Revolutionizing Phenylphosphonic Dichloride Production with Recyclable Ionic Liquid Catalysts for Commercial Scale
Revolutionizing Phenylphosphonic Dichloride Production with Recyclable Ionic Liquid Catalysts for Commercial Scale
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable manufacturing processes, particularly in the synthesis of critical organophosphorus intermediates. Patent CN101671366A introduces a groundbreaking environmentally-friendly synthesis method for phenylphosphonic dichloride, a vital building block for flame retardants, stabilizers, and pharmaceutical intermediates. This technology replaces the traditional, waste-intensive aluminum chloride catalysis with a novel Lewis acid ionic liquid system that operates under milder conditions. By utilizing benzene and phosphorus trichloride as raw materials in the presence of this tunable ionic catalyst, the process achieves high selectivity while fundamentally altering the downstream purification landscape. For R&D directors and procurement managers seeking a reliable phenylphosphonic dichloride supplier, this patent represents a significant leap forward in process efficiency and environmental compliance, offering a pathway to reduce both operational complexity and ecological footprint without compromising on product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of phenylphosphonic dichloride has relied heavily on the Friedel-Crafts reaction between benzene and phosphorus trichloride catalyzed by anhydrous aluminum chloride. While chemically effective, this traditional approach suffers from severe engineering and environmental drawbacks that hinder modern cost reduction in fine chemical intermediates manufacturing. The primary issue lies in the strong complexation between the product, phenylphosphonic dichloride, and the aluminum chloride catalyst, forming a stable adduct that cannot be separated by simple physical means. To break this complex, manufacturers are forced to introduce dissociating agents such as sodium chloride, pyridine, or phosphorus oxychloride, followed by intricate organic solvent extraction procedures. This not only consumes excessive amounts of catalyst—often in stoichiometric rather than catalytic quantities—but also generates substantial quantities of hazardous waste slag and wastewater containing aluminum salts and organic amines, creating a massive burden on waste treatment facilities and driving up disposal costs significantly.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a Lewis acid ionic liquid, specifically formed from quaternary ammonium salts and aluminum chloride, which acts as a liquid catalyst with tunable acidity. This innovation eliminates the need for solid catalyst handling and the subsequent complex dissociation steps entirely. The reaction mixture spontaneously separates into two distinct liquid phases upon cooling: a lower ionic liquid layer containing the active catalyst species and an upper organic layer comprising the product, unreacted benzene, and phosphorus trichloride. This physical separation allows for the direct decantation of the crude product mixture, bypassing the need for water quenching or chemical dissociation agents. Furthermore, the ionic liquid layer can be directly recycled back into the reactor after simple extraction to recover entrained product, enabling a closed-loop catalytic cycle that drastically simplifies the unit operations required for commercial scale-up of complex organophosphorus compounds.
Mechanistic Insights into Lewis Acid Ionic Liquid Catalysis
To fully appreciate the technical superiority of this method, one must understand the unique coordination chemistry occurring within the ionic liquid medium. Unlike solid AlCl3, the ionic liquid exists as a dynamic equilibrium of chloroaluminate anions, such as [AlCl4]- and [Al2Cl7]-, which serve as the active Lewis acid sites. The concentration of these species is governed by the molar ratio of the quaternary amine chloride to aluminum chloride, allowing chemists to precisely tune the electrophilicity of the catalyst to match the reactivity of phosphorus trichloride. This tunability prevents the over-chlorination or degradation of the aromatic ring, ensuring high selectivity for the mono-substituted phenylphosphonic dichloride. The liquid nature of the catalyst ensures homogeneous mixing at the molecular level during the reaction, enhancing mass transfer rates compared to heterogeneous solid catalysts, while the immiscibility with the organic product phase post-reaction facilitates the effortless separation that defines this green chemistry breakthrough.
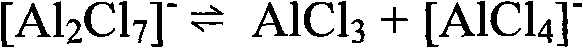
The reaction mechanism proceeds through the activation of phosphorus trichloride by the Lewis acidic ionic liquid. The electron-deficient aluminum centers in the ionic liquid coordinate with the lone pair electrons on the phosphorus atom of PCl3, increasing its electrophilic character and making it more susceptible to nucleophilic attack by the pi-electron cloud of the benzene ring. This activation step is crucial as phosphorus trichloride alone is often insufficiently reactive to undergo efficient Friedel-Crafts phosphorylation under mild conditions. The formation of the intermediate complex between the activated phosphorus species and the catalyst lowers the activation energy of the rate-determining step, allowing the reaction to proceed smoothly at reflux temperatures rather than requiring extreme thermal conditions that could lead to side reactions or safety hazards associated with handling large volumes of volatile chlorides.
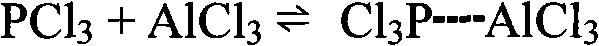
Following the activation, the electrophilic phosphorus species attacks the benzene ring to form a sigma-complex, which subsequently loses a proton to regenerate the aromatic system and release hydrogen chloride gas. A critical advantage of the ionic liquid system is its ability to stabilize the transition states without irreversibly binding the final product. In traditional systems, the product binds tightly to AlCl3, but in the ionic liquid matrix, the equilibrium dynamics favor the release of phenylphosphonic dichloride into the organic phase. This is partly due to the solvation properties of the ionic liquid, which prefers to solvate the ionic intermediates while excluding the neutral organic product, effectively pushing the equilibrium towards product formation and phase separation. This mechanistic nuance is what allows the process to avoid the formation of intractable sludge and enables the high-purity isolation of the target molecule.
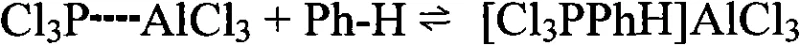
How to Synthesize Phenylphosphonic Dichloride Efficiently
The synthesis protocol outlined in the patent offers a robust framework for laboratory and pilot-scale production, emphasizing strict control over moisture and temperature to maintain catalyst integrity. The process begins with the in-situ or ex-situ preparation of the ionic liquid catalyst by reacting a quaternary amine hydrochloride, such as N-butylpyridinium chloride or triethylamine hydrochloride, with anhydrous aluminum chloride under a dry nitrogen atmosphere. Once the catalyst is prepared, it is charged into a reactor along with phosphorus trichloride and benzene in specific molar ratios, typically ranging from 1.5 to 2.5 equivalents of PCl3 relative to benzene to drive conversion. The mixture is heated to reflux, typically maintained for 6 to 18 hours depending on the specific catalyst composition and desired conversion levels, ensuring complete consumption of the limiting reagent while minimizing thermal stress on the equipment.
- Prepare the Lewis acid ionic liquid catalyst by mixing a quaternary amine salt (such as N-butylpyridinium chloride) with anhydrous aluminum chloride in a molar ratio between 1: 1 and 1:4 under nitrogen protection at 40-160°C.
- React benzene and phosphorus trichloride in the presence of the prepared ionic liquid catalyst under reflux conditions for 6 to 18 hours, maintaining a molar ratio of PCl3 to benzene between 1.5: 1 and 2.5:1.
- Allow the reaction mixture to settle for phase separation, extract the ionic liquid layer with phosphorus trichloride to recover product, and purify the combined organic layers via atmospheric and vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ionic liquid-catalyzed process translates into tangible strategic advantages beyond mere technical novelty. The most significant benefit is the drastic simplification of the downstream processing train, which directly correlates to reduced capital expenditure (CAPEX) and operating expenditure (OPEX). By eliminating the need for dissociating agents, extensive washing steps, and the handling of solid aluminum waste, the facility requirements are streamlined, leading to faster batch cycles and higher throughput. This efficiency gain is critical for maintaining a competitive edge in the global market for high-purity organophosphorus compounds, where delivery speed and consistency are paramount. Furthermore, the ability to recycle the catalyst multiple times reduces the dependency on fluctuating raw material markets for aluminum chloride, providing a more stable cost structure for long-term supply contracts.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the catalytic nature of the ionic liquid and the elimination of auxiliary chemicals. In traditional methods, the cost of dissociating agents like pyridine or sodium chloride, combined with the loss of catalyst in the waste stream, adds a significant premium to the final product cost. Here, the catalyst is retained within the system and reused, meaning the variable cost per kilogram of product is driven primarily by the commodity prices of benzene and phosphorus trichloride, which are generally stable. Additionally, the recovery of unreacted benzene and phosphorus trichloride via distillation further enhances the atom economy, ensuring that raw material spend is maximized and waste disposal fees are minimized, resulting in substantial overall cost savings for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and simplicity of the reaction system. Traditional processes involving solid catalysts and complex workups are prone to bottlenecks, such as filtration issues or emulsion formation during extraction, which can delay shipments and disrupt just-in-time inventory models. The liquid-liquid separation inherent in this ionic liquid method is rapid and predictable, reducing the risk of batch failures or extended cycle times. Moreover, the catalyst components are commercially available and easy to handle, reducing the risk of supply disruptions for specialized reagents. This reliability ensures that a reliable phenylphosphonic dichloride supplier can meet tight delivery windows consistently, fostering stronger partnerships with downstream pharmaceutical and agrochemical clients who depend on uninterrupted material flow.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify globally, this technology offers a future-proof solution for commercial scale-up of complex organophosphorus intermediates. The process generates negligible solid waste and avoids the discharge of aluminum-rich wastewater, aligning perfectly with stringent environmental regulations in Europe and North America. The closed-loop nature of the catalyst recycling minimizes the release of volatile organic compounds and corrosive gases, enhancing workplace safety and reducing the need for expensive scrubbing systems. This environmental compatibility not only mitigates regulatory risk but also enhances the brand value of the end-product, appealing to multinational corporations with strict sustainability mandates who prioritize green chemistry in their sourcing decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what potential partners can expect regarding performance and operational parameters. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or for assessing the quality profile of the resulting intermediates.
Q: How does the ionic liquid catalyst improve separation compared to traditional AlCl3?
A: Traditional methods form stable complexes between phenylphosphonic dichloride and AlCl3 requiring harsh dissociating agents. The ionic liquid method creates a biphasic system where the product separates naturally into an organic layer, allowing direct decantation without complexing agents.
Q: What is the expected purity and yield of this green synthesis method?
A: According to patent embodiments, this method achieves yields around 60-67% based on benzene, with product purity exceeding 98.8% as confirmed by gas chromatography, containing minimal phosphorus trichloride residues.
Q: Can the ionic liquid catalyst be recycled for industrial production?
A: Yes, the ionic liquid catalyst demonstrates excellent recyclability. It can be reused up to 8 times with maintained activity before a noticeable decline in yield occurs, significantly reducing catalyst consumption costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylphosphonic Dichloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic technologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably translated into industrial reality. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of phenylphosphonic dichloride meets the exacting standards required for pharmaceutical and high-performance polymer applications. Our commitment to process innovation allows us to offer products that are not only chemically superior but also produced with a focus on sustainability and cost-efficiency.
We invite you to collaborate with us to optimize your supply chain for organophosphorus intermediates. By leveraging our expertise in ionic liquid catalysis and green synthesis, we can help you achieve significant operational improvements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing processes can support your R&D and production goals effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
