Advanced Manufacturing of 2-Bromo-3-Fluorobenzoic Acid for Global Pharmaceutical Supply Chains
Advanced Manufacturing of 2-Bromo-3-Fluorobenzoic Acid for Global Pharmaceutical Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective synthetic routes for critical fluorinated building blocks. A significant technological advancement in this domain is detailed in patent CN102898301A, which discloses a novel preparation method for 2-bromo-3-fluorobenzoic acid. This compound serves as a vital intermediate in the synthesis of various bioactive molecules, including kinase inhibitors and agrochemical agents. The disclosed methodology represents a paradigm shift from traditional cryogenic lithiation strategies to a more pragmatic, ambient-temperature sequence involving nitration, bromination, reduction, and deamination. For R&D directors and procurement specialists, understanding the nuances of this patent is crucial, as it offers a pathway to secure supply chains that are less dependent on hazardous reagents and extreme energy consumption. The transition from laboratory-scale curiosity to industrial reality requires a deep appreciation of the chemical engineering principles embedded in this invention, particularly its ability to maintain high purity while utilizing commodity chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ortho-substituted fluorobenzoic acids has been plagued by significant operational challenges that hinder commercial viability. Prior art, such as the methods described in PCT Int. Appl. 2008154221, relies heavily on the diazotization of 2-amino-3-fluorobenzoic acid, a starting material that is itself expensive and difficult to procure in bulk quantities. Furthermore, alternative routes documented in chemical literature often necessitate the use of organolithium reagents, such as sec-butyllithium or LDA, under strictly cryogenic conditions ranging from -75°C to lower. These low-temperature requirements impose a massive burden on manufacturing infrastructure, demanding specialized refrigeration equipment and resulting in exorbitant energy costs. The use of pyrophoric reagents also introduces severe safety risks, requiring inert atmosphere handling and complex quenching protocols that slow down production throughput. As illustrated in the reaction scheme below, the direct lithiation of m-fluorobenzoic acid followed by bromination is chemically elegant but industrially cumbersome, creating a bottleneck for reliable pharmaceutical intermediate supplier networks seeking to minimize risk.
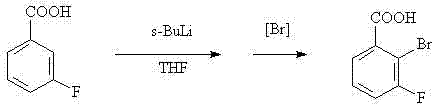
The Novel Approach
In stark contrast to the hazardous legacy methods, the process outlined in CN102898301A utilizes fluorobenzotrifluoride as a readily available and inexpensive starting material. This new route operates under mild conditions, with key reaction steps such as nitration and bromination occurring at ambient temperatures between 20°C and 30°C. The elimination of cryogenic cooling not only drastically reduces energy consumption but also simplifies the reactor design, allowing for the use of standard glass-lined or stainless-steel vessels common in fine chemical plants. The bromination step employs dibromohydantoin as a controlled bromine source, which offers better handling safety compared to elemental bromine. Following bromination, the reduction is performed using reduced iron powder in an aqueous phase, a classic yet highly effective method that avoids the need for expensive noble metal catalysts like palladium on carbon. This strategic selection of reagents ensures that the cost reduction in pharma intermediate manufacturing is realized through both raw material savings and operational efficiency. The overall process design prioritizes safety and scalability, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
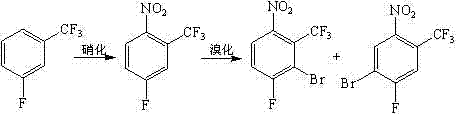
Mechanistic Insights into Electrophilic Aromatic Substitution and Deamination
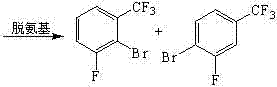
From a mechanistic perspective, the success of this synthesis relies on the precise control of electrophilic aromatic substitution and the subsequent manipulation of the amino group. The initial nitration of fluorobenzotrifluoride is directed by the electronic effects of the trifluoromethyl and fluoro substituents, yielding 4-fluoro-2-trifluoromethyl nitrobenzene with high regioselectivity. The subsequent bromination step is critical, as it introduces the bromine atom ortho to the nitro group. The use of dibromohydantoin in sulfuric acid generates the electrophilic bromine species in situ, allowing for a controlled reaction that minimizes poly-bromination side products. Following the reduction of the nitro group to an amine using iron powder, the process employs a reductive deamination strategy. This involves the formation of a diazonium salt intermediate, which is then treated with phosphorous acid. This specific deamination mechanism is superior to traditional copper-catalyzed Sandmeyer reactions because it avoids the introduction of heavy metal impurities that are difficult to remove from the final API intermediate. The ability to cleanly remove the amino functionality while retaining the bromine and fluoro substituents is the key to achieving the high-purity fluorinated building blocks required by modern drug discovery programs.
Impurity control is a paramount concern for R&D directors evaluating this technology, particularly regarding the separation of isomeric byproducts. The bromination and deamination steps generate mixtures containing both the desired 2-bromo-3-fluoro isomer and the 4-bromo-3-fluoro isomer. The patent specifies a vacuum fractionation step to separate these isomers based on their boiling point differences. This physical separation method is highly scalable and does not require complex chromatography, which is often a bottleneck in large-scale production. The final hydrolysis step converts the trifluoromethyl group into the carboxylic acid functionality using concentrated sulfuric acid at elevated temperatures (150-175°C). This harsh condition is necessary to cleave the strong carbon-fluorine bonds of the CF3 group, but the robustness of the intermediate ensures that the aromatic ring remains intact. The rigorous QC labs required for such a process are standard in top-tier manufacturing facilities, ensuring that stringent purity specifications are met for every batch produced. The mechanistic clarity of this route provides confidence in its reproducibility and robustness across different manufacturing sites.
How to Synthesize 2-Bromo-3-Fluorobenzoic Acid Efficiently
Implementing this synthesis route requires a systematic approach to unit operations, starting from the careful dosing of nitrating agents to the final crystallization of the acid. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing overall cycle time. For example, the bromination mixture can often be carried forward to the reduction step after a simple aqueous workup, avoiding the need for full purification of the nitro-bromo intermediate. The reduction step utilizes a heterogeneous mixture of iron powder and water, which requires efficient stirring and temperature control to manage the exotherm. Following reduction, the deamination step must be conducted with precise pH control to ensure complete diazotization without decomposing the sensitive diazonium species. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and workup procedures necessary to achieve optimal yields and purity.
- Nitration of fluorobenzotrifluoride using sulfuric acid and nitric acid at 20-30°C to form 4-fluoro-2-trifluoromethyl nitrobenzene.
- Bromination using dibromohydantoin in sulfuric acid, followed by reduction with iron powder in aqueous phase.
- Deamination using phosphorous acid, vacuum fractionation for isomer separation, and final hydrolysis with sulfuric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible benefits that extend beyond simple chemistry. The primary advantage lies in the substantial cost savings achieved by eliminating the need for cryogenic infrastructure and expensive organometallic reagents. By operating at ambient temperatures, the process significantly reduces energy consumption, which is a major component of the cost of goods sold (COGS) in chemical manufacturing. Furthermore, the use of commodity chemicals like sulfuric acid, nitric acid, and iron powder ensures that the supply chain is resilient to fluctuations in the market prices of specialty reagents. This stability is crucial for long-term supply agreements with pharmaceutical companies that require consistent pricing over the lifecycle of a drug product. The process also enhances supply chain reliability by utilizing standard equipment that is widely available, reducing the risk of production delays due to equipment scarcity or specialized maintenance requirements.
- Cost Reduction in Manufacturing: The elimination of cryogenic cooling and pyrophoric reagents leads to a drastic simplification of the manufacturing process, resulting in significant operational expenditure savings. The use of iron powder for reduction instead of catalytic hydrogenation or noble metals removes the cost associated with catalyst recovery and precious metal procurement. Additionally, the high atom economy of the nitration and bromination steps ensures that raw material utilization is optimized, further driving down the variable costs per kilogram of product. These factors combine to create a highly competitive cost structure that allows for better margin management in a price-sensitive market.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as fluorobenzotrifluoride ensures that production is not held hostage by the supply constraints of exotic intermediates. The robustness of the reaction conditions means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or reagent quality. This consistency is vital for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for re-processing or batch rejection. A stable and predictable manufacturing process allows supply chain planners to maintain lower safety stock levels while still meeting customer demand reliably.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as vacuum fractionation and aqueous extraction that are standard in multi-purpose chemical plants. The avoidance of heavy metal catalysts simplifies waste stream treatment, as there is no need for complex metal scavenging or hazardous waste disposal protocols associated with palladium or copper residues. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the facility more attractive to eco-conscious partners. The ability to scale from pilot plant to commercial production without significant process redesign ensures a smooth technology transfer and faster time-to-market for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2-bromo-3-fluorobenzoic acid using this patented methodology. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation, providing clarity on safety, purity, and scalability. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their supply chain. The responses highlight the practical advantages of the new route over traditional methods, focusing on operational safety and economic efficiency.
Q: What are the primary safety advantages of this new synthesis route?
A: The novel process eliminates the need for cryogenic conditions (-75°C) and pyrophoric reagents like s-BuLi or LDA, significantly reducing operational hazards and safety infrastructure costs.
Q: How is the regioselectivity controlled during the bromination step?
A: Regioselectivity is managed through precise temperature control (20-25°C) and the use of dibromohydantoin, yielding a mixture of isomers that are subsequently separated via vacuum fractionation.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of standard industrial unit operations such as vacuum fractionation and aqueous phase reduction makes the process highly scalable and compatible with existing chemical manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-3-Fluorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development. Our technical team has extensively analyzed the route described in CN102898301A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that translating a patent into a commercial reality requires more than just following a recipe; it demands deep process engineering expertise to optimize yield, safety, and environmental performance. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of 2-bromo-3-fluorobenzoic acid meets the exacting standards required for GMP manufacturing. We are committed to providing a reliable 2-bromo-3-fluorobenzoic acid supplier partnership that supports your long-term strategic goals.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic advantages of switching to this manufacturing method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs. Our goal is to be more than just a vendor; we aim to be a strategic partner in your supply chain, offering the technical depth and commercial flexibility needed to navigate the complexities of modern drug development.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
