Advanced Biocatalytic Flow Synthesis for High-Purity Antibiotic Intermediates
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable manufacturing, particularly in the synthesis of critical antibiotic intermediates. Patent CN112280773B introduces a groundbreaking bio-enzyme catalysis flow process for preparing 2-amino-3-substituted phenyl-3-hydroxypropionic acid, a key precursor for drugs like chloramphenicol and florfenicol. This technology leverages immobilized aldolase within a continuous flow reactor system, fundamentally altering the economic and environmental landscape of amino acid derivative production. By integrating genetic engineering with process intensification, the method achieves conversion rates exceeding 96% while utilizing water as the primary solvent. For R&D directors and procurement strategists, this represents a significant opportunity to transition away from hazardous chemical synthesis towards a greener, more efficient biological platform that aligns with modern regulatory standards and corporate sustainability goals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of 2-amino-3-substituted phenyl-3-hydroxypropionic acid has relied on traditional organic reactions that are increasingly viewed as unsustainable in a modern regulatory environment. These conventional pathways frequently necessitate the use of highly toxic reagents such as hydrogen sulfide and various heavy metal catalysts, which pose severe safety risks to personnel and require complex, costly waste treatment infrastructure. The energy consumption associated with these batch processes is typically high due to stringent temperature controls and extensive downstream purification steps needed to remove metal residues to acceptable pharmaceutical limits. Furthermore, the atomic economy of these chemical routes is often poor, resulting in substantial material loss and the generation of large volumes of hazardous waste that do not comply with emerging green chemistry policies. The reliance on organic solvents further exacerbates the environmental footprint, creating supply chain vulnerabilities related to solvent availability and disposal costs.
The Novel Approach
In stark contrast, the novel biocatalytic flow process described in the patent data offers a transformative solution by replacing hazardous chemical reagents with a highly specific immobilized enzyme system. This approach utilizes water as a benign solvent, effectively eliminating the need for volatile organic compounds and significantly reducing the fire and health hazards associated with traditional manufacturing. The integration of a fixed-bed reactor allows for continuous processing, which dramatically improves space-time yield and reduces the physical footprint required for production compared to large batch tanks. By employing a genetically modified aldolase, the process achieves high selectivity and conversion under mild conditions, typically between 35-65°C, which lowers energy demands for heating and cooling. The design also incorporates a closed-loop recycling system for both the solvent and unreacted starting materials, ensuring maximum resource utilization and minimizing waste discharge to near-zero levels.
Mechanistic Insights into Immobilized Aldolase Catalysis
The core of this technological advancement lies in the sophisticated application of protein engineering to enhance enzyme stability and activity under flow conditions. The patent specifies the use of a genetically modified aldolase from Brevibacterium crescentum, designated as Cc-H91A178, where specific amino acid residues at positions 91 and 178 have been mutated to optimize performance. This engineered biocatalyst is immobilized on macroporous adsorption resins, such as D301 or ZGA451, which provides a robust solid support that withstands the hydraulic pressure and shear forces of a continuous flow system. The reaction mechanism involves the condensation of substituted benzaldehyde with glycine, facilitated by the enzyme in the presence of divalent metal ion promoters like zinc or nickel. This catalytic cycle is highly efficient, driving the equilibrium towards the desired product while minimizing the formation of by-products that typically complicate downstream purification in chemical synthesis.
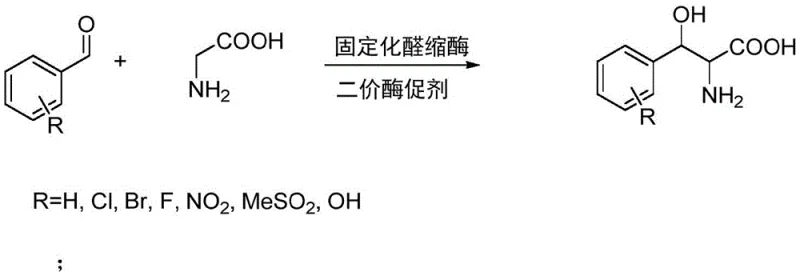
Impurity control is inherently superior in this enzymatic route due to the high stereoselectivity and chemoselectivity of the biocatalyst. Unlike chemical catalysts which may promote side reactions such as over-oxidation or non-specific substitutions, the enzyme active site ensures that only the specific aldol condensation occurs. The subsequent separation step utilizes a complexing resin fixed bed which specifically adsorbs the divalent metal ion promoters, ensuring that the final product is free from heavy metal contamination without requiring additional extraction or chromatography steps. This integrated purification strategy simplifies the workflow and ensures that the impurity profile of the final API intermediate meets stringent pharmacopeial standards. The ability to recycle the dilute glycine solution further enhances the purity of the overall process by preventing the accumulation of degradation products that can occur in batch recycling systems.
How to Synthesize 2-Amino-3-Substituted Phenyl-3-Hydroxypropionic Acid Efficiently
Implementing this synthesis route requires a precise understanding of the flow dynamics and enzyme immobilization parameters to ensure consistent quality and yield. The process begins with the preparation of the immobilized biocatalyst, followed by the continuous pumping of substrate solutions through the reactor bed at controlled flow rates ranging from 100 to 800 BV/h. Detailed operational protocols regarding buffer preparation, pH adjustment, and temperature gradients are critical for maintaining enzyme longevity and reaction efficiency over extended production runs. The standardized synthesis steps outlined below provide a framework for scaling this technology from laboratory validation to commercial manufacturing, ensuring that all critical process parameters are monitored and controlled.
- Immobilize genetically modified aldolase (Cc-H91A178) on macroporous adsorption resin in phosphate buffer.
- Pass a solution of glycine, substituted benzaldehyde, and divalent metal ions through the enzyme fixed bed at 35-65°C.
- Separate products using a complexing resin fixed bed to remove metal ions and recycle glycine solution.
- Concentrate, crystallize, and dry the product while recycling water and unreacted materials.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this biocatalytic flow process offers compelling economic and operational benefits that extend beyond simple unit cost calculations. The elimination of toxic reagents and heavy metals fundamentally alters the cost structure by removing the need for expensive waste disposal services and specialized containment equipment. The continuous nature of the flow reaction significantly enhances production throughput, allowing for a more responsive supply chain that can adapt quickly to fluctuations in market demand for antibiotic intermediates. Furthermore, the use of water as a solvent reduces dependency on petrochemical-derived organic solvents, mitigating risks associated with price volatility and supply disruptions in the raw material market. These factors combine to create a more resilient and cost-effective manufacturing model that supports long-term strategic sourcing goals.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts and toxic hydrogen sulfide from the process workflow leads to substantial cost savings by eliminating complex purification and waste treatment steps. Traditional methods require extensive downstream processing to meet residual metal specifications, whereas this enzymatic route inherently produces a cleaner crude product. The ability to recycle unreacted glycine and water further reduces raw material consumption, driving down the variable cost per kilogram of the final intermediate. These efficiencies translate into a more competitive pricing structure for the finished pharmaceutical ingredient without compromising on quality or regulatory compliance.
- Enhanced Supply Chain Reliability: The continuous flow design ensures a steady and predictable output of material, reducing the batch-to-batch variability often seen in traditional chemical synthesis. This consistency is crucial for maintaining uninterrupted supply to downstream API manufacturers who rely on just-in-time delivery models. The robustness of the immobilized enzyme system allows for extended operation cycles, minimizing downtime for reactor cleaning and catalyst replacement. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly reduced, providing a strategic advantage in a fast-paced market environment.
- Scalability and Environmental Compliance: Scaling this process is straightforward due to the modular nature of fixed-bed reactors, allowing capacity to be increased by adding parallel units rather than building massive new vessels. The green chemistry profile of the process, characterized by zero discharge of three wastes and low energy consumption, ensures compliance with increasingly strict environmental regulations globally. This future-proofs the supply chain against regulatory changes that might otherwise force the shutdown of older, more polluting manufacturing facilities, ensuring long-term continuity of supply for critical antibiotic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented biocatalytic technology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the process. Understanding these details is essential for stakeholders evaluating the potential integration of this route into their existing manufacturing portfolios.
Q: How does this enzymatic process compare to traditional chemical synthesis regarding environmental impact?
A: Traditional methods often utilize toxic hydrogen sulfide and heavy metals, generating significant hazardous waste. This patented flow process uses water as a solvent, eliminates heavy metal pollution, and achieves zero discharge of three wastes through material recycling.
Q: What specific enzyme modifications are utilized to achieve high conversion rates?
A: The process employs a genetically modified aldolase from Brevibacterium crescentum (Cc-H91A178), where specific amino acid positions are mutated to enhance stability and activity in a flow system, achieving conversion rates exceeding 96%.
Q: Can this process be scaled for industrial production of antibiotic intermediates?
A: Yes, the design utilizes a fixed-bed flow reaction system which is inherently scalable. The high space-time yield and continuous operation mode make it suitable for large-scale commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-3-Substituted Phenyl-3-Hydroxypropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this enzymatic flow technology for the production of high-value antibiotic intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-amino-3-substituted phenyl-3-hydroxypropionic acid meets the highest international standards. We are committed to leveraging advanced biocatalytic strategies to deliver superior quality intermediates that support the global supply of essential medicines.
We invite pharmaceutical partners to collaborate with us on optimizing their supply chains through the adoption of this green synthesis route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this sustainable technology into your production network. Together, we can drive efficiency and sustainability in the manufacturing of critical pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
