Optimizing Epoxiconazole Intermediate Production via Novel Wittig-Horner Methodology
The global demand for high-performance fungicides such as Epoxiconazole necessitates robust and efficient supply chains for critical intermediates. Patent CN102643162A introduces a transformative methodology for the preparation of 1-(2-chlorophenyl)-2-(4-fluorophenyl) propylene, a pivotal building block in agrochemical synthesis. This technical disclosure addresses long-standing inefficiencies in traditional manufacturing by leveraging a modified Wittig-Horner reaction sequence that prioritizes safety, yield, and environmental sustainability. By shifting away from hazardous Grignard reagents and expensive phosphine ligands, this route offers a compelling value proposition for industrial scale-up. The process utilizes readily available starting materials like o-chlorobenzyl chloride and trialkyl phosphite, ensuring supply chain stability. Furthermore, the operational simplicity and mild reaction conditions described in the patent significantly lower the barrier for commercial adoption, making it an ideal candidate for integration into existing fine chemical production facilities seeking to optimize their agrochemical intermediate portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key agrochemical intermediates has relied heavily on Grignard chemistry, a pathway fraught with significant operational hazards and economic inefficiencies. The traditional route, as referenced in prior art such as US Patent No. 50812317A, involves the use of Grignard reagents which are highly reactive and moisture-sensitive, requiring rigorous anhydrous conditions that drive up energy and infrastructure costs. A critical safety concern is the evolution of large volumes of hydrogen gas during the reaction, necessitating specialized explosion-proof equipment and complex ventilation systems to mitigate fire risks. Moreover, the reliance on triphenylphosphine in alternative pathways generates stoichiometric amounts of triphenylphosphine oxide, a solid waste byproduct that is notoriously difficult to separate and dispose of, creating environmental burdens. These factors collectively result in higher production costs, extended cycle times, and increased regulatory scrutiny, rendering conventional methods less attractive for modern, sustainability-focused manufacturing environments.
The Novel Approach
The methodology outlined in CN102643162A represents a paradigm shift by employing a Wittig-Horner reaction mediated by trialkyl phosphites rather than traditional phosphines. This innovative approach eliminates the generation of hydrogen gas, thereby removing a major safety bottleneck and allowing for operation in standard chemical reactors without specialized explosion-proof modifications. The substitution of triphenylphosphine with trialkyl phosphite not only reduces raw material costs but also simplifies the downstream purification process, as the phosphonate byproducts are more manageable. The process operates under mild alkaline conditions with reaction temperatures ranging from 0°C to 50°C, which significantly reduces energy consumption compared to high-temperature alternatives. Additionally, the use of polar solvents like DMF or DMSO as catalysts enhances reaction kinetics, shortening cycle times and improving equipment throughput. This combination of safety, cost-efficiency, and operational ease makes the novel approach superior for large-scale industrial application.
Mechanistic Insights into Phosphonate-Mediated Wittig-Horner Olefination
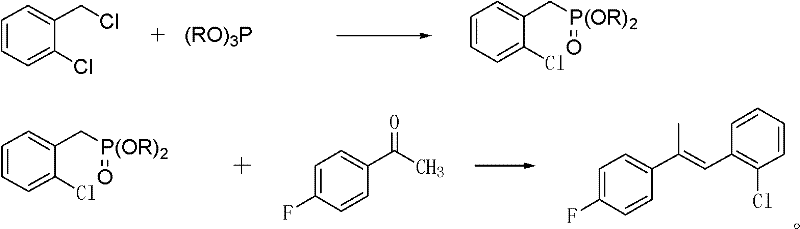
The core of this synthesis lies in the generation of a stabilized carbanion from the o-chlorobenzyl phosphonate diester, which serves as the nucleophile in the olefination step. In the first stage, an Arbuzov reaction occurs where o-chlorobenzyl chloride reacts with a trialkyl phosphite under reflux conditions at 170°C to 180°C to form the phosphonate diester. This intermediate is crucial as the electron-withdrawing phosphoryl group stabilizes the adjacent carbanion formed upon deprotonation by a strong base such as potassium hydroxide or sodium methoxide. Unlike unstabilized ylides used in classic Wittig reactions, this stabilized anion reacts with the ketone, specifically 4-fluoroacetophenone, to form the alkene with high E-selectivity. The presence of polar aprotic solvents like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) acts as a phase transfer catalyst or solvation enhancer, facilitating the interaction between the ionic base and the organic substrates. This mechanistic nuance ensures high conversion rates and minimizes side reactions, leading to the observed high yields.
Impurity control is inherently built into the reaction design through the choice of reagents and conditions. The use of trialkyl phosphite instead of triphenylphosphine avoids the formation of triphenylphosphine oxide, a common impurity that often co-crystallizes with products and requires chromatographic purification. The mild reaction temperature of 20°C to 30°C in the second step prevents thermal degradation of the sensitive fluoro-substituted aromatic rings, preserving the structural integrity of the molecule. Furthermore, the reaction endpoint is easily monitored via gas chromatography, ensuring that the consumption of 4-fluoroacetophenone is complete before workup, which minimizes the presence of unreacted starting materials in the final crude. The aqueous workup effectively removes inorganic salts and polar byproducts, while the organic layer retains the target propylene derivative. This streamlined purification profile results in a product content exceeding 94%, reducing the need for extensive recrystallization and lowering solvent waste.
How to Synthesize 1-(2-Chlorophenyl)-2-(4-Fluorophenyl) Propylene Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and reaction parameters to maximize yield and safety. The process begins with the preparation of the phosphonate diester, followed by the olefination step under strictly controlled basic conditions. Operators must ensure the complete removal of excess phosphite via distillation before proceeding to the second step to prevent interference with the base catalyst. The selection of the base, whether alkali metal hydroxides or alkoxides, should be matched with the appropriate polar solvent to optimize solubility and reaction rate. Detailed standard operating procedures regarding temperature ramping and quenching protocols are essential to maintain consistency across batches. For a comprehensive breakdown of the specific molar ratios, solvent volumes, and workup techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Preparation of o-chlorobenzyl phosphonate diester via refluxing o-chlorobenzyl chloride with trialkyl phosphite.
- Reaction of the phosphonate diester with p-fluoroacetophenone using polar solvents as catalysts under strong alkaline conditions.
- Workup involving water washing and solvent removal to isolate the high-purity propylene derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic advantages by addressing key cost drivers and risk factors associated with agrochemical intermediate manufacturing. The elimination of hazardous gas evolution reduces the need for specialized safety infrastructure, leading to significant capital expenditure savings for production facilities. The substitution of expensive reagents with cost-effective alternatives directly lowers the bill of materials, enhancing margin potential for downstream fungicide producers. Furthermore, the simplified waste profile reduces disposal costs and environmental compliance burdens, aligning with global sustainability goals. The robustness of the process ensures consistent supply continuity, mitigating the risk of production delays caused by safety incidents or complex purification bottlenecks. These factors collectively contribute to a more resilient and cost-efficient supply chain for high-value agrochemical intermediates.
- Cost Reduction in Manufacturing: The replacement of triphenylphosphine with trialkyl phosphite represents a direct material cost saving, as phosphites are generally more economical and atom-efficient. By avoiding the formation of solid phosphine oxide waste, the process eliminates the need for expensive waste treatment and disposal services, further reducing operational expenditures. The mild reaction conditions also translate to lower energy consumption for heating and cooling, contributing to overall utility cost reductions. Additionally, the high yield minimizes raw material waste, ensuring that a greater proportion of input materials are converted into saleable product. These cumulative efficiencies result in a significantly lower cost of goods sold without compromising on product quality or purity specifications.
- Enhanced Supply Chain Reliability: The safety profile of this method, which avoids hydrogen gas generation, reduces the likelihood of unplanned shutdowns due to safety incidents or regulatory inspections. The use of commercially available and stable starting materials ensures that supply is not dependent on niche or volatile reagent markets. The simplified workup procedure shortens the production cycle time, allowing for faster turnaround and increased production capacity to meet fluctuating market demands. This reliability is crucial for maintaining just-in-time inventory levels and ensuring uninterrupted supply to downstream formulation plants. Consequently, partners can expect a more stable and predictable supply of critical intermediates, reducing the need for excessive safety stock.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing standard reactor types and common solvents that are easily sourced in bulk quantities. The reduction in hazardous waste generation simplifies environmental permitting and compliance reporting, facilitating smoother operations in regions with strict environmental regulations. The ability to recover and recycle excess phosphite further enhances the green chemistry profile of the synthesis, appealing to environmentally conscious stakeholders. This scalability ensures that production can be ramped up from pilot scale to multi-ton commercial production without significant process re-engineering. As a result, the method supports sustainable growth and long-term viability in the competitive agrochemical market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation. They provide clarity on safety, cost, and quality aspects that are critical for decision-makers evaluating this technology for adoption. Understanding these details helps in assessing the feasibility of integrating this method into existing production lines or new facility designs. For further technical specifications or custom feasibility studies, direct consultation with our engineering team is recommended.
Q: What are the safety advantages of this method over traditional Grignard routes?
A: Unlike traditional methods utilizing Grignard reagents which release large volumes of hydrogen gas and require stringent safety protocols, this Wittig-Horner approach operates under mild conditions without hazardous gas evolution, significantly enhancing workshop safety.
Q: How does the use of trialkyl phosphite impact production costs?
A: Replacing expensive triphenylphosphine with trialkyl phosphite reduces raw material costs substantially. Additionally, the byproduct is easier to handle and recover, contributing to overall process economics and environmental compliance.
Q: What purity levels can be achieved with this synthesis route?
A: The patented method consistently achieves product yields and content exceeding 94%, ensuring high-quality intermediates suitable for the rigorous standards required in fungicide manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-(2-Chlorophenyl)-2-(4-Fluorophenyl) Propylene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of agrochemical formulations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-(2-chlorophenyl)-2-(4-fluorophenyl) propylene meets the exacting standards required for Epoxiconazole synthesis. Our commitment to technical excellence allows us to optimize the Wittig-Horner process described in CN102643162A, delivering consistent quality and reliability to our global partners. We are dedicated to supporting your R&D and production goals with a supply chain that is both robust and responsive.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your current manufacturing setup, highlighting potential efficiencies and ROI. We encourage you to reach out to request specific COA data and route feasibility assessments to validate the compatibility of this intermediate with your downstream processes. By partnering with us, you gain access to not just a product, but a comprehensive technical solution that drives value and competitiveness in the agrochemical market. Let us help you optimize your supply chain with our proven expertise and commitment to quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
