Scalable Synthesis of Novel Anthraquinone Imidazole Riboside Analogues for Oncology Applications
Scalable Synthesis of Novel Anthraquinone Imidazole Riboside Analogues for Oncology Applications
The pharmaceutical industry is constantly seeking next-generation anticancer agents that retain the potent efficacy of traditional anthracyclines while mitigating their severe side effects, particularly cardiotoxicity. Patent CN108822170B introduces a groundbreaking class of anthraquinone imidazole riboside analogues that represent a significant leap forward in medicinal chemistry. This technology leverages a unique structural modification where an imidazole ring serves as a linker between the anthraquinone chromophore and various sugar moieties. This innovation not only preserves the critical DNA intercalation capability of the anthraquinone core but also dramatically improves the water solubility and bioavailability of the final compounds. For R&D directors and procurement specialists in the oncology sector, this patent offers a robust pathway to develop high-purity pharmaceutical intermediates with enhanced therapeutic indices. The synthesis described provides a reliable foundation for creating novel nucleoside analogues that could redefine treatment protocols for leukemia and lung cancer.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional anthracycline antibiotics, such as Daunorubicin and Doxorubicin, have long been the cornerstone of chemotherapy regimens due to their broad-spectrum antitumor activity. However, their clinical utility is severely constrained by dose-dependent cardiotoxicity and the development of multidrug resistance. These conventional molecules rely on a natural O-glycosidic bond to connect the sugar to the aglycone, a linkage that is often metabolically unstable and contributes to the generation of reactive oxygen species in cardiac tissue. Furthermore, the reliance on fermentation processes for the production of natural anthracycline precursors introduces significant variability in supply chains and limits the ability to structurally modify the molecule for improved safety profiles. The extraction and purification of these natural products are resource-intensive, leading to high manufacturing costs and inconsistent batch-to-batch quality, which poses a significant risk for supply chain heads managing global inventory.
The Novel Approach
The technology disclosed in CN108822170B circumvents these limitations by employing a fully synthetic route to construct the anthraquinone-imidazole-sugar architecture. By replacing the traditional glycosidic oxygen with a nitrogen-containing imidazole ring, the resulting analogues exhibit superior chemical stability and altered metabolic pathways. This structural innovation allows for the precise attachment of diverse pentose and hexose sugars, enabling fine-tuning of the pharmacokinetic properties without the constraints of natural biosynthesis. The synthetic approach ensures a consistent and scalable supply of high-purity intermediates, independent of biological fermentation variables. For procurement managers, this shift from extraction to synthesis translates into a more predictable cost structure and reduced dependency on agricultural or biological raw materials, thereby enhancing supply chain resilience and enabling cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Imidazole-Mediated Glycosylation
The core of this technological advancement lies in the strategic construction of the imidazole ring fused to the anthraquinone system, followed by a highly efficient glycosylation step. The synthesis begins with the nitration and subsequent reduction of 1,4-dihydroxy-9,10-anthraquinone to form a diamine intermediate, which is then cyclized with formic acid to generate the 4,11-dihydroxyanthra[2,3-d]imidazole-5,11-dione core. This core structure retains the planar tetracyclic system essential for DNA intercalation, ensuring that the fundamental mechanism of action against cancer cells is preserved. The imidazole nitrogen at the 1-position then acts as a nucleophile in the subsequent glycosylation reaction. This nitrogen atom is less basic than typical amines but sufficiently nucleophilic under Lewis acid catalysis to attack activated glycosyl donors, forming a stable N-glycosidic bond that is resistant to enzymatic hydrolysis in plasma.
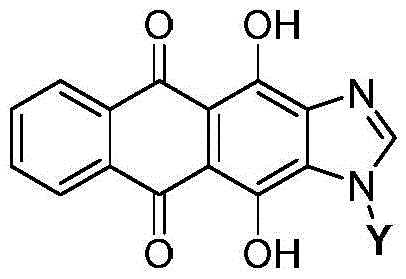
The glycosylation step is critical for imparting water solubility and targeting specificity to the molecule. The patent details multiple catalytic systems to achieve this coupling, including the use of trimethylsilyl trifluoromethanesulfonate (TMSOTf) with DBU or BSA, as well as an N-iodosuccinimide (NIS) promoted pathway. These methods allow for the attachment of peracetylated sugars with high stereocontrol, ensuring the formation of the desired beta-anomers which are crucial for biological activity. The ability to use various protected sugars, such as tetraacetylribose or peracetylmannose, provides a modular platform for generating a library of analogues. This modularity is invaluable for R&D teams aiming to optimize the impurity profile and potency of the final drug candidate. The subsequent deprotection using sodium methoxide is a mild and efficient process that yields the final hydroxyl-functionalized analogues without degrading the sensitive anthraquinone core.
![Synthetic route for the glycosyl acceptor 4,11-dihydroxyanthra[2,3-d]imidazole-5,11-dione from anthraquinone](/insights/img/anthraquinone-imidazole-riboside-synthesis-pharma-supplier-20260315011027-06.webp)
How to Synthesize Anthraquinone Imidazole Riboside Efficiently
The synthesis of these complex nucleoside analogues requires precise control over reaction conditions to ensure high yields and purity. The process involves a sequential multi-step pathway starting from readily available anthraquinone derivatives. The initial functionalization of the anthraquinone ring requires careful temperature control during nitration and reduction to prevent over-oxidation or side reactions. The cyclization to form the imidazole ring is driven by refluxing in formic acid, a step that demands efficient removal of water to push the equilibrium towards product formation. Once the aglycone acceptor is prepared, the glycosylation reaction must be performed under anhydrous conditions to prevent hydrolysis of the activated sugar donor. The detailed standardized synthetic steps for this process are outlined below, providing a clear roadmap for laboratory scale-up and process optimization.
- Synthesize the glycosyl acceptor 4,11-dihydroxyanthra[2,3-d]imidazole-5,11-dione via nitration and reduction of 1,4-dihydroxy-9,10-anthraquinone.
- Perform glycosylation using peracetylated sugars with promoters like TMSOTf or NIS/TfOH to attach the sugar moiety to the imidazole nitrogen.
- Deprotect the acetyl groups using sodium methoxide in methanol to yield the final hydroxyl-functionalized anthraquinone imidazole riboside analogues.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from natural extraction to this fully synthetic route offers profound strategic advantages. The reliance on fermentation for traditional anthracyclines creates bottlenecks related to biological variability, long lead times, and complex purification workflows. In contrast, the synthetic pathway described in this patent utilizes commodity chemicals and standard organic synthesis techniques that are easily scalable in existing fine chemical manufacturing facilities. This shift eliminates the risk of crop failure or strain degradation associated with biological sources, ensuring a continuous and reliable supply of critical intermediates. Furthermore, the synthetic route allows for tighter control over impurity profiles, reducing the burden on quality control laboratories and minimizing the risk of batch rejection. This reliability is essential for maintaining the continuity of drug production schedules in a highly regulated pharmaceutical environment.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive natural extraction and fermentation processes, which are capital-intensive and have low throughput. By using readily available starting materials like 1,4-dihydroxy-9,10-anthraquinone and common glycosyl donors, the raw material costs are significantly stabilized. The elimination of transition metal catalysts in favor of organic promoters like TMSOTf and DBU further reduces the cost associated with heavy metal removal and waste treatment. This streamlined process results in substantial cost savings across the entire manufacturing value chain, allowing for more competitive pricing of the final API intermediates without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Synthetic manufacturing is inherently more predictable than biological fermentation, as it is not subject to the vagaries of biological growth cycles or contamination risks. This predictability allows for more accurate forecasting and inventory management, reducing the need for safety stock and freeing up working capital. The ability to source raw materials from multiple chemical suppliers rather than a single biological source diversifies the supply base and mitigates the risk of disruption. For supply chain heads, this means a more resilient procurement strategy that can withstand market fluctuations and ensure the timely delivery of high-purity API intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are amenable to large-scale production, with steps like nitration, reduction, and glycosylation being well-understood unit operations in the fine chemical industry. The use of standard solvents like dichloromethane and acetonitrile allows for efficient recovery and recycling, minimizing solvent waste and environmental impact. The avoidance of toxic heavy metals simplifies the waste treatment process and ensures compliance with increasingly stringent environmental regulations. This scalability ensures that the production can be ramped up from kilograms to tons to meet commercial demand without the need for significant process re-engineering or new facility construction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel anthraquinone imidazole riboside analogues. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating these intermediates into their drug development pipelines. The information covers aspects of synthesis, biological activity, and regulatory considerations to provide a comprehensive overview of the technology's potential.
Q: What is the primary advantage of introducing an imidazole ring to anthraquinone structures?
A: Introducing an imidazole ring allows for the linkage of various sugar moieties, significantly enhancing water solubility and biological activity while retaining the DNA intercalation properties of the anthraquinone core.
Q: How does this synthetic route address the cardiotoxicity associated with traditional anthracyclines?
A: By modifying the glycosidic linkage through an imidazole bridge rather than a traditional O-glycosidic bond, the resulting analogues exhibit altered pharmacokinetics and reduced accumulation in cardiac tissue, potentially lowering cardiotoxicity.
Q: Is this synthesis suitable for large-scale commercial production?
A: Yes, the route utilizes common reagents like nitric acid, formic acid, and standard glycosyl donors, avoiding rare natural extraction, which makes it highly amenable to cost-effective commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anthraquinone Imidazole Riboside Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation oncology therapeutics. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the complex synthesis of anthraquinone imidazole riboside analogues can be executed with precision and efficiency. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify identity and potency. Our capability to handle sensitive glycosylation reactions and multi-step heterocyclic synthesis makes us an ideal partner for bringing these innovative compounds from the laboratory to the clinic.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize their supply chains for these valuable intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can support your drug development goals. By partnering with us, you gain access to a reliable source of complex chemical intermediates that can accelerate your timeline to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
