Revolutionizing Lyral Production: A Catalyst-Free Diels-Alder Strategy for Global Fragrance Markets
The global fragrance and flavor industry constantly seeks innovative synthetic pathways that balance molecular complexity with operational efficiency. A pivotal advancement in this domain is detailed in patent CN102531858B, which outlines a novel preparation method for Lyral, a critical ingredient known for its stable and lasting lily-of-the-valley note. This technology represents a significant departure from traditional high-energy synthesis routes, offering a streamlined approach that eliminates the need for harsh Lewis acid catalysts or extreme high-pressure conditions. For R&D Directors and Procurement Managers alike, understanding the nuances of this Diels-Alder cycloaddition is essential for optimizing supply chains and reducing the environmental footprint of daily chemical production. The method leverages precise thermal control and reactant addition strategies to achieve exceptional purity levels, setting a new benchmark for the reliable synthetic flavors & fragrances supplier market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Lyral has been plagued by significant operational hurdles that impact both cost and safety. Traditional methods, such as those disclosed in earlier literature like US4007137, often require sealing reactants in high-pressure vessels at temperatures exceeding 150°C for extended periods. This approach not only demands expensive, specialized reactor equipment capable of withstanding immense pressure but also poses substantial safety risks due to the volatile nature of acrolein at elevated temperatures. Furthermore, alternative routes utilizing Lewis acid catalysts like zinc chloride or tin tetrachloride introduce a different set of complications. These catalytic systems necessitate the use of organic solvents such as toluene, which must be meticulously removed post-reaction. The subsequent neutralization and washing steps generate large volumes of waste acid, leading to severe equipment corrosion and creating a heavy burden on environmental compliance teams. These factors collectively drive up the cost of Lyral manufacturing and complicate the commercial scale-up of complex fragrance intermediates.
The Novel Approach
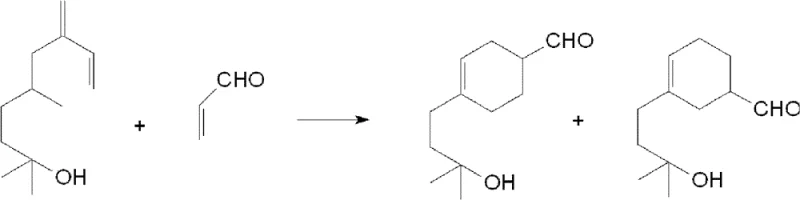
In stark contrast, the methodology described in CN102531858B introduces a remarkably elegant solution that simplifies the entire production workflow. By heating myrcenol to a moderate range of 50°C to 130°C and subsequently dropwise adding acrolein, the process effectively manages the exothermic nature of the Diels-Alder reaction without external pressure. This controlled addition prevents the steep temperature spikes that typically cause acrolein volatilization and side reactions. The absence of Lewis acid catalysts and organic solvents means that the post-reaction workup is drastically simplified to a basic water wash followed by vacuum rectification. This not only reduces the consumption of auxiliary chemicals but also eliminates the generation of corrosive waste acid, aligning perfectly with modern green chemistry principles. For supply chain heads, this translates to a more robust and continuous production capability, reducing lead time for high-purity fragrance intermediates while minimizing downtime associated with complex cleaning and maintenance procedures.
Mechanistic Insights into Solvent-Free Diels-Alder Cyclization
The core of this technological breakthrough lies in the precise manipulation of reaction kinetics through thermal regulation and reactant stoichiometry. Myrcenol, acting as the diene component, possesses a conjugated double bond system that is highly susceptible to self-polymerization if not properly managed. To counteract this, the process incorporates specific polymerization inhibitors, such as resorcinol or 2,6-di-tert-butyl-p-cresol, at concentrations between 0.1wt% and 0.3wt%. As the acrolein, serving as the dienophile, is introduced dropwise over a period of 6 to 10 hours, it immediately reacts with the heated myrcenol. This slow addition rate ensures that the concentration of free acrolein remains low, thereby suppressing its tendency to self-polymerize while favoring the desired [4+2] cycloaddition. The reaction heat is continuously dissipated, maintaining the system within the optimal 50°C to 130°C window, which is critical for maximizing the yield of the target cyclohexene derivative.
Following the completion of the acrolein addition, the reaction mixture is held at temperature for an additional 1 to 2 hours to ensure full conversion of the starting materials. This curing phase is vital for minimizing residual impurities that could affect the olfactory profile of the final fragrance. The resulting crude product is then subjected to a straightforward purification process involving washing with deionized water to remove any water-soluble by-products, followed by high-vacuum rectification at pressures below 2mmHg. This gentle distillation allows for the separation of Lyral based on its boiling point range of 120°C to 125°C, yielding a product with a purity exceeding 99% as confirmed by gas chromatography. The structural integrity of the final molecule, preserving the delicate aldehyde functionality essential for its scent, is maintained throughout this mild processing.
How to Synthesize Lyral Efficiently
Implementing this synthesis route requires strict adherence to the thermal and addition parameters defined in the patent to ensure reproducibility and safety on an industrial scale. The process begins with the preparation of the myrcenol feedstock, ensuring it contains the necessary inhibitors to prevent premature polymerization during the heating phase. Operators must utilize precise metering pumps to control the dropwise addition of acrolein, as the rate of addition is directly correlated with the ability to control the exothermic reaction temperature. Detailed standardized synthesis steps see the guide below for specific operational protocols that guarantee the high purity and yield required for commercial fragrance applications.
- Heat Myrcenol to a controlled temperature range of 50°C to 130°C while incorporating a polymerization inhibitor.
- Dropwise add Acrolein over a period of 6 to 10 hours, maintaining strict temperature control to manage exothermic heat.
- Continue reaction for 1 to 2 hours post-addition, followed by water washing and high-vacuum rectification to isolate pure Lyral.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this catalyst-free synthesis method offers profound economic and logistical benefits that extend beyond simple raw material costs. The elimination of Lewis acid catalysts and organic solvents removes an entire layer of supply chain complexity, as there is no longer a need to source, store, and dispose of these hazardous auxiliary chemicals. This simplification directly contributes to substantial cost savings in Lyral manufacturing by reducing the overhead associated with chemical procurement and waste management. Furthermore, the mild reaction conditions significantly lower the barrier to entry for production facilities, as standard atmospheric reactors can be utilized instead of costly high-pressure autoclaves. This flexibility enhances supply chain reliability by allowing for production across a wider range of manufacturing sites without requiring specialized infrastructure upgrades.
- Cost Reduction in Manufacturing: The removal of Lewis acid catalysts and organic solvents from the process flow eliminates the need for expensive post-reaction neutralization and solvent recovery steps. This reduction in unit operations translates to significantly lower energy consumption and labor costs per kilogram of product. Additionally, the absence of corrosive waste acid extends the lifespan of reaction vessels and piping, reducing capital expenditure on equipment replacement and maintenance. By streamlining the purification process to a simple water wash and distillation, the overall production cycle time is optimized, allowing for higher throughput and better asset utilization without compromising on quality standards.
- Enhanced Supply Chain Reliability: Operating under atmospheric pressure and moderate temperatures drastically reduces the safety risks associated with high-pressure chemical synthesis. This improved safety profile minimizes the likelihood of unplanned shutdowns due to safety incidents or regulatory inspections, ensuring a more consistent and continuous supply of product. The use of readily available raw materials like myrcenol and acrolein, combined with a robust process that tolerates minor variations, further stabilizes the supply chain. This reliability is crucial for meeting the demanding delivery schedules of global fragrance houses, reducing lead time for high-purity fragrance intermediates and strengthening long-term supplier partnerships.
- Scalability and Environmental Compliance: The green chemistry attributes of this method, specifically the absence of heavy metal catalysts and volatile organic solvents, align perfectly with increasingly stringent global environmental regulations. This compliance reduces the administrative burden and costs associated with environmental permitting and waste disposal. The process is inherently scalable, as the heat management strategy via dropwise addition can be effectively translated from pilot scale to multi-ton commercial production. This scalability ensures that manufacturers can respond agilely to market demand fluctuations, providing a secure source of supply for daily chemical flavors without the environmental liabilities associated with traditional synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Lyral synthesized via this advanced method. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages for potential partners. Understanding these details is key for R&D teams evaluating process feasibility and for procurement officers assessing the total cost of ownership.
Q: Why is the catalyst-free method superior for Lyral manufacturing?
A: Eliminating Lewis acid catalysts removes the need for complex post-treatment neutralization and washing steps, significantly reducing waste acid generation and equipment corrosion risks.
Q: How does this process ensure high purity without organic solvents?
A: By utilizing a controlled dropwise addition of acrolein into heated myrcenol, the reaction minimizes side reactions and self-polymerization, achieving over 99% purity through simple vacuum rectification.
Q: What are the safety advantages of avoiding high-pressure conditions?
A: Operating at atmospheric pressure and moderate temperatures (50-130°C) reduces the requirement for specialized high-pressure vessels, lowering capital expenditure and enhancing operational safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lyral Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern fragrance industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the catalyst-free Lyral synthesis can be seamlessly integrated into your supply chain. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Lyral performs consistently in your final formulations. Our capability to handle complex Diels-Alder chemistries positions us as a strategic partner for companies seeking to optimize their raw material sourcing.
We invite you to collaborate with us to explore how this advanced manufacturing technology can drive value for your organization. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your transition to this superior grade of Lyral is smooth and commercially viable.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
