Revolutionizing Production of 2-Nitro-4-Trifluoromethylbenzoic Acid via Advanced One-Step Nitration Technology
Revolutionizing Production of 2-Nitro-4-Trifluoromethylbenzoic Acid via Advanced One-Step Nitration Technology
In the rapidly evolving landscape of fine chemical synthesis, the demand for efficient, cost-effective routes to fluorinated aromatic intermediates has never been more critical for global supply chains. A groundbreaking technical disclosure found in patent CN108558672B presents a transformative approach to synthesizing 2-nitro-4-trifluoromethylbenzoic acid and its isomer, 3-nitro-4-trifluoromethylbenzoic acid. This innovation addresses long-standing inefficiencies in prior art by utilizing 4-trifluoromethyl benzonitrile as a robust starting material, subjected to a novel one-step nitration and hydrolysis protocol. For R&D directors and procurement strategists alike, this methodology represents a significant leap forward, replacing expensive precursors with accessible nitriles while achieving total yields that consistently surpass 90 percent under optimized conditions. The strategic implementation of this technology allows manufacturers to bypass complex multi-step sequences, thereby streamlining operations and enhancing the economic viability of producing these vital building blocks for pharmaceuticals and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-nitro-4-trifluoromethylbenzoic acid has been plagued by economic and operational inefficiencies that hindered large-scale adoption. Prior art, such as the process disclosed in US4845279, relied heavily on the use of 4-trifluoromethyl-benzoyl halides or 4-trifluoromethyl-trihalotoluenes as primary raw materials. These starting materials are not only prohibitively expensive due to complex upstream synthesis requirements but also introduce significant safety hazards associated with handling reactive halides. Furthermore, the conventional pathways often suffered from mediocre total yields, typically capping out between 70 percent and 76 percent, which resulted in substantial material loss and increased waste generation. The necessity for separate nitration and hydrolysis steps in older methods further complicated the workflow, requiring rigorous control over reaction conditions to prevent side reactions, ultimately driving up the cost of goods sold and extending production lead times for downstream users.
The Novel Approach
The innovative strategy detailed in the patent data fundamentally reimagines the synthetic route by leveraging the unique reactivity of 4-trifluoromethyl benzonitrile. By employing a potent nitrating agent composed of mixed acids, this method facilitates a concurrent nitration and hydrolysis reaction in a single vessel, effectively collapsing multiple processing stages into one seamless operation. This one-pot transformation not only drastically simplifies the operational workflow but also leverages the electron-withdrawing nature of the trifluoromethyl group to direct nitration efficiently. The result is a robust process capable of delivering high-purity 2-nitro-4-trifluoromethylbenzoic acid alongside its valuable isomer with exceptional efficiency. For a reliable pharmaceutical intermediate supplier, adopting this route means accessing a feedstock that is significantly cheaper and more stable than acyl halides, while simultaneously unlocking yield improvements that can reach upwards of 94.6 percent under optimal thermal conditions, thereby maximizing asset utilization.
Mechanistic Insights into Mixed-Acid Catalyzed Nitration and Hydrolysis
The core of this technological breakthrough lies in the precise manipulation of reaction thermodynamics and kinetics to overcome the inherent stability of the cyano group. Under typical mild acidic conditions, the hydrolysis of a nitrile to a carboxylic acid is kinetically sluggish and often stalls at the amide intermediate stage, particularly when competing with electrophilic aromatic substitution. However, the patented process utilizes a carefully balanced mixture of concentrated sulfuric acid and fuming nitric acid to generate a highly reactive nitronium ion species while maintaining a sufficiently acidic environment to drive the hydration of the nitrile. The presence of the strong electron-withdrawing trifluoromethyl group deactivates the aromatic ring, necessitating vigorous conditions; yet, the specific acid matrix employed ensures that the nitration occurs regioselectively at the ortho and meta positions relative to the cyano group before or concurrent with its conversion to the carboxyl functionality.
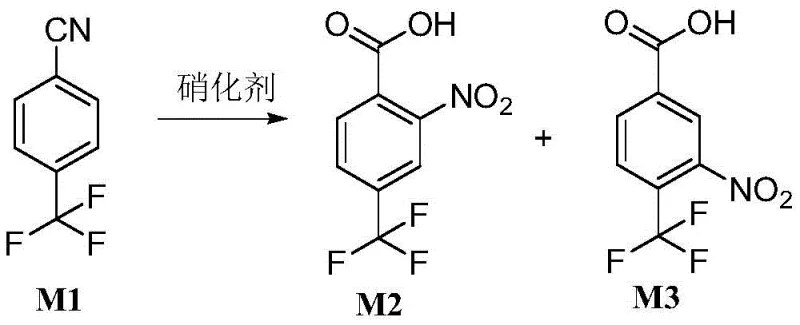
Furthermore, the mechanism involves a delicate interplay between temperature and acid concentration to suppress the formation of unwanted byproducts. Experimental data indicates that at lower temperatures, such as 40°C, the reaction favors the formation of the amide or proceeds with incomplete conversion, whereas elevating the temperature to the 60-70°C range provides the necessary activation energy to fully hydrolyze the intermediate amide to the target carboxylic acid. This mechanistic understanding is crucial for quality control, as it explains why strict temperature regulation is paramount for achieving the high selectivity observed in the examples. By optimizing the molar ratio of nitric acid to the substrate between 0.9 and 2.5:1, the process minimizes oxidative degradation while ensuring complete conversion, resulting in a clean impurity profile that simplifies downstream purification and meets the stringent requirements for high-purity agrochemical intermediates.
How to Synthesize 2-Nitro-4-Trifluoromethylbenzoic Acid Efficiently
Implementing this synthesis requires careful attention to the preparation of the nitrating agent and the control of exothermic events during the addition phase. The process is remarkably versatile, accommodating both solvent-free conditions and the use of halogenated hydrocarbons like dichloromethane or 1,2-dichloroethane, offering flexibility based on existing plant infrastructure. The operational simplicity allows for a straightforward workup procedure involving aqueous quenching and organic extraction, which is highly advantageous for reducing solvent consumption and waste treatment costs. Below is the standardized procedural framework derived from the patent examples, designed to guide process engineers in replicating these high-yield results.
- Prepare the nitrating agent by mixing concentrated sulfuric acid and fuming nitric acid under cooling conditions to ensure stability and reactivity.
- Add 4-trifluoromethyl benzonitrile to a reactor, optionally with a halogenated hydrocarbon solvent like dichloromethane or 1,2-dichloroethane, or proceed under solvent-free conditions.
- Dropwise add the nitrating agent to the substrate at a controlled temperature between 60-70°C, maintain reaction for 3-7 hours, and isolate products via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits that extend far beyond simple chemistry. The shift from expensive acyl halides to widely available benzonitriles fundamentally alters the cost structure of the final product, enabling significant margin improvements without compromising quality. Additionally, the reduction in unit operations—from multiple steps to a single reaction pot—drastically lowers energy consumption and labor requirements, contributing to a leaner manufacturing footprint. This efficiency translates directly into enhanced supply chain resilience, as the reliance on fewer, more commoditized raw materials mitigates the risk of shortages and price volatility often associated with specialized halogenated precursors.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like 4-trifluoromethyl-benzoyl halides results in a substantial decrease in raw material expenditure, which is often the largest component of COGS in fine chemical production. Moreover, the consolidation of nitration and hydrolysis into a single step removes the need for intermediate isolation and purification, thereby saving on solvent usage, filtration media, and drying energy. This streamlined approach inherently reduces the overall processing time per batch, increasing throughput capacity and allowing manufacturers to respond more agilely to market demand fluctuations without incurring proportional cost increases.
- Enhanced Supply Chain Reliability: By utilizing 4-trifluoromethyl benzonitrile, a stable and commercially abundant commodity chemical, the supply chain becomes less vulnerable to the bottlenecks associated with custom-synthesized halides. The robustness of the reaction conditions, which tolerate a range of solvent choices including solvent-free operation, provides manufacturing flexibility that ensures continuity of supply even if specific solvent markets face disruptions. This reliability is critical for maintaining long-term contracts with downstream pharmaceutical and agrochemical clients who require consistent delivery schedules and uninterrupted material flow for their own production lines.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability, with data showing consistent high yields across various batch sizes and reaction times, making it ideal for commercial scale-up of complex fluorinated intermediates. The ability to operate under solvent-free conditions or with recoverable chlorinated solvents significantly reduces the volume of hazardous waste generated, aligning with increasingly strict environmental regulations and sustainability goals. Simplified workup procedures involving basic extraction minimize the use of auxiliary chemicals, further lowering the environmental impact and reducing the costs associated with waste disposal and regulatory compliance reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of using 4-trifluoromethyl benzonitrile over benzoyl halides?
A: Using 4-trifluoromethyl benzonitrile significantly reduces raw material costs compared to expensive benzoyl halides or trihalotoluenes. Furthermore, it enables a direct one-step conversion to the target carboxylic acid, eliminating the need for separate hydrolysis steps and improving overall process efficiency.
Q: How does this method address the selectivity issues between amide and acid formation?
A: Traditional mild acidic conditions often stall at the amide stage. This patented method utilizes optimized mixed acid concentrations and elevated temperatures (specifically around 70°C) to drive the hydrolysis of the cyano group directly to the carboxyl group, ensuring high selectivity for the desired benzoic acid derivatives.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable. It operates effectively under solvent-free conditions or with common solvents like dichloromethane, uses readily available reagents, and achieves total yields exceeding 90%, making it economically viable for commercial production of pharmaceutical and agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Nitro-4-Trifluoromethylbenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists has thoroughly analyzed the potential of the one-step nitration technology described in CN108558672B and is prepared to assist partners in scaling this efficient route from laboratory benchtop to full industrial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition to this new process is seamless and risk-mitigated. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-nitro-4-trifluoromethylbenzoic acid delivered meets the exacting standards required for API and crop protection synthesis.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability. Let us be your trusted partner in navigating the complexities of fluorinated intermediate production.
