Revolutionizing End-Group Alkyne Production: Safe, Scalable, and Cost-Effective Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety, particularly for reactive intermediates like terminal alkynes. Patent CN101863732A introduces a groundbreaking synthesis method for end-group alkynes that fundamentally shifts the paradigm from hazardous cryogenic processes to manageable, ambient-pressure reactions. This technology utilizes a lithium acetylide ethylenediamine complex to facilitate nucleophilic substitution with haloalkanes, effectively bypassing the need for liquid ammonia at -78°C. For R&D Directors and Procurement Managers, this represents a significant opportunity to enhance the reliability of pharmaceutical intermediates supply chains while drastically reducing the safety risks associated with traditional alkyne manufacturing. The method ensures high yields and simplified post-reaction processing, making it an ideal candidate for the commercial scale-up of complex polymer additives and electronic chemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of terminal alkynes has relied heavily on the use of acetylene gas undergoing nucleophilic substitution in liquid ammonia at extremely low temperatures, typically around -78°C. This conventional approach presents severe logistical and safety challenges, including the requirement for specialized cryogenic equipment capable of maintaining such low temperatures consistently. The handling of liquid ammonia introduces significant occupational health hazards, necessitating rigorous safety protocols and expensive containment systems to prevent leaks and exposure. Furthermore, the energy consumption required to sustain these cryogenic conditions contributes substantially to the overall manufacturing cost, making the process economically inefficient for large-scale production. The complexity of the setup often leads to longer lead times for high-purity intermediates, as any deviation in temperature control can result in side reactions or reduced yields, compromising the quality of the final API intermediate.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a pre-formed lithium acetylide ethylenediamine complex, which allows the reaction to proceed at much milder temperatures ranging from -20°C to 20°C. This shift eliminates the dependency on liquid ammonia, thereby removing the associated high-pressure risks and the need for energy-intensive cooling infrastructure. The use of common organic solvents such as dimethyl sulfoxide (DMSO), tetrahydrofuran (THF), or ether further simplifies the process, enabling easier solvent recovery and waste management. By operating under these moderate conditions, the method significantly reduces the barrier to entry for manufacturers, allowing for more flexible production scheduling and cost reduction in pharmaceutical intermediates manufacturing. The streamlined workflow not only enhances safety but also improves the overall throughput, ensuring a more reliable supply of critical chemical building blocks for downstream applications.
Mechanistic Insights into Lithium Acetylide Ethylenediamine Catalyzed Substitution
The core of this innovative synthesis lies in the formation and stability of the lithium acetylide ethylenediamine complex, which acts as a potent nucleophile without the volatility of free acetylide ions. In this mechanism, ethylenediamine serves as a stabilizing ligand, coordinating with the lithium cation to solubilize the acetylide anion in organic media effectively. When the haloalkane is introduced to the cooled solution, the acetylide anion attacks the electrophilic carbon attached to the halogen, displacing the halide ion through a classic SN2 nucleophilic substitution pathway. The reaction temperature is strictly controlled between -10°C and 10°C during the addition phase to manage the exothermic nature of the bond formation, preventing thermal runaway. This precise thermal management is crucial for maintaining the integrity of the triple bond and ensuring that the reaction proceeds to completion without polymerization or decomposition of the sensitive alkyne product.
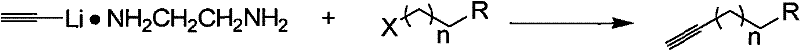
Impurity control is inherently built into this mechanistic design, as the mild reaction conditions minimize the formation of side products that typically arise from harsh basic environments or extreme temperatures. The stoichiometric ratio of lithium acetylide ethylenediamine to haloalkane is optimized between 1:1 and 2:1, ensuring that the limiting reagent is fully consumed while preventing excess base from degrading the product. Post-reaction workup involves simple extraction and washing steps, followed by distillation to isolate the fraction with the boiling point close to that of the target product. This high selectivity reduces the burden on purification columns, resulting in a final product with a cleaner impurity profile that meets the stringent requirements for high-purity OLED material or agrochemical intermediate applications. The robustness of this mechanism ensures consistent batch-to-batch reproducibility, a key factor for regulatory compliance in pharmaceutical synthesis.
How to Synthesize End-Group Alkyne Efficiently
Implementing this synthesis route requires careful attention to the preparation of the lithium acetylide complex and the subsequent addition of the haloalkane substrate under controlled thermal conditions. The process begins with the generation of the active complex in toluene, followed by dissolution in a polar aprotic solvent to facilitate the nucleophilic attack. Operators must monitor the temperature closely during the dropwise addition of the alkyl halide to manage the heat of reaction effectively. The detailed standardized synthesis steps see the guide below for specific parameters regarding reaction times and workup procedures tailored for different chain lengths.
- Prepare the lithium acetylide ethylenediamine complex by reacting metallic lithium with ethylenediamine in toluene under nitrogen protection at 90-105°C.
- Dissolve the complex in a solvent like DMSO or THF and cool the solution to a temperature range between -10°C and 10°C.
- Add the haloalkane substrate dropwise while maintaining the reaction temperature between -20°C and 20°C for 0.5 to 5 hours.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits that extend beyond mere chemical efficiency, directly impacting the bottom line and operational resilience. By eliminating the need for liquid ammonia and cryogenic cooling systems, manufacturers can achieve substantial cost savings through reduced capital expenditure on specialized equipment and lower energy consumption during production. The simplified safety profile allows for production in a wider range of facilities, enhancing supply chain reliability by diversifying the potential manufacturing base. This flexibility is critical for reducing lead time for high-purity intermediates, as it mitigates the risk of production stoppages due to safety inspections or equipment failures associated with hazardous materials. Furthermore, the use of readily available solvents and reagents ensures a stable supply of raw materials, protecting against market volatility.
- Cost Reduction in Manufacturing: The elimination of expensive cryogenic infrastructure and the reduction in energy usage for cooling significantly lower the operational costs associated with alkyne production. By avoiding the handling of hazardous liquid ammonia, facilities also save on compliance costs and safety monitoring systems, leading to a more economically viable process. The higher yields and simplified purification steps further contribute to cost efficiency by maximizing the output from each batch of raw materials. These factors combined result in a more competitive pricing structure for the final chemical products without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The milder reaction conditions and use of stable reagents reduce the risk of unplanned downtime caused by equipment malfunctions or safety incidents. This stability ensures a consistent flow of products to downstream customers, strengthening the partnership between suppliers and pharmaceutical or agrochemical companies. The ability to produce these intermediates in standard chemical reactors rather than specialized cryogenic vessels allows for greater flexibility in production scheduling and capacity allocation. Consequently, supply chain heads can plan with greater confidence, knowing that the production process is robust and less susceptible to external disruptions.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of extreme pressure or temperature requirements, facilitating the commercial scale-up of complex pharmaceutical intermediates. The reduced use of hazardous gases and the implementation of standard waste treatment protocols for organic solvents align with increasingly strict environmental regulations. This compliance minimizes the risk of regulatory penalties and enhances the company's reputation as a responsible manufacturer. The process generates less hazardous waste compared to traditional methods, simplifying disposal and contributing to a more sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkyne synthesis technology, based on the specific advantages outlined in the patent data. These answers are designed to provide clarity on the operational benefits and safety improvements offered by the new method. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing production lines.
Q: Why is the ethylenediamine complex method safer than traditional liquid ammonia synthesis?
A: Traditional methods require liquid ammonia at -78°C, posing significant safety risks and requiring specialized cryogenic equipment. The new method operates at -10°C to 20°C using a stable complex, eliminating high-pressure ammonia hazards.
Q: What solvents are compatible with this alkyne synthesis protocol?
A: The patent specifies dimethyl sulfoxide (DMSO), tetrahydrofuran (THF), or ether as effective solvents, providing flexibility for different scale-up requirements and downstream processing needs.
Q: Can this method be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the method avoids extreme cryogenic conditions and hazardous gases, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates with improved operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable End-Group Alkyne Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes to meet the evolving demands of the global pharmaceutical and fine chemical markets. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the lithium acetylide ethylenediamine method are translated into reliable industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of end-group alkyne meets the highest standards required for sensitive applications. Our commitment to technical excellence allows us to offer customized solutions that align with your specific project timelines and quality requirements.
We invite you to collaborate with us to optimize your supply chain and leverage these advanced synthesis technologies for your next project. Contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals. Let us help you secure a stable and cost-effective supply of high-quality chemical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
