Advanced Cytarabine Prodrug Intermediates: Enhancing Bioavailability and Organ Specificity
Advanced Cytarabine Prodrug Intermediates: Enhancing Bioavailability and Organ Specificity
The global landscape of oncology treatment is constantly evolving, driven by the urgent need to overcome the pharmacokinetic limitations of established chemotherapeutic agents. A pivotal innovation in this domain is detailed in patent CN102115485A, which introduces a novel class of prodrugs based on the cytosine arabinoside (cytarabine) structure. This technology addresses the critical challenge of rapid metabolic inactivation that plagues traditional nucleoside therapies, particularly their susceptibility to deamination in the liver. By strategically modifying the N4 position with heterocyclic carboxylic acid fatty esters and optionally phosphorylating the O5 position, this invention creates compounds with enhanced lipophilicity, improved bioavailability, and distinct organ specificity. For pharmaceutical developers, this represents a significant leap forward in designing next-generation anticancer therapeutics that can effectively target resistant tumors in the liver and colon.
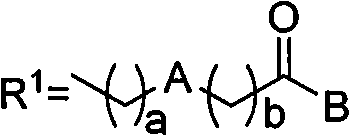
As illustrated in the general structure, the core innovation lies in the versatility of the substituent groups attached to the cytarabine backbone. The patent defines 'A' as a diverse array of aromatic or heterocyclic compounds, including pyridine, pyrazine, thiophene, and biphenyl derivatives, connected via alkyl spacers. This structural flexibility allows medicinal chemists to fine-tune the physicochemical properties of the drug, optimizing it for specific delivery pathways. Furthermore, the inclusion of long-chain alkoxy groups (C6-C18) dramatically increases the lipid solubility of the molecule, facilitating passive diffusion across cell membranes. This is a crucial improvement over native cytarabine, which relies on active transport mechanisms that are often downregulated in resistant cancer cell lines, thereby restoring sensitivity in difficult-to-treat malignancies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional cytarabine therapy is severely hampered by its poor pharmacokinetic profile, characterized by rapid clearance and extensive first-pass metabolism. Upon administration, native cytarabine is swiftly deaminated by cytidine deaminase, primarily in the liver, converting the active drug into the inactive metabolite uracil arabinoside (ara-U). This metabolic bottleneck results in a very short plasma half-life, typically ranging from 3 to 15 minutes, necessitating continuous intravenous infusions to maintain therapeutic levels. Moreover, the conversion of cytarabine to its active triphosphate form (Ara-CTP) inside the cell is a rate-limiting step dependent on deoxycytidine kinase, an enzyme that is frequently deficient in resistant leukemia and solid tumor cells. Consequently, conventional formulations often fail to achieve sufficient intracellular concentrations to induce apoptosis, leading to treatment failure and disease progression in patients with advanced hepatic or colorectal cancers.
The Novel Approach
The synthetic strategy outlined in the patent offers a transformative solution by chemically masking the vulnerable amine group and pre-activating the phosphate pathway. By acylating the N4 position with bulky heterocyclic esters, the prodrug sterically hinders the access of deaminase enzymes, effectively protecting the molecule from premature degradation during systemic circulation. This "Trojan Horse" approach allows the drug to reach the target tissue intact, where specific esterases or amidases can cleave the promoiety to release the active parent drug. Additionally, the optional phosphorylation at the 5'-hydroxyl position bypasses the initial, rate-limiting phosphorylation step catalyzed by deoxycytidine kinase. This dual-modification strategy not only extends the biological half-life but also ensures a higher flux of the active triphosphate metabolite within the tumor microenvironment, significantly enhancing antitumor efficacy against resistant cell lines.
Mechanistic Insights into N4-Acylation and O5-Phosphorylation
The chemical elegance of this synthesis lies in its use of robust coupling reagents that ensure high yields and purity under mild conditions. The N4-acylation step utilizes phosphonium-based coupling agents like PyBOP (Benzotriazol-1-yloxytripyrrolidinophosphonium hexafluorophosphate) in the presence of DMAP (4-Dimethylaminopyridine) as a nucleophilic catalyst. This combination activates the carboxylic acid moiety of the heterocyclic linker, facilitating a smooth nucleophilic attack by the exocyclic amine of cytarabine. The reaction proceeds efficiently at room temperature in polar aprotic solvents such as DMF or N-methylpyrrolidone, minimizing the risk of sugar ring degradation or epimerization which can occur under harsh acidic or basic conditions. This selectivity is paramount for maintaining the stereochemical integrity of the arabinose sugar, which is essential for the drug's recognition by DNA polymerase.
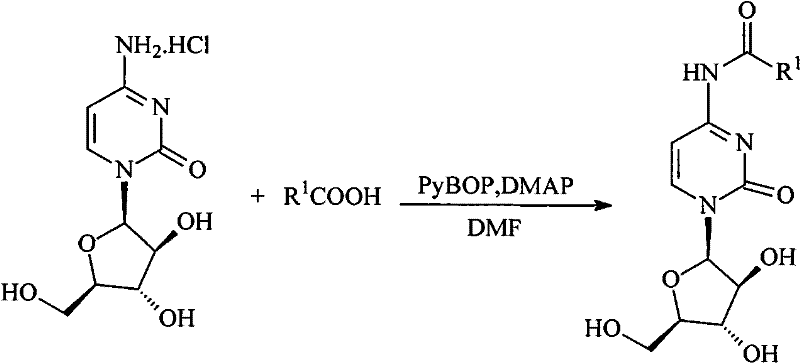
For the O5-phosphorylation, the mechanism involves the formation of a reactive phosphorodichloridate intermediate using phosphorus oxychloride (POCl3) and a proton scavenger like diisopropylethylamine (DIEPA). Conducted at low temperatures (0°C), this step ensures mono-phosphorylation at the primary 5'-hydroxyl group without affecting the secondary 2'- and 3'-hydroxyls. Subsequent alcoholysis with long-chain alcohols converts the dichloridate into a stable diester phosphate. This lipophilic phosphate mask further enhances membrane permeability and allows the drug to bypass the first kinase step entirely upon intracellular hydrolysis. The resulting prodrug exhibits a unique ability to accumulate in lipid-rich tissues, providing a targeted delivery mechanism that reduces systemic toxicity while maximizing local drug concentration in organs like the liver.
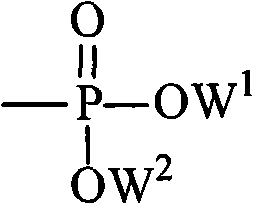
How to Synthesize N4-Modified Cytarabine Prodrugs Efficiently
The preparation of these high-value intermediates follows a streamlined, scalable protocol designed for industrial feasibility. The process begins with the preparation of the acylating agent, where heterocyclic anhydrides are reacted with long-chain alcohols to generate the requisite carboxylic acids. These acids are then coupled directly to cytarabine hydrochloride using the PyBOP/DMAP system, a method known for its reliability and ease of workup. For derivatives requiring phosphate masking, the crude N4-acylated product is subjected to phosphorylation conditions without the need for extensive purification in between, telescoping the process to save time and resources. Detailed standardized synthetic steps for specific derivatives can be found in the guide below.
- Prepare the acylating agent by reacting heterocyclic anhydrides or acyl chlorides with long-chain alcohols (C6-C18) to form the corresponding carboxylic acid intermediates.
- Perform N4-acylation of cytarabine hydrochloride using PyBOP and DMAP in DMF at room temperature for 12-24 hours to attach the lipophilic side chain.
- Optionally phosphorylate the 5'-hydroxyl group using phosphorus oxychloride (POCl3) and diisopropylethylamine at 0°C, followed by alcoholysis to introduce phosphate ester groups.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technology offers substantial benefits for procurement managers and supply chain directors looking to optimize the production of anticancer intermediates. The synthetic route relies on commodity chemicals and widely available coupling reagents, eliminating the dependency on scarce or exotic catalysts that often bottleneck production schedules. By simplifying the protection and deprotection sequences typically required for nucleoside modification, the process drastically reduces the number of unit operations, leading to significant cost reductions in API manufacturing. Furthermore, the high purity profiles reported in the patent examples (often exceeding 95% by LC-UV) minimize the need for complex downstream purification, directly translating to lower operational expenditures and faster batch release times.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of room temperature reactions significantly lowers energy consumption and waste disposal costs. The high atom economy of the PyBOP-mediated coupling ensures that raw material utilization is maximized, reducing the overall cost of goods sold (COGS). Additionally, the robustness of the chemistry allows for the use of technical grade solvents in early steps, further driving down input costs without compromising final product quality.
- Enhanced Supply Chain Reliability: The starting materials, such as pyridine dicarboxylic anhydrides and long-chain fatty alcohols, are produced on a multi-ton scale by the global chemical industry, ensuring a stable and continuous supply. This reduces the risk of raw material shortages that can disrupt production timelines. The synthetic pathway is also insensitive to minor fluctuations in moisture or temperature, making it highly reproducible across different manufacturing sites and equipment setups, which is critical for maintaining consistent supply to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic heavy metals or genotoxic reagents, simplifying the environmental health and safety (EHS) compliance burden. Workup procedures involve standard aqueous extractions and silica gel chromatography, techniques that are easily scaled from kilogram to multi-ton production without requiring specialized reactor configurations. This scalability ensures that the technology can meet the growing global demand for advanced cancer therapeutics while adhering to increasingly stringent green chemistry regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the stability, formulation, and regulatory aspects of these cytarabine prodrug intermediates. Understanding these nuances is essential for R&D teams planning to integrate this technology into their drug development pipelines. The answers provided are based on the experimental data and mechanistic insights disclosed in the patent literature, offering a reliable foundation for feasibility assessments.
Q: How does N4-modification improve the therapeutic index of cytarabine?
A: N4-modification with lipophilic heterocyclic carboxylic acid esters significantly enhances the solubility and membrane permeability of cytarabine. This structural change protects the amine group from rapid deamination by cytidine deaminase in the liver, thereby extending the half-life and allowing for better accumulation in target organs like the liver and colon.
Q: What are the critical reaction conditions for the phosphorylation step?
A: The phosphorylation step requires strict temperature control at 0°C using an ice bath to prevent side reactions. Phosphorus oxychloride (POCl3) is activated with diisopropylethylamine (DIEPA) in anhydrous organic solvents like THF, followed by the addition of long-chain alcohols to form the diester phosphate prodrug.
Q: Can this synthesis platform be applied to other nucleoside analogues?
A: Yes, the synthetic methodology described in patent CN102115485A is highly versatile. While optimized for cytarabine, the N4-acylation and O5-phosphorylation strategy is applicable to other nucleoside anticancer drugs such as gemcitabine, azacitidine, and decitabine, offering a broad platform for prodrug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cytarabine Prodrug Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of N4-modified cytarabine prodrugs in the fight against resistant cancers. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your innovative therapies can reach the market without supply constraints. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards for safety and efficacy. We are committed to supporting your R&D efforts with high-quality materials that accelerate your path to clinical success.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation oncology portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and route feasibility assessments, and let us help you optimize your supply chain for the future of cancer treatment.
