Revolutionizing Aromatic Aldehyde Production: High-Efficiency Carbonylation for Global Pharmaceutical Intermediates
The landscape of fine chemical synthesis is constantly evolving, driven by the relentless demand for higher purity intermediates and more sustainable manufacturing processes. A significant breakthrough in this domain is documented in patent CN111072464A, which introduces a novel method for the direct introduction of aldehyde groups onto aromatic rings. This technology addresses the longstanding inefficiencies in producing key pharmaceutical and agrochemical building blocks, such as p-tolualdehyde, which serves as a critical precursor for terephthalic acid and various polymer additives. By leveraging a sophisticated catalyst system comprising a silicon-based carrier, a specialized ionic liquid, and transition metal auxiliaries, this method achieves unprecedented conversion rates and selectivity. For global procurement leaders and R&D directors, this patent represents a pivotal shift towards more atom-economical and cost-effective pathways for generating high-value aromatic aldehydes, positioning it as a cornerstone technology for modern fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic aldehydes has been plagued by significant technical and economic hurdles that impact supply chain stability and production costs. Traditional routes, such as the direct high-temperature oxidation of p-xylene, suffer from inherently low aromatic utilization rates, often yielding total conversion figures as low as 26.7 percent, which necessitates extensive recycling loops and energy-intensive separation processes. Alternatively, indirect electrosynthesis methods, while offering better selectivity, are constrained by high capital expenditure due to complex equipment requirements and the prohibitive cost of specialized catalysts. Furthermore, classical Gattermann-Koch carbonylation reactions, though atom-economical in theory, have historically struggled with low toluene conversion rates and poor selectivity when utilizing conventional liquid acid or solid superacid catalysts. These inefficiencies result in substantial waste generation and inconsistent product quality, creating bottlenecks for companies seeking a reliable aromatic aldehyde supplier capable of meeting stringent pharmaceutical grade specifications.
The Novel Approach
The innovative methodology described in the patent data overcomes these legacy challenges through the deployment of a supported ionic liquid phase (SILP) catalyst system enhanced with specific transition metal auxiliaries. Unlike traditional homogeneous systems that are difficult to separate, this approach immobilizes the active ionic liquid species onto a robust silicon-based support, such as silica gel, SBA-15, or MCM-41. The core of this catalytic power lies in the unique structure of the ionic liquid, which combines aluminum halides with imidazolium salts to create a highly active Lewis acidic environment. 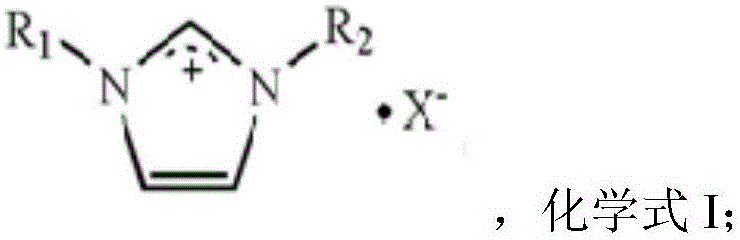 This structural configuration, further boosted by the synergistic presence of metals like Chromium, Vanadium, or Manganese, facilitates a highly efficient electrophilic substitution of carbon monoxide onto the aromatic ring. The result is a dramatic improvement in process metrics, with experimental data showing toluene conversion rates soaring to 90.3 percent and selectivity for the target para-isomer reaching 93.6 percent, effectively solving the dual problems of low yield and poor selectivity that have hindered the industry for decades.
This structural configuration, further boosted by the synergistic presence of metals like Chromium, Vanadium, or Manganese, facilitates a highly efficient electrophilic substitution of carbon monoxide onto the aromatic ring. The result is a dramatic improvement in process metrics, with experimental data showing toluene conversion rates soaring to 90.3 percent and selectivity for the target para-isomer reaching 93.6 percent, effectively solving the dual problems of low yield and poor selectivity that have hindered the industry for decades.
Mechanistic Insights into Supported Ionic Liquid Carbonylation
To fully appreciate the technical superiority of this method, one must delve into the mechanistic interplay between the ionic liquid matrix and the transition metal promoters. The reaction proceeds via an electrophilic aromatic substitution mechanism where the carbon monoxide molecule is activated by the strong Lewis acidity of the chloroaluminate anions within the ionic liquid. The imidazolium cation stabilizes the reactive intermediates, while the silicon-based support provides a high surface area that maximizes the exposure of active sites to the reactants. Crucially, the addition of compound auxiliary metals such as Cr, V, and Mn introduces a synergistic effect that further polarizes the CO bond, making the carbonyl carbon more susceptible to nucleophilic attack by the electron-rich aromatic ring. This multi-component catalytic system ensures that the reaction proceeds rapidly even at moderate temperatures ranging from 30 to 120 degrees Celsius, minimizing thermal degradation of sensitive functional groups and reducing the formation of unwanted by-products like benzoic acid or over-oxidized species.
From an impurity control perspective, this mechanism offers distinct advantages for the production of high-purity aromatic aldehydes. The heterogenized nature of the catalyst prevents the leaching of active species into the product stream, a common issue with homogeneous acid catalysts that complicates downstream neutralization and washing steps. Moreover, the high regioselectivity towards the para-position, dictated by the steric and electronic properties of the catalyst pore structure, significantly reduces the burden of isomer separation. In pharmaceutical synthesis, where isomeric impurities can be toxic or alter biological activity, this level of control is paramount. The ability to tune the catalyst by adjusting the molar ratio of the imidazolium salt to the aluminum halide, or by varying the loading of the transition metal auxiliaries, allows process chemists to optimize the impurity profile specifically for their target application, ensuring compliance with rigorous regulatory standards for API intermediates.
How to Synthesize P-Tolualdehyde Efficiently
Implementing this advanced carbonylation route requires precise adherence to the preparation and reaction protocols outlined in the patent to ensure optimal performance. The process begins with the meticulous preparation of the supported catalyst, involving the calcination of the silicon carrier followed by impregnation with the ionic liquid under an inert nitrogen atmosphere to prevent moisture deactivation. Once the catalyst is prepared and loaded into a high-pressure reactor along with the transition metal auxiliaries and a solvent like cyclohexane, the system is purged and pressurized with carbon monoxide. The detailed standardized synthesis steps, including specific stirring rates, gas replacement cycles, and workup procedures involving ice water washing and extraction, are critical for reproducibility and safety. For a comprehensive breakdown of the operational parameters required to achieve the reported 90 percent plus conversion rates, please refer to the technical guide below.
- Preparation of the supported catalyst by calcining a silicon-based carrier (such as silica gel or SBA-15) and impregnating it with an aluminum halide-based ionic liquid under inert atmosphere.
- Addition of compound auxiliary metals (Cr, V, or Mn) to the reactor containing the supported ionic liquid catalyst and a solvent like cyclohexane.
- Introduction of aromatic hydrocarbon substrate and CO gas into the high-pressure reactor, followed by heating to 30-120°C and maintaining pressure at 1-6 MPa to achieve carbonylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates directly into tangible strategic advantages regarding cost structure and supply reliability. The shift from inefficient oxidation processes or complex electrosynthesis to this streamlined carbonylation method eliminates several cost centers associated with traditional manufacturing. By utilizing a catalyst system that operates at moderate pressures and temperatures with high turnover numbers, the process significantly reduces energy consumption and reactor occupancy time. Furthermore, the use of readily available raw materials such as toluene and carbon monoxide, coupled with the robustness of the silicon-supported catalyst, mitigates the risks associated with volatile raw material markets. This stability is essential for maintaining consistent pricing and avoiding supply disruptions in the competitive landscape of fine chemical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic improvement in atom economy and the simplification of downstream processing. Because the catalyst achieves such high conversion and selectivity, the volume of unreacted starting material that needs to be recovered and recycled is minimized, leading to substantial savings in utility costs and solvent usage. Additionally, the heterogeneous nature of the supported ionic liquid catalyst simplifies the separation of the catalyst from the product mixture, removing the need for expensive and time-consuming neutralization steps typically required for liquid acid catalysts. This streamlined workflow reduces the overall operational expenditure per kilogram of product, allowing for more competitive pricing structures in the global market without compromising on margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of synthesis routes that rely on scarce reagents or fragile catalytic systems. This technology enhances reliability by employing a catalyst formulation based on abundant silicon materials and common transition metals like chromium and manganese, which are less susceptible to geopolitical supply shocks compared to precious metals. The robustness of the catalyst also implies a longer operational lifespan and the potential for multiple reuse cycles, reducing the frequency of catalyst replenishment orders. For supply chain heads, this means a more predictable production schedule and the ability to scale output rapidly to meet surging demand for derivatives like terephthalic acid precursors, ensuring that downstream customers never face stockouts.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to scale a process while minimizing waste is a critical commercial asset. This carbonylation method generates significantly less hazardous waste compared to traditional oxidation routes that produce heavy metal sludge or large volumes of acidic wastewater. The closed-loop nature of the high-pressure carbonylation reaction contains volatile organic compounds, and the ease of catalyst filtration reduces the load on wastewater treatment facilities. These factors facilitate smoother regulatory approvals for commercial scale-up and align with the sustainability goals of major multinational corporations, making the resulting aromatic aldehydes more attractive to environmentally conscious buyers in the pharmaceutical and polymer industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid-catalyzed carbonylation technology. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, aiming to clarify the operational feasibility and strategic value of adopting this method for large-scale production. Understanding these nuances is vital for technical teams evaluating the transition from legacy processes to this next-generation synthetic route.
Q: What represents the primary advantage of this ionic liquid catalyst system over traditional Gattermann-Koch methods?
A: The primary advantage lies in the significantly enhanced conversion rates and selectivity. While traditional methods often struggle with low aromatic utilization and complex separation, this supported ionic liquid system achieves toluene conversion rates exceeding 90% with para-selectivity above 93%, drastically reducing downstream purification costs.
Q: Can this carbonylation process be scaled for industrial production of terephthalic acid precursors?
A: Yes, the process is highly amenable to scale-up. The use of a solid silicon-based carrier (like silica gel or MCM-41) transforms the typically homogeneous ionic liquid into a heterogeneous or semi-heterogeneous system, facilitating catalyst recovery and reuse, which is critical for continuous industrial manufacturing of intermediates like p-tolualdehyde.
Q: What role do the transition metal auxiliaries play in the reaction mechanism?
A: The transition metal auxiliaries, specifically Chromium (Cr), Vanadium (V), and Manganese (Mn), act as synergistic promoters. They enhance the electrophilic substitution capability of the CO molecule on the aromatic ring, working in concert with the Lewis acidic chloroaluminate ionic liquid to lower the activation energy and improve overall reaction kinetics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable P-Tolualdehyde Supplier
The technological advancements detailed in patent CN111072464A underscore the immense potential for optimizing the production of aromatic aldehydes, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this transformation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative chemistries to the market. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced high-pressure reactors capable of handling the specific conditions required for ionic liquid catalysis, ensuring that every batch meets stringent purity specifications demanded by the global pharmaceutical sector. We understand that the transition to a new catalytic system involves careful validation, and our team is dedicated to managing every aspect of the scale-up process to guarantee consistency and quality.
We invite forward-thinking organizations to collaborate with us to unlock the full commercial potential of this high-efficiency synthesis route. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a product, but to a comprehensive solution that includes a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive down your total cost of ownership while securing a stable supply of critical intermediates for your supply chain.
