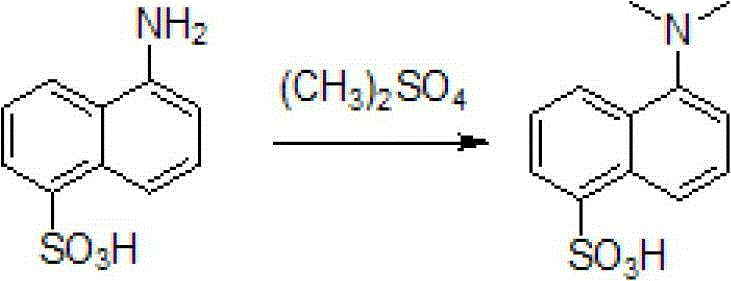
How to Synthesize Dansyl Acid Efficiently
The synthesis of dansyl acid via this patented route requires strict adherence to the specified addition rates and temperature profiles to ensure safety and reproducibility. The process begins with the preparation of an aqueous bicarbonate solution, into which the sulfonic acid starting material is introduced to form a homogeneous suspension. The addition of dimethyl sulfate must be performed dropwise under low-temperature conditions to manage the exothermic nature of the methylation reaction, preventing thermal runaway. Following the initial reaction period, the temperature is raised to facilitate the completion of the methylation, after which the product is isolated through acidification and purified via a double recrystallization process. For detailed standardized synthesis steps, please refer to the guide below.
- Dissolve sodium bicarbonate in water and add 1-naphthylamine-5-sulfonic acid slowly under stirring to form a buffered mixture.
- Add dimethyl sulfate dropwise at low temperature, maintain reaction for 5 hours, then heat to 60-70°C overnight.
- Acidify with concentrated hydrochloric acid, filter the solid, boil filter cake with dilute acid, and recrystallize twice to obtain pure crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis protocol offers significant advantages for procurement managers and supply chain heads focused on stability and efficiency. The reliance on commercially available raw materials such as sodium bicarbonate, dimethyl sulfate, and 1-naphthylamine-5-sulfonic acid ensures that the supply chain is not vulnerable to the shortages of exotic catalysts or specialized reagents. This accessibility translates directly into enhanced supply chain reliability, as sourcing can be diversified across multiple global vendors without compromising the quality of the input materials. Furthermore, the simplified purification process reduces the dependency on expensive chromatography resins and large volumes of organic solvents, leading to a more streamlined manufacturing workflow that is less prone to operational delays.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the use of inexpensive inorganic bases significantly lower the operational expenditure associated with production. By avoiding the need for transition metal catalysts, the process also removes the costly and time-consuming step of heavy metal removal, which is often required to meet pharmaceutical regulatory standards. This qualitative improvement in process efficiency results in substantial cost savings, allowing for more competitive pricing structures in the market for commercial scale-up of complex polymer additives and fine chemicals.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate standard industrial equipment and common reagents, minimizes the risk of batch failures due to equipment incompatibility or reagent instability. The high yield reported in the patent examples indicates a material-efficient process, reducing the volume of raw materials required per unit of output and mitigating the impact of raw material price fluctuations. This stability is crucial for reducing lead time for high-purity dansyl acid derivatives, ensuring that downstream manufacturers can maintain consistent production schedules without interruption.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing aqueous media and standard heating protocols that are easily transferred from laboratory to pilot and commercial scales. The reduced use of organic solvents and the generation of manageable inorganic waste streams align with increasingly strict environmental regulations, reducing the compliance burden on manufacturing facilities. This environmental compatibility not only safeguards the company against regulatory risks but also enhances the brand value associated with sustainable manufacturing practices in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of dansyl acid based on the patented methodology. These insights are derived from the specific process parameters and beneficial effects detailed in the intellectual property documentation, providing clarity for technical decision-makers. Understanding these aspects is essential for evaluating the feasibility of integrating this intermediate into broader manufacturing workflows.
Q: What is the primary advantage of using sodium bicarbonate in this dansyl acid synthesis?
A: Sodium bicarbonate acts as a mild buffering base that neutralizes the sulfuric acid byproduct generated during methylation, preventing the degradation of the sensitive naphthalene ring and ensuring a cleaner reaction profile compared to stronger bases.
Q: How does the recrystallization process impact the final purity of the dansyl acid?
A: The double recrystallization step, specifically boiling the filter cake with dilute hydrochloric acid followed by cooling, effectively removes unreacted starting materials and dimethylated byproducts, achieving the stringent purity specifications required for fluorescence labeling applications.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly outlines a scalable protocol using common industrial reagents and standard temperature controls, avoiding exotic catalysts or high-pressure equipment, which facilitates straightforward commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dansyl Acid Supplier
The technical potential of the synthesis route described in patent CN102875426A is immense, offering a pathway to high-quality dansyl acid that meets the exacting standards of the global pharmaceutical and diagnostic industries. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that this laboratory-grade protocol is seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of dansyl acid delivered to our partners is free from critical impurities and ready for immediate use in sensitive fluorescence applications.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for fluorescence labels and pharmaceutical intermediates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical confidence and commercial security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
