Advanced Synthetic Route for Upadacitinib Intermediates Enabling Scalable Commercial Production
The pharmaceutical landscape for Janus Kinase (JAK) inhibitors has evolved rapidly, with Upadacitinib emerging as a critical therapeutic agent for treating severe atopic dermatitis and rheumatoid arthritis. As detailed in the Chinese patent CN111217819A, a groundbreaking synthetic methodology has been developed that addresses the longstanding inefficiencies associated with the commercial production of this complex molecule. This innovation represents a significant leap forward in process chemistry, shifting away from traditional, costly pathways toward a more streamlined and economically viable manufacturing protocol. By re-engineering the construction of the key chiral pyrrolidine core and the pyrazolo-pyrazine fusion, this new approach offers a robust solution for meeting the escalating global demand for high-purity active pharmaceutical ingredients. For industry stakeholders, understanding the technical nuances of this patent is essential for securing a competitive edge in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Upadacitinib and its key intermediates has been plagued by reliance on expensive precious metal catalysts and cumbersome purification steps that hinder scalable production. Prior art, such as the methods disclosed in US2017129902, typically necessitates the use of palladium catalysts for multiple hydrogenation stages, which not only inflates raw material costs but also introduces significant challenges in removing trace metal residues to meet regulatory standards. Furthermore, conventional routes often depend on chiral resolution techniques that inherently cap the maximum theoretical yield at fifty percent, resulting in substantial material waste and increased environmental burden. The use of hazardous reagents like Lawesson's reagent for cyclization in older pathways further complicates the process, creating safety risks and requiring specialized waste treatment protocols that drive up operational expenditures. These cumulative inefficiencies create a fragile supply chain vulnerable to cost fluctuations and production delays.
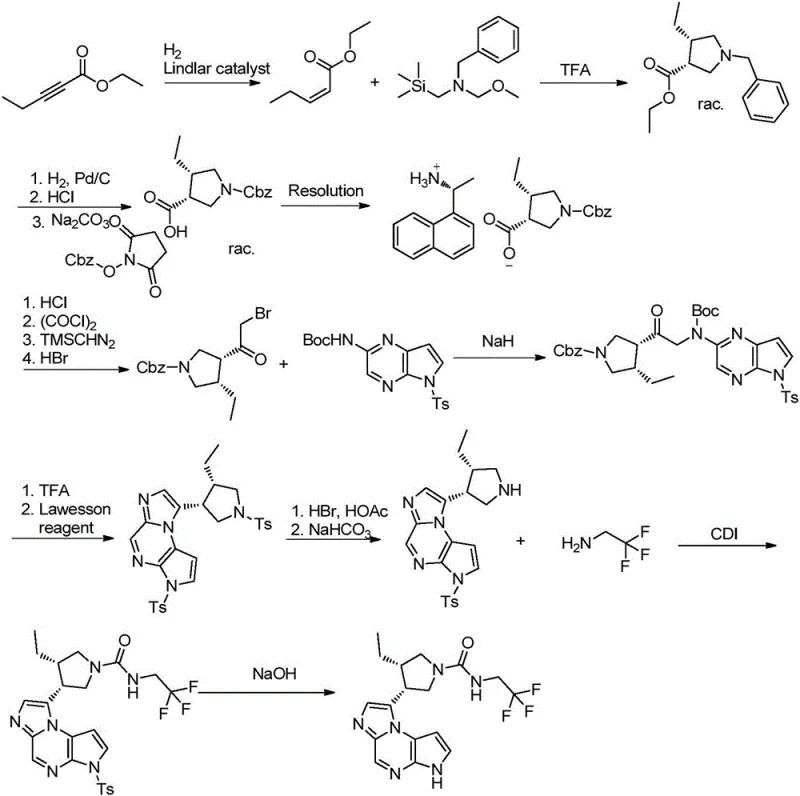
The Novel Approach
In stark contrast, the methodology outlined in CN111217819A introduces a paradigm shift by utilizing nickel-catalyzed coupling and advanced asymmetric hydrogenation to bypass these traditional bottlenecks. This innovative route strategically replaces palladium with nickel for the critical carbon-carbon bond formation, drastically reducing the dependency on high-value precious metals while maintaining high reaction efficiency. The process eliminates the need for low-yield chiral resolution by employing a stereoselective hydrogenation step that directly establishes the required (3S,4R) configuration with high fidelity. Additionally, the replacement of sulfur-based cyclization reagents with trifluoroacetic anhydride streamlines the ring-closing step, enhancing safety profiles and simplifying downstream processing. This holistic optimization results in a shorter synthetic sequence that is inherently more robust and adaptable to large-scale manufacturing requirements.
Mechanistic Insights into Nickel-Catalyzed Coupling and Chiral Hydrogenation
The core technical advancement of this patent lies in the precise orchestration of transition metal catalysis to construct the chiral pyrrolidine scaffold with exceptional stereocontrol. The process initiates with the activation of a pyrroline carboxylate via chlorination, followed by a nickel-catalyzed cross-coupling with an ethyl Grignard reagent. This specific transformation is critical as it installs the ethyl side chain under mild conditions, avoiding the harsh environments that often lead to racemization or decomposition in alternative methods. Subsequently, the introduction of chirality is achieved through a ruthenium-catalyzed asymmetric hydrogenation using a chiral ligand system such as S-Sunphos. This step is pivotal, as it converts the prochiral olefin into the desired chiral saturated ring with high enantiomeric excess, effectively setting the stereochemical foundation for the entire molecule without the need for subsequent resolution.
Following the establishment of the chiral center, the synthesis proceeds through a derivatization sequence involving Meldrum's acid to facilitate halogenation and subsequent coupling with the heterocyclic fragment. The final assembly involves a condensation reaction between the chiral acid fragment and the protected pyrazolo-pyrazine amine, followed by a cyclization mediated by trifluoroacetic anhydride. This cyclization step is particularly elegant, as it forms the fused tricyclic core essential for biological activity while simultaneously removing protecting groups under controlled conditions. The entire sequence is designed to minimize the formation of diastereomers and regioisomers, ensuring that the final crude product possesses a purity profile that significantly reduces the burden on final crystallization and purification stages.
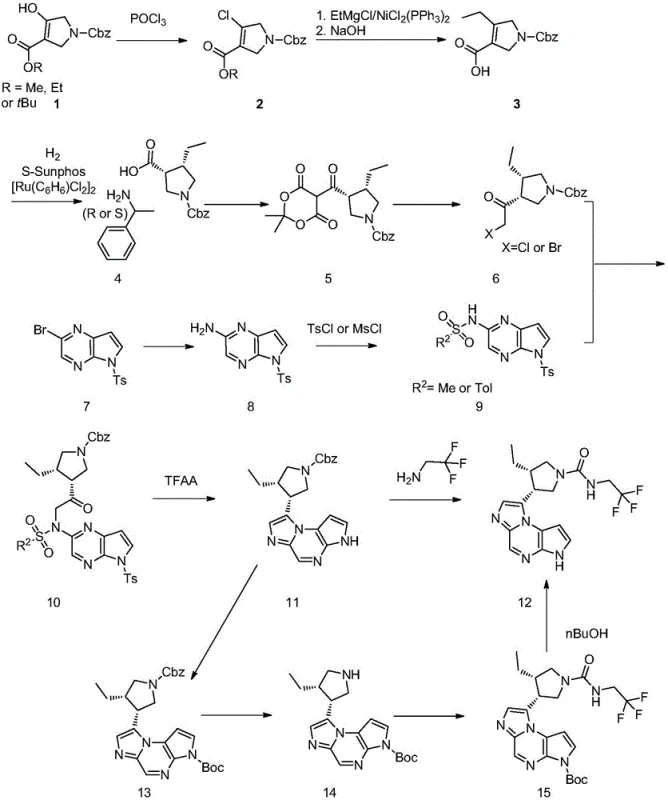
How to Synthesize Upadacitinib Efficiently
The implementation of this synthetic strategy requires careful attention to reaction parameters such as temperature control, stoichiometry, and catalyst loading to maximize yield and purity. The process is divided into distinct phases involving the preparation of the chiral acid intermediate, the synthesis of the heterocyclic amine fragment, and their final convergence. Operators must ensure strict anhydrous conditions during the Grignard coupling phase and maintain precise hydrogen pressure during the asymmetric reduction to guarantee consistent stereochemical outcomes. While the general workflow is outlined here, the specific operational details regarding solvent choices, workup procedures, and crystallization conditions are critical for successful technology transfer.
- Perform chlorination of the starting pyrroline carboxylate using phosphorus oxychloride to activate the position for coupling.
- Execute a nickel-catalyzed coupling reaction with an ethyl Grignard reagent followed by hydrolysis to introduce the ethyl group.
- Conduct asymmetric hydrogenation using a chiral ruthenium catalyst and resolve with chiral amine to establish stereochemistry.
- Derivatize with Meldrum's acid, halogenate, and couple with the protected pyrazolo-pyrazine fragment followed by cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthetic route offers profound advantages for procurement managers and supply chain directors seeking to optimize cost structures and mitigate supply risks. The fundamental redesign of the chemical pathway directly translates into tangible economic benefits by eliminating expensive reagents and reducing the total number of unit operations required to reach the final API. This efficiency gain is not merely theoretical but is rooted in the chemical mechanism itself, where the substitution of noble metals with base metals and the avoidance of yield-limiting resolution steps create a leaner manufacturing model. For organizations managing tight budgets and complex vendor relationships, this technology represents a strategic opportunity to secure a more stable and cost-effective source of critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the replacement of palladium catalysts with nickel-based systems, which are significantly less expensive and more abundant globally. By removing the need for multiple palladium hydrogenation steps and the associated scavenging processes required to meet residual metal specifications, the overall cost of goods sold is substantially decreased. Furthermore, the elimination of chiral resolution prevents the loss of half the material inventory, effectively doubling the yield potential of the chiral synthesis step and maximizing the return on raw material investment.
- Enhanced Supply Chain Reliability: Dependence on precious metals often exposes pharmaceutical supply chains to geopolitical volatility and price spikes, whereas nickel and standard organic reagents offer a more stable procurement landscape. The simplified reaction sequence reduces the number of intermediate handovers and quality control checkpoints, thereby shortening the overall production cycle time and improving responsiveness to market demand fluctuations. This robustness ensures a continuous supply of high-purity intermediates, minimizing the risk of stockouts that can disrupt downstream drug formulation and packaging schedules.
- Scalability and Environmental Compliance: The avoidance of malodorous and toxic sulfur-containing reagents like Lawesson's reagent significantly simplifies waste management and aligns the process with increasingly stringent environmental regulations. A cleaner reaction profile with fewer by-products facilitates easier scale-up from pilot plant to commercial tonnage without the need for extensive re-engineering of safety systems. This environmental compatibility not only reduces disposal costs but also enhances the sustainability profile of the manufacturing site, which is a growing priority for corporate social responsibility initiatives within the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Upadacitinib synthesis method. These insights are derived directly from the comparative data and experimental examples provided within the patent documentation, offering clarity on how this route outperforms legacy technologies. Understanding these specifics is vital for technical teams evaluating the feasibility of adopting this new process for their own manufacturing portfolios or for procurement teams negotiating supply agreements.
Q: How does this new synthetic route improve upon previous palladium-based methods?
A: The novel route replaces expensive palladium catalysts with more cost-effective nickel catalysts for the coupling step and eliminates the need for complex chiral resolution steps found in prior art, significantly lowering raw material costs and simplifying purification.
Q: What measures are taken to ensure high enantiomeric purity in the final product?
A: The process utilizes a highly selective chiral ruthenium catalyst for asymmetric hydrogenation followed by salt formation with chiral phenylethylamine, which effectively minimizes the generation of secondary chiral by-products and ensures stringent stereochemical control.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the route is designed for scalability by avoiding hazardous reagents like Lawesson's reagent and reducing the total number of reaction steps, which facilitates easier process amplification and consistent quality control in commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Upadacitinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthetic route and possess the technical expertise to bring it to fruition on a global scale. As a premier CDMO partner, we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing settings. Our facilities are equipped with rigorous QC labs and stringent purity specifications capable of handling complex chiral syntheses and sensitive catalytic reactions, guaranteeing that every batch meets the highest international standards for pharmaceutical intermediates.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-efficient technology for their Upadacitinib supply needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic advantages of switching to this nickel-catalyzed route for your project. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of JAK inhibitor therapeutics.
