Advanced Cytarabine Structural Analogs: Enhancing Oral Bioavailability and Commercial Scalability
The pharmaceutical landscape for acute myeloid leukemia treatment is undergoing a significant transformation driven by the need for more patient-compliant oral therapies. Patent CN112409431B introduces a groundbreaking series of cytarabine structural analogs, specifically designated as P1 through P4, which address the critical pharmacokinetic limitations of the parent drug. Traditional cytarabine suffers from extremely low oral bioavailability and a very short plasma half-life, necessitating continuous intravenous infusion which burdens hospital resources and patient quality of life. These novel analogs employ a sophisticated phosphoramidate prodrug approach that masks the polar phosphate group, thereby facilitating passive diffusion across cell membranes. Once inside the target leukemia cells, enzymatic cleavage releases the active triphosphate metabolite at concentrations significantly higher than those achieved with standard cytarabine hydrochloride. This technological leap not only promises enhanced therapeutic efficacy but also opens new avenues for outpatient management of hematological malignancies, representing a pivotal shift in oncology intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Cytarabine, a cornerstone in leukemia chemotherapy, faces severe physicochemical challenges that hinder its optimal clinical deployment. The molecule possesses high polarity due to its multiple hydroxyl groups and the cytosine base, resulting in poor permeability across the lipid bilayer of the intestinal epithelium. Consequently, oral administration yields negligible systemic exposure, forcing reliance on invasive intravenous routes. Furthermore, cytarabine is rapidly inactivated by cytosine deaminase in the liver and blood, converting it into the inactive uracil arabinoside within minutes. This rapid metabolic clearance demands high-frequency dosing or continuous infusion pumps to maintain therapeutic levels, which increases the risk of dose-dependent toxicities such as myelosuppression and neurotoxicity. The logistical complexity of IV administration limits treatment to specialized clinical settings, creating bottlenecks in healthcare delivery and increasing the overall cost of care for chronic management protocols.
The Novel Approach
The innovative strategy outlined in the patent data circumvents these biological barriers through rational molecular design. By modifying the 5'-hydroxyl position of the arabinose sugar with a lipophilic phosphoramidate moiety, the new analogs effectively bypass the rate-limiting first phosphorylation step typically catalyzed by deoxycytidine kinase. This ProTide technology allows the drug to enter cells as a neutral species, avoiding efflux transporters that often pump charged nucleotides out of the cell. 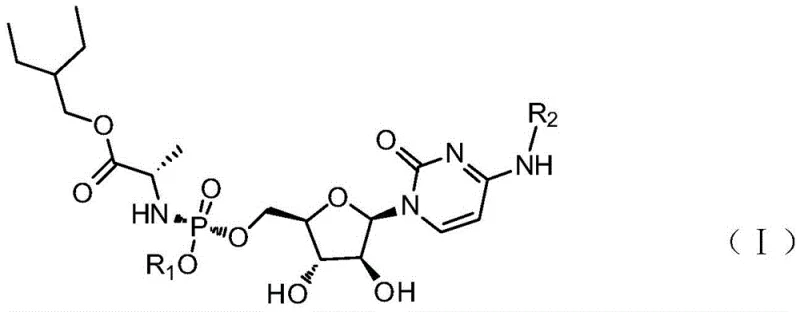 . Once intracellular, esterases and phosphoramidases sequentially cleave the masking groups to release the active monophosphate, which is then efficiently converted to the triphosphate form. This mechanism results in sustained intracellular drug concentrations and a markedly prolonged half-life, enabling once-daily oral dosing regimens that could revolutionize patient adherence and treatment outcomes in home-care settings.
. Once intracellular, esterases and phosphoramidases sequentially cleave the masking groups to release the active monophosphate, which is then efficiently converted to the triphosphate form. This mechanism results in sustained intracellular drug concentrations and a markedly prolonged half-life, enabling once-daily oral dosing regimens that could revolutionize patient adherence and treatment outcomes in home-care settings.
Mechanistic Insights into Phosphoramidate Prodrug Activation
The chemical elegance of this synthesis lies in the precise construction of the phosphorus-nitrogen bond which serves as the trigger for intracellular activation. The reaction utilizes phosphoryl chloride derivatives where the chlorine atom acts as a leaving group, allowing for nucleophilic attack by the 5'-hydroxyl of cytarabine under basic conditions. The presence of the 1-methylimidazole base is critical as it scavenges the generated hydrochloric acid, preventing degradation of the acid-sensitive glycosidic bond in the nucleoside structure. The stereochemistry at the phosphorus center is also controlled to ensure consistent metabolic processing, although the patent indicates that mixtures may be formed which are still biologically active. This pathway avoids the use of harsh phosphorylating agents like phosphorus oxychloride directly on the nucleoside without protection, which often leads to extensive decomposition and difficult-to-remove inorganic phosphate impurities. The result is a cleaner reaction profile that simplifies the purification burden for process chemists aiming for GMP compliance.
Impurity control in this synthesis is paramount given the genotoxic potential of many nucleoside analogs. The process described relies on standard organic workup procedures involving aqueous washes to remove water-soluble salts and unreacted starting materials. The use of column chromatography with dichloromethane and methanol gradients allows for the separation of diastereomers and side products such as bis-phosphorylated species. By optimizing the molar ratios of cytarabine to the phosphoryl chloride derivative, specifically maintaining a ratio around 1:1.5, the formation of over-reacted byproducts is minimized. This attention to stoichiometric balance ensures that the final product meets stringent purity specifications required for clinical trials. The stability of the phosphoramidate bond during storage is another key factor, and the solid-state properties of the resulting white powder suggest good handling characteristics for downstream formulation into tablets or capsules without significant degradation over time.
How to Synthesize Cytarabine Structural Analogs Efficiently
The preparation of these high-value intermediates follows a robust two-stage protocol designed for reproducibility and safety. The initial step involves the coupling of the nucleoside with the phosphoramidochloridate reagent at cryogenic temperatures to control exothermicity and selectivity. Following the reaction, a careful quenching and extraction sequence isolates the crude material, which is then subjected to catalytic hydrogenation if benzyl protecting groups are present on the amino acid moiety. 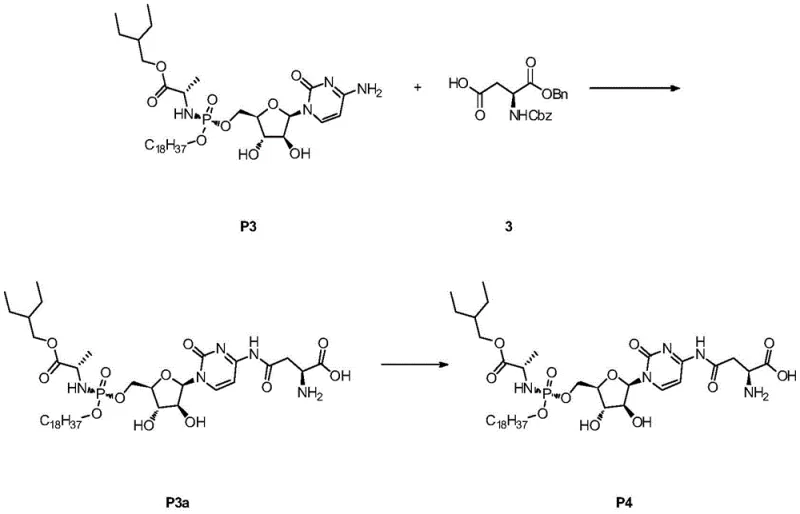 . Detailed standardized operating procedures regarding solvent drying, inert atmosphere maintenance, and filtration techniques are essential to prevent hydrolysis of the sensitive phosphate ester bonds. Operators must adhere to strict temperature controls during the concentration phase to avoid thermal decomposition, ensuring that the final isolated yield maintains the integrity of the chiral centers essential for biological activity.
. Detailed standardized operating procedures regarding solvent drying, inert atmosphere maintenance, and filtration techniques are essential to prevent hydrolysis of the sensitive phosphate ester bonds. Operators must adhere to strict temperature controls during the concentration phase to avoid thermal decomposition, ensuring that the final isolated yield maintains the integrity of the chiral centers essential for biological activity.
- Dissolve cytarabine and phosphoryl chloride derivative in anhydrous tetrahydrofuran under nitrogen protection.
- Cool the mixture to -70 to -78°C and slowly add 1-methylimidazole, then warm to room temperature for reaction.
- Perform post-treatment purification via concentration, washing, and column chromatography to isolate the target analog.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthetic route offers substantial strategic benefits regarding cost structure and supply security. The elimination of transition metal catalysts, which are often required in cross-coupling reactions for nucleoside modification, removes the need for expensive metal scavenging resins and rigorous testing for residual heavy metals. This simplification directly translates to reduced manufacturing costs and shorter batch cycle times, as the validation for metal clearance is no longer a bottleneck. Furthermore, the starting materials, including the specific phosphoryl chloride derivatives and cytarabine, are sourced from established supply chains with high availability, mitigating the risk of raw material shortages that frequently plague specialty chemical markets. The scalability of the process is evidenced by the use of common industrial solvents like tetrahydrofuran and dichloromethane, which are easily recovered and recycled, aligning with modern sustainability goals and waste reduction mandates.
- Cost Reduction in Manufacturing: The process logic inherently drives down operational expenditures by streamlining the purification workflow. Since the reaction proceeds with high selectivity under mild conditions, the burden on downstream processing is significantly alleviated compared to traditional multi-step protection-deprotection sequences. The avoidance of cryogenic reagents beyond the initial cooling phase and the use of ambient temperature for the main reaction duration reduce energy consumption associated with heating and cooling cycles. Additionally, the high purity of the crude product minimizes the loss of valuable material during chromatography, improving the overall mass balance and yield efficiency. These factors collectively contribute to a more economical production model that allows for competitive pricing in the generic oncology intermediate market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Securing a stable supply of critical oncology intermediates is a top priority for pharmaceutical buyers, and this chemistry supports that goal through reagent accessibility. The reliance on commodity chemicals rather than bespoke, single-source reagents ensures that multiple vendors can qualify to supply the necessary inputs, fostering a competitive sourcing environment. The robustness of the reaction to minor variations in moisture or oxygen levels, provided standard inert gas protocols are followed, makes the technology transfer to different manufacturing sites smoother and less prone to failure. This flexibility allows supply chain managers to diversify their manufacturing footprint across different geographic regions, reducing the risk of disruption due to local regulatory changes or logistical bottlenecks. Consequently, lead times for high-purity intermediates can be stabilized, ensuring continuous production schedules for finished dosage forms.
- Scalability and Environmental Compliance: Scaling this synthesis from laboratory to commercial tonnage is facilitated by the absence of hazardous reagents that require special containment or disposal procedures. The waste streams generated are primarily organic solvents and aqueous salt solutions, which can be treated using standard wastewater management infrastructure found in most fine chemical parks. The atom economy of the reaction is favorable, as the majority of the reagent mass is incorporated into the final product or converted into manageable byproducts. This environmental profile simplifies the permitting process for new production lines and aligns with the increasingly strict environmental, social, and governance (ESG) criteria demanded by global pharmaceutical partners. The ability to produce these complex molecules with a lower environmental footprint enhances the corporate reputation of manufacturers and strengthens long-term partnerships with eco-conscious clients.
Frequently Asked Questions (FAQ)
The following inquiries address common technical and commercial considerations regarding the implementation of this technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding the nuances of the prodrug mechanism and the manufacturing requirements is essential for stakeholders evaluating the feasibility of integrating these analogs into their development pipelines. The information below clarifies the operational advantages and the scientific rationale behind the superior performance of these new entities compared to legacy treatments.
Q: How do these cytarabine analogs improve upon traditional therapy?
A: Unlike traditional cytarabine which requires intravenous injection due to poor oral bioavailability, these structural analogs utilize a phosphoramidate prodrug strategy. This modification significantly enhances cell membrane permeability and protects the drug from rapid metabolic deamination, allowing for effective oral administration and prolonged half-life.
Q: What are the key advantages for large-scale manufacturing?
A: The synthesis route described in patent CN112409431B avoids the use of expensive transition metal catalysts and operates under relatively mild temperature conditions. The workup procedure involves standard extraction and column chromatography, which simplifies downstream processing and reduces the complexity of impurity removal compared to traditional nucleoside modifications.
Q: Is the supply chain for raw materials stable?
A: Yes, the key starting materials, including cytarabine and specific phosphoryl chloride derivatives, are commercially available from established chemical suppliers. The reliance on common organic solvents like tetrahydrofuran and dichloromethane further ensures that the supply chain remains robust and less susceptible to shortages of exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cytarabine Analog Supplier
As the demand for next-generation leukemia therapeutics grows, partnering with an experienced CDMO becomes critical for navigating the complexities of nucleoside chemistry. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of detecting trace impurities and verifying stereochemical purity, guaranteeing that every batch meets stringent purity specifications required by global regulatory agencies. We understand the sensitivity of phosphate-containing compounds and have optimized our drying and packaging processes to maintain stability throughout the logistics chain. Our technical team is ready to collaborate on process optimization to further enhance yields and reduce environmental impact, aligning with your corporate sustainability objectives.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our manufacturing efficiencies can lower your overall cost of goods sold. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target product profile. Whether you require custom synthesis of the phosphoryl chloride precursors or full-scale production of the final cytarabine analog, our integrated services are designed to accelerate your time to market. Let us help you overcome engineering bottlenecks and secure a reliable supply of high-quality oncology intermediates for your vital medications.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
