Advanced Purification Strategies for Semaglutide: Enhancing Yield and Commercial Scalability
The pharmaceutical industry is currently witnessing an unprecedented surge in demand for glucagon-like peptide-1 (GLP-1) receptor agonists, driven by their dual efficacy in managing type II diabetes and facilitating weight loss. At the forefront of this therapeutic revolution is Semaglutide, a long-acting analog that requires sophisticated downstream processing to meet stringent regulatory standards. Patent CN112175068A introduces a groundbreaking method for purifying Semaglutide that addresses critical bottlenecks in solubility and impurity clearance. This technical insight report analyzes the proprietary two-step chromatographic approach detailed in the patent, highlighting its potential to redefine manufacturing efficiency for high-purity peptide intermediates. By shifting from unstable ammonia-based systems to a controlled phosphate buffer environment, this innovation offers a robust pathway for reliable API intermediate supplier operations seeking to optimize their production lines for complex polypeptide drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification protocols for polypeptide drugs like Semaglutide have long struggled with the physicochemical instability of crude peptide samples during the dissolution phase. Conventional methods often rely on dilute ammonia water or pure acetonitrile to solubilize the crude material, which frequently results in unpredictable pH fluctuations and poor solution stability. This instability leads to the precipitation of the peptide during the loading phase, causing severe blockage of chromatographic columns and associated equipment, which drastically halts production flow. Furthermore, the low solubility limits the sample loading concentration, often forcing manufacturers to process smaller batches to avoid column damage, thereby inflating the cost reduction in pharmaceutical manufacturing. The inability to effectively control the pH of the dissolved crude peptide solution also compromises the separation resolution, making it difficult to remove specific structural impurities that closely resemble the target molecule.
The Novel Approach
The innovative method disclosed in the patent fundamentally reengineers the dissolution and purification workflow by introducing a phosphate buffer solution containing an organic solvent as the primary dissolution medium. This specific solvent system maintains a stable pH environment, typically adjusted to between 7 and 8 using triethylamine, which prevents the crude peptide from precipitating even at high concentrations. By ensuring the crude peptide solution remains clear and stable, the process eliminates the risk of column blockage and allows for a significant increase in sample loading capacity, reaching up to 50 g/L in optimized embodiments. This stability is further enhanced by matching the composition of the dissolution buffer with the initial mobile phase of the first purification step, minimizing band broadening at the column head. Consequently, this novel approach not only improves the separation effect but also streamlines the entire downstream process, offering a scalable solution for the commercial scale-up of complex polymer additives and peptide therapeutics.
Mechanistic Insights into Reversed-Phase Chromatographic Purification
The core of this purification strategy lies in a sophisticated two-step reversed-phase chromatography sequence that leverages distinct stationary phases to target different classes of impurities. The first purification step utilizes octaalkylsilane bonded silica filler, which provides a specific hydrophobic interaction profile ideal for separating the target peptide from fragment impurities and defective sequences. By employing a gradient elution with a phosphate buffer containing an organic solvent as phase A and acetonitrile as phase B, the system effectively resolves the main product from early-eluting impurities. This stage is critical for removing bulk contaminants such as deletion sequences, ensuring that the intermediate fraction achieves a purity of greater than 95% before proceeding to the final polishing step. The careful selection of triethylamine phosphate as the buffer salt system further inhibits peak tailing, enhancing the resolution and recovery of the target compound.
The second purification step is specifically engineered to address the most challenging impurities: racemic variants that arise during synthesis. This stage employs octadecylsilane bonded silica filler, which offers higher hydrophobicity and resolution power, paired with a dilute acetic acid solution and acetonitrile gradient. This change in mobile phase chemistry is crucial for separating diastereomers and racemates, such as the His1 and Ala18 racemization impurities that are structurally nearly identical to Semaglutide. The mechanism relies on the subtle differences in interaction between the chiral centers of the impurities and the hydrophobic stationary phase under acidic conditions. As demonstrated in the patent data, this step successfully reduces single impurity levels to less than 0.1%, achieving a final purity exceeding 99.5%. 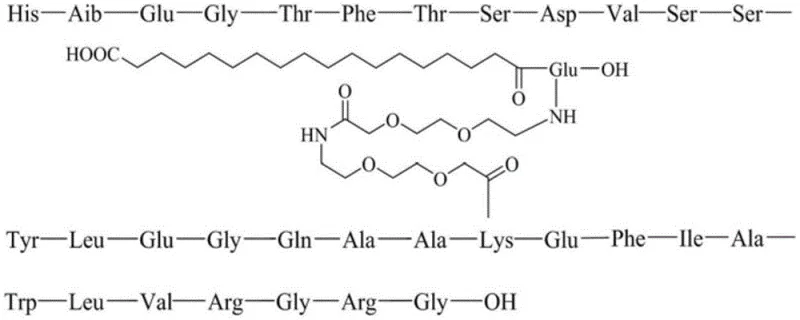
How to Synthesize High-Purity Semaglutide Efficiently
Implementing this purification protocol requires precise control over buffer preparation and chromatographic parameters to replicate the high yields reported in the patent examples. The process begins with the preparation of a dissolving buffer where phosphoric acid and triethylamine are used to establish a stable pH of 8.0, followed by the addition of acetonitrile to enhance solubility. Once the crude peptide is dissolved and filtered through a 0.22-micron organic membrane, it is loaded onto the first column for gradient elution, collecting fractions that meet the initial purity threshold. The detailed standardized synthesis steps see the guide below for specific flow rates, gradient slopes, and column dimensions that ensure reproducibility.
- Dissolve crude Semaglutide in a phosphate buffer solution containing an organic solvent to ensure stability and prevent precipitation.
- Perform first-step purification using octaalkylsilane bonded silica filler with gradient elution to remove fragment impurities.
- Execute second-step purification using octadecylsilane bonded silica filler to eliminate racemic impurities and achieve final purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates into tangible operational efficiencies that directly impact the bottom line without compromising quality. The primary advantage lies in the drastic simplification of the dissolution process, which eliminates the need for complex pH adjustments and reduces the risk of batch failure due to precipitation. By enabling higher loading concentrations, the method significantly increases throughput per batch, allowing manufacturers to produce more active pharmaceutical ingredient within the same timeframe and equipment footprint. This efficiency gain leads to substantial cost savings in manufacturing by reducing solvent consumption and energy usage associated with processing larger volumes of dilute solutions. Furthermore, the robustness of the phosphate buffer system ensures consistent performance across different batches, enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of unstable dissolution conditions means that expensive chromatography columns are protected from blockage and damage, extending their operational lifespan and reducing capital expenditure on replacement parts. Additionally, the ability to load the column with higher concentrations of crude peptide reduces the total volume of mobile phase required for elution, leading to significant savings in solvent procurement and waste disposal costs. The high recovery yield reported in the patent examples indicates that less raw material is wasted during purification, further optimizing the cost structure of the final API. These qualitative improvements collectively drive down the unit cost of production, making the supply of Semaglutide more economically viable.
- Enhanced Supply Chain Reliability: The stability of the crude peptide solution in the new buffer system ensures that the manufacturing process is less susceptible to interruptions caused by equipment fouling or filtration issues. This reliability is critical for maintaining continuous production schedules, which is a key concern for supply chain heads managing global inventory levels. By minimizing the risk of batch rejection due to poor separation or column failure, the method guarantees a more consistent output of qualified product. This consistency allows for better forecasting and planning, ensuring that downstream formulation teams receive their materials on time without unexpected delays caused by purification bottlenecks.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reversed-phase fillers and common solvent systems that are easily adapted from laboratory to industrial scale. The use of triethylamine and phosphate buffers, while requiring proper waste management, is well-understood in the industry and facilitates compliance with environmental regulations regarding effluent treatment. The high purity achieved in just two steps reduces the need for additional polishing rounds, which in turn minimizes the overall environmental footprint of the manufacturing process. This alignment with green chemistry principles supports the long-term sustainability goals of modern pharmaceutical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this purification method, based on the specific data and embodiments provided in the patent documentation. These answers are designed to clarify the operational advantages and technical feasibility for R&D and production teams evaluating this technology for adoption. Understanding these details is essential for assessing the compatibility of this method with existing manufacturing infrastructure and quality control standards.
Q: How does the new purification method improve crude peptide solubility?
A: The method utilizes a phosphate buffer solution with an organic solvent at a controlled pH, which significantly stabilizes the crude peptide solution compared to traditional ammonia water systems, preventing column blockage and allowing higher loading concentrations up to 50 g/L.
Q: What specific impurities are targeted in the two-step process?
A: The first step primarily removes fragment impurities and defective peptides, while the second step is specifically designed to eliminate difficult-to-remove racemic impurities such as His1 racemization and Ala18 racemization, ensuring final purity exceeds 99.5%.
Q: Is this purification process suitable for large-scale commercial production?
A: Yes, the process demonstrates strong operability and stability with high yields (approximately 73.86% in validation examples), making it highly viable for scaling up from laboratory synthesis to industrial manufacturing of GLP-1 analogs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Semaglutide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust purification technologies in the production of high-value peptide therapeutics like Semaglutide. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into efficient industrial processes. We are committed to meeting stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical equipment to verify the absence of racemic impurities and deletion sequences. Our capability to handle complex purification challenges makes us an ideal partner for companies seeking to secure a stable supply of high-quality GLP-1 analogs.
We invite you to collaborate with us to optimize your supply chain and achieve significant operational efficiencies. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your commercial goals. Let us help you navigate the complexities of peptide production with confidence and precision.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
