Advanced Purification Technology for WF11899A: Enhancing Commercial Scalability and Purity Standards
Advanced Purification Technology for WF11899A: Enhancing Commercial Scalability and Purity Standards
The pharmaceutical industry continuously seeks robust methodologies for isolating high-value antifungal intermediates, and patent CN103483427A presents a transformative approach to the purification of Echinocandin compounds, specifically WF11899A. This intellectual property details a sophisticated three-stage process encompassing prepurification solution preparation, macroporous resin chromatography separation, and a refined crystallization protocol that collectively address longstanding inefficiencies in downstream processing. By eliminating the reliance on complex mixed solvent systems and mitigating the risks of acid-induced degradation common in prior art, this technology offers a pathway to significantly higher sample purity and operational simplicity. For global procurement and R&D leaders, understanding the nuances of this purification breakthrough is essential for securing a reliable supply chain of critical pharmaceutical intermediates used in the synthesis of next-generation antifungal APIs like Caspofungin and Anidulafungin.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of Echinocandin compounds has been plagued by cumbersome multi-step procedures that compromise both yield and structural integrity. Prior art, such as US5021403 and US20100249371, often relies on repeated resin absorption combined with silica gel column chromatography or salt sorbent materials that exhibit poor stability during actual operation. A critical failure point in existing technologies, notably disclosed in patents like US6506726, is the dependence on acidic gradient elution with acetic acid concentrations between 0.1% and 10%, which frequently fails to effectively wash out WF11899A while simultaneously exposing the sensitive cyclic peptide structure to degradative acidic conditions. Furthermore, traditional precipitation methods described in WO2008048627 necessitate the addition of mixed solvents and precipitating carriers like diatomite to prevent the formation of oily residues that are difficult to filter, thereby introducing additional contaminants and complicating the waste treatment profile for large-scale manufacturing facilities.
The Novel Approach
In stark contrast to these legacy methodologies, the novel purification process defined in CN103483427A leverages a targeted macroporous resin strategy that operates under neutral or controlled pH conditions to preserve molecular stability. This innovative route utilizes specific polystyrene resins, such as HP20SS or CG161, to achieve selective adsorption and subsequent elution using aqueous organic solvent solutions with concentrations optimized between 50% and 70% (V/V), ensuring complete recovery of the target compound without the need for harsh acidic gradients. The subsequent crystallization step represents a paradigm shift by employing a single solvent system, such as isopropanol or ethanol, added directly to the concentrated eluent at controlled temperatures below 40°C, which facilitates the formation of high-purity white crystals without the requirement for filtration aids or complex solvent exchanges. This streamlined workflow not only enhances the chromatographic purity from initial eluate levels of approximately 70% to final crystal purities exceeding 94% but also drastically simplifies the equipment requirements and solvent recovery logistics for commercial production.
Mechanistic Insights into Resin Chromatography and Crystallization Dynamics
The core of this technological advancement lies in the precise manipulation of hydrophobic interactions between the Echinocandin macrocycle and the porous surface of the selected polystyrene resin matrix. Unlike reversed-phase chromatography media that may require aggressive acidic modifiers to disrupt binding, the macroporous resins specified in this patent, including HP20 and SP825, offer a balanced surface energy that allows for strong adsorption from aqueous fermentation broths followed by clean desorption using moderate concentrations of alcohols or acetonitrile. This mechanism minimizes the residence time of the WF11899A molecule in potentially degrading environments and reduces the co-elution of structurally related by-products that typically contaminate the final API intermediate. The selectivity is further enhanced by the specific pore size distribution of resins like HZ20SS, which physically excludes larger impurity molecules while retaining the target echinocandin, thereby acting as a molecular sieve that complements the chemical adsorption properties.
Following the chromatographic enrichment, the crystallization mechanism is driven by the controlled reduction of solubility through the addition of a water-miscible organic anti-solvent to the concentrated aqueous eluate. By maintaining the crystallization temperature between -5°C and 10°C, the process kinetically favors the nucleation of the WF11899A lattice over the incorporation of residual impurities, resulting in a highly ordered crystal structure with minimal solvent inclusion. 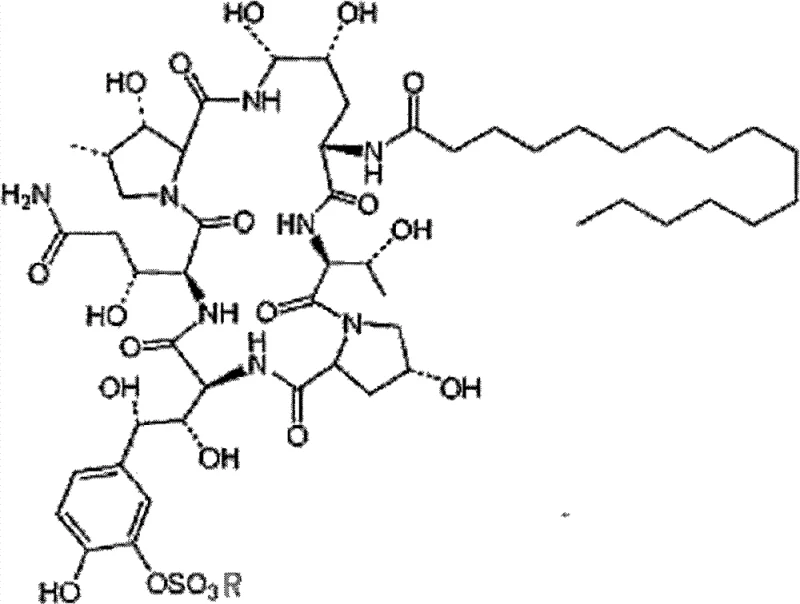 This structural integrity is paramount, as the molecule features a complex cyclic hexapeptide core linked to a fatty acid side chain, which is susceptible to hydrolysis or oxidation if exposed to improper processing conditions. The ability to achieve purities of 96% through this single-solvent crystallization confirms that the thermodynamic conditions are precisely tuned to exclude the specific by-products generated during microbial fermentation, ensuring the material is suitable for subsequent semi-synthetic modifications.
This structural integrity is paramount, as the molecule features a complex cyclic hexapeptide core linked to a fatty acid side chain, which is susceptible to hydrolysis or oxidation if exposed to improper processing conditions. The ability to achieve purities of 96% through this single-solvent crystallization confirms that the thermodynamic conditions are precisely tuned to exclude the specific by-products generated during microbial fermentation, ensuring the material is suitable for subsequent semi-synthetic modifications.
How to Synthesize WF11899A Efficiently
The implementation of this purification protocol requires strict adherence to the defined parameters for resin selection, solvent concentration, and temperature control to replicate the high-yield results observed in the patent examples. Operators must first ensure the fermentation broth is properly clarified and concentrated to the specified range of 100-500mg/ml before loading onto the resin column to prevent channeling and ensure maximum adsorption capacity. The detailed standardized synthesis steps, including specific flow rates, resin bed volumes, and exact crystallization times, are critical for maintaining batch-to-batch consistency and achieving the reported purity benchmarks in a GMP environment.
- Prepare the prepurification solution by collecting mycelium through filtration, soaking in organic solvent, and concentrating under reduced pressure.
- Perform resin chromatography separation using polystyrene resins like HP20SS, involving adsorption, washing with aqueous organic solvent, and elution.
- Execute crystallization by concentrating the eluent and adding a specific solvent like isopropanol at controlled temperatures below 40°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this purification technology offers substantial benefits by fundamentally simplifying the manufacturing workflow and reducing the dependency on specialized, hard-to-source reagents. The elimination of mixed solvent systems in the crystallization phase translates directly to simplified solvent recovery operations, as distillation units can be dedicated to single-component streams rather than complex azeotropic separations, leading to significant operational expenditure savings over the lifecycle of the product. Furthermore, the robustness of the macroporous resin against acidic degradation means that the stationary phase has a prolonged operational lifespan, reducing the frequency of column repacking and the associated downtime that often disrupts supply continuity for high-demand pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the removal of expensive and hazardous processing aids such as diatomite and the reduction of solvent consumption associated with mixed-solvent precipitation techniques. By utilizing common industrial solvents like ethanol and isopropanol at moderate concentrations, the facility can leverage bulk purchasing power and minimize waste disposal costs associated with halogenated or exotic organic solvents. Additionally, the higher purity achieved in the initial crystallization step reduces the burden on downstream polishing processes, effectively lowering the overall cost of goods sold by minimizing yield losses during secondary purification stages.
- Enhanced Supply Chain Reliability: The simplicity of the resin chromatography method ensures that production campaigns can be executed with greater predictability, as the risk of batch failure due to compound degradation is substantially mitigated compared to acidic elution methods. This reliability is crucial for maintaining consistent inventory levels of WF11899A, allowing procurement managers to plan API synthesis schedules with confidence and avoid the costly expedited shipping fees often incurred when reacting to unexpected production shortfalls. The use of stable, commercially available polystyrene resins also insulates the supply chain from fluctuations in the availability of specialized silica-based or salt-based sorbent materials.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is facilitated by the linear scalability of column chromatography and the straightforward nature of the single-solvent crystallization, which does not require complex mixing dynamics to prevent oiling out. From an environmental standpoint, the reduction in solvent variety and the absence of solid filtration aids like diatomite significantly lower the volume of hazardous solid waste generated, aligning with increasingly stringent global environmental regulations and corporate sustainability goals. This green chemistry profile enhances the marketability of the final intermediate to eco-conscious pharmaceutical partners who prioritize suppliers with robust environmental management systems.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific WF11899A purification technology. These insights are derived directly from the comparative data and experimental embodiments provided in the patent documentation, offering clarity on how this method outperforms historical precedents in terms of purity and operational ease. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing manufacturing infrastructure.
Q: Why is the new WF11899A purification method superior to traditional acidic gradient elution?
A: Traditional acidic gradient elution often fails to wash out WF11899A effectively and risks degrading the compound under acidic conditions. The new method utilizes specific resin adsorption and neutral pH conditions to prevent degradation while ensuring complete elution.
Q: How does this process eliminate the need for mixed solvents in crystallization?
A: By optimizing the resin elution concentration and selecting specific single solvents like isopropanol or ethanol for crystallization, the process avoids the complexity and waste associated with mixed solvent systems, simplifying downstream processing.
Q: What purity levels can be achieved with this resin-based purification technology?
A: Experimental data indicates that chromatographic purity can be significantly increased from approximately 70-78% in the eluate to over 93-96% in the final crystallized product, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable WF11899A Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced purification technologies like the one described in CN103483427A to deliver exceptional quality pharmaceutical intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of WF11899A meets the exacting standards required for antifungal API synthesis.
We invite international partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of these superior processing methods. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the competitive pharmaceutical landscape.
