Advanced Immobilized Enzyme Technology for Commercial (S)-Nornicotine Production
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift in the production of chiral alkaloids, driven by the urgent need for higher purity and sustainable manufacturing processes. Patent CN115786418A introduces a groundbreaking method for synthesizing high-purity (S)-nornicotine utilizing a sophisticated immobilized enzyme catalytic system. This technology addresses the critical limitations of traditional plant extraction, which often suffers from batch-to-batch variability and the presence of potentially carcinogenic impurities inherent to tobacco sources. Furthermore, it outperforms conventional chemical resolution methods that typically involve complex separation steps, harsh reaction conditions, and significant material loss. By leveraging a dynamic kinetic resolution strategy, this biocatalytic route converts racemic (R,S)-nornicotine directly into the desired (S)-enantiomer with exceptional efficiency. For R&D Directors and Supply Chain Heads, this represents a viable pathway to secure a stable, high-quality supply of this critical intermediate, which is essential for the rapidly expanding electronic cigarette market and various pharmaceutical applications including smoking cessation therapies and neurological treatments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of (S)-nicotine and its precursor (S)-nornicotine has relied heavily on extraction from tobacco plants, a process fraught with significant supply chain vulnerabilities and quality control challenges. The extraction yield is heavily dependent on agricultural factors such as climate, harvest period, and soil conditions, leading to inconsistent availability and fluctuating market prices that disrupt long-term procurement planning. Moreover, naturally extracted alkaloids often contain a complex matrix of impurities, including nitrosamines and other potentially carcinogenic compounds, which necessitate rigorous and costly purification steps to meet safety standards for human consumption. On the synthetic front, traditional chemical methods often produce racemic mixtures that require chiral resolution, a process that inherently caps the maximum theoretical yield at 50% unless a dynamic kinetic resolution is employed, which is chemically difficult to achieve. These chemical routes frequently utilize expensive chiral auxiliaries or transition metal catalysts that require removal, adding to the environmental burden and production costs while complicating the regulatory approval process for pharmaceutical grades.
The Novel Approach
The innovative approach disclosed in patent CN115786418A circumvents these historical bottlenecks by employing a multi-enzyme immobilized system that functions as a self-regenerating catalytic engine. Instead of discarding the unwanted (R)-enantiomer, the process utilizes a high (R)-selectivity monoamine oxidase to oxidize the (R)-nornicotine into myosmine, which is subsequently reduced back to racemic nornicotine by an imine reductase. This cyclic mechanism effectively funnels the entire racemic starting material into the desired (S)-nornicotine product, theoretically allowing for yields approaching 100% rather than the 50% limit of standard resolution. The immobilization of these enzymes on epoxy resin carriers not only enhances their stability under operational conditions but also facilitates easy separation from the reaction mixture, enabling the catalyst to be recovered and reused for multiple batches. This technological leap transforms the production landscape by offering a route that is both economically superior due to higher atom economy and environmentally friendlier due to the elimination of heavy metal catalysts and harsh organic solvents.
Mechanistic Insights into Immobilized Enzyme Dynamic Kinetic Resolution
The core of this technological advancement lies in the precise orchestration of four distinct enzymatic activities co-immobilized on a single solid support, creating a highly efficient cascade reaction system. The process initiates with the high (R)-selectivity monoamine oxidase, which specifically recognizes and oxidizes the (R)-enantiomer of the nornicotine substrate to form myosmine, leaving the (S)-enantiomer untouched in the reaction medium. This myosmine intermediate is then immediately subjected to asymmetric reduction by the imine reductase, which, in the presence of a cofactor regeneration system involving glucose dehydrogenase and glucose, converts the myosmine back into a racemic mixture of nornicotine. As the cycle repeats, the (R)-enantiomer is continuously consumed and recycled, while the (S)-enantiomer accumulates in the solution, driving the equilibrium towards the exclusive formation of the target chiral product. The inclusion of catalase in the immobilized matrix is crucial for decomposing hydrogen peroxide, a byproduct of the oxidase reaction, thereby preventing enzyme inactivation and ensuring the longevity of the biocatalyst throughout the extended reaction period.
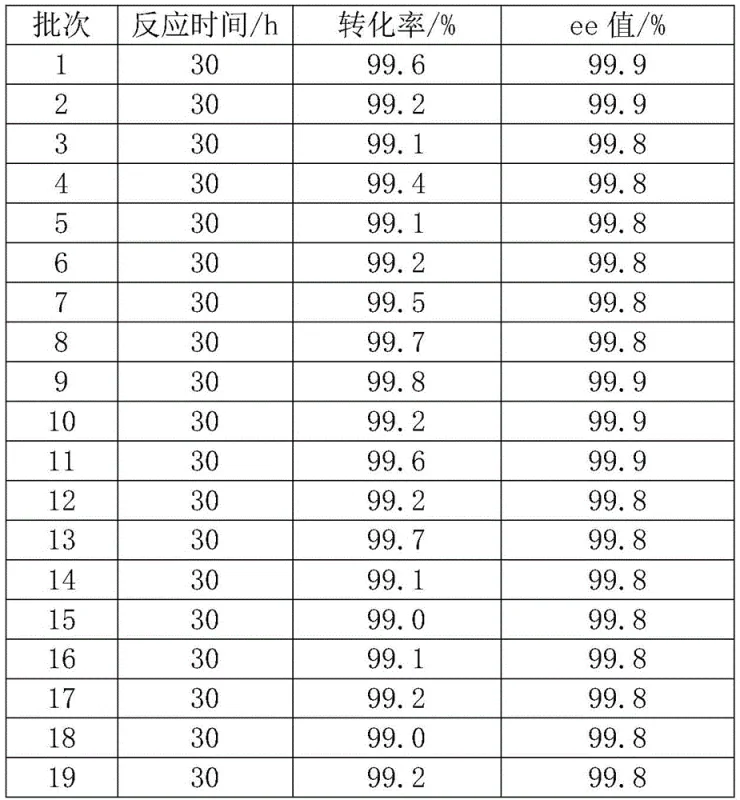
Controlling the impurity profile in such a complex biocatalytic system is paramount for meeting the stringent requirements of the pharmaceutical and e-liquid industries. The high selectivity of the engineered enzymes ensures that side reactions are minimized, resulting in a product with an optical purity (ee value) exceeding 99.8%, as demonstrated in the patent examples. The immobilization matrix itself acts as a barrier against contamination, and the mild aqueous reaction conditions prevent the formation of thermal degradation products often seen in high-temperature chemical synthesis. Furthermore, the ability to wash and reuse the immobilized enzyme for over 20 batches without significant loss of activity or selectivity indicates a robust process control mechanism that maintains consistent product quality over time. For quality assurance teams, this means a predictable impurity spectrum that is far easier to manage and validate compared to the variable impurity profiles associated with botanical extracts or multi-step chemical syntheses involving diverse reagents.
How to Synthesize (S)-Nornicotine Efficiently
Implementing this synthesis route requires careful attention to the preparation of the biocatalyst and the optimization of reaction parameters to maximize throughput and catalyst lifespan. The patent outlines a specific protocol for co-immobilizing the enzyme cocktail onto epoxy resin, which serves as the foundation for a scalable and reproducible manufacturing process. Operators must ensure that the ratio of the four enzymes is balanced to prevent the accumulation of intermediates that could inhibit the reaction, and the cofactor regeneration system must be sufficiently supplied to sustain the reductase activity. While the specific laboratory-scale parameters are detailed in the intellectual property, the general workflow involves preparing the substrate solution, introducing the immobilized biocatalyst, and maintaining the system under controlled pH and temperature conditions for a defined period.
- Preparation of the multi-enzyme immobilized system by covalently bonding monoamine oxidase, imine reductase, glucose dehydrogenase, and catalase onto epoxy resin carriers.
- Introduction of racemic (R,S)-nornicotine substrate into the reaction system containing the immobilized enzyme complex and glucose as a cofactor regenerator.
- Maintenance of reaction conditions at 15-40°C and pH 6.8-7.2 for 16-48 hours to allow dynamic kinetic resolution and accumulation of (S)-nornicotine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized enzyme technology offers substantial strategic advantages that extend beyond simple unit cost calculations. The primary value driver is the drastic simplification of the downstream processing workflow, as the solid nature of the biocatalyst allows for simple filtration to separate the product from the reaction mixture, eliminating the need for complex extraction and purification units. This reduction in processing steps translates directly into lower capital expenditure for manufacturing facilities and reduced operational costs related to solvent consumption and waste disposal. Additionally, the high atom economy of the dynamic kinetic resolution means that the raw material utilization is maximized, reducing the overall demand for starting materials and mitigating the risk of supply shortages for key precursors. The ability to operate under mild conditions also lowers energy consumption requirements, contributing to a more sustainable and cost-effective production model that aligns with modern corporate environmental goals.
- Cost Reduction in Manufacturing: The elimination of expensive chiral resolving agents and transition metal catalysts significantly lowers the direct material costs associated with production. By avoiding the use of heavy metals, the manufacturer also bypasses the costly and time-consuming steps required for metal scavenging and residual metal testing, which are mandatory for pharmaceutical compliance. The reusability of the immobilized enzyme over multiple batches further amortizes the cost of the biocatalyst, making the cost per kilogram of the final product highly competitive compared to traditional methods. This economic efficiency allows for more flexible pricing strategies and improved margin protection in a volatile market environment.
- Enhanced Supply Chain Reliability: Unlike agricultural extraction which is subject to seasonal fluctuations and geopolitical risks associated with tobacco farming, this synthetic biocatalytic route relies on fermentation-derived enzymes and chemically synthesized starting materials that can be sourced consistently year-round. The robustness of the immobilized enzyme system ensures that production schedules can be maintained without unexpected downtime due to catalyst failure or batch inconsistencies. This reliability is critical for maintaining continuous supply to downstream customers in the e-cigarette and pharmaceutical sectors, where interruptions can lead to significant contractual penalties and loss of market share. The scalability of the process from laboratory to industrial fermenters ensures that supply can be ramped up quickly to meet surging demand without compromising quality.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system and the absence of hazardous organic solvents simplify the waste treatment process, reducing the environmental footprint of the manufacturing site. This aligns with increasingly strict global environmental regulations and reduces the liability and costs associated with hazardous waste disposal. The mild reaction conditions also enhance workplace safety by reducing the risk of thermal runaways or exposure to toxic reagents, fostering a safer operational environment. Furthermore, the high efficiency of the process minimizes the generation of byproduct waste, supporting a circular economy approach to chemical manufacturing that is increasingly valued by stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route for (S)-nornicotine. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or for qualifying new suppliers who utilize this advanced method.
Q: How does the immobilized enzyme method improve purity compared to extraction?
A: Unlike plant extraction which carries carcinogenic impurities, this enzymatic route starts from synthetic racemic nornicotine and uses high-selectivity enzymes to specifically consume the (R)-enantiomer, yielding (S)-nornicotine with over 99.8% ee value without natural contaminants.
Q: Is the biocatalyst reusable for industrial scale-up?
A: Yes, the patent data demonstrates that the immobilized enzyme system maintains high conversion rates and optical purity over at least 20 repeated batches, significantly reducing catalyst consumption costs for large-scale manufacturing.
Q: What are the reaction conditions for this synthesis?
A: The process operates under mild conditions, specifically between 15-40°C and a neutral pH range of 6.8 to 7.2, which simplifies equipment requirements and enhances safety compared to harsh chemical resolution methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-Nornicotine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalytic technologies like the one described in patent CN115786418A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the high optical purity and low impurity profiles required for pharmaceutical and consumer health applications. We are committed to leveraging such advanced enzymatic routes to provide our clients with a competitive edge through superior product quality and supply chain stability.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain for (S)-nornicotine and related alkaloids. By partnering with us, you gain access to a secure, scalable, and technologically advanced source of critical chemical intermediates that can drive the success of your final products in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
