Advanced Nano-Polymer Chromatography for Commercial Scale Production of High-Purity Teicoplanin API
The pharmaceutical industry continuously seeks robust purification technologies to meet the stringent quality standards of modern glycopeptide antibiotics. Patent CN102690333B introduces a transformative preparation method for high-purity teicoplanin, addressing critical challenges in component ratio control and scalability. Unlike traditional methods that struggle with the complex separation of teicoplanin analogues, this innovation utilizes nano-polymer microspheres to achieve a purity exceeding 95.0% while strictly adhering to European Pharmacopoeia specifications. For R&D directors and procurement specialists, this represents a significant leap forward in reliable antibiotic API supplier capabilities, ensuring that the final active pharmaceutical ingredient possesses the precise structural integrity required for clinical efficacy. The method simplifies the downstream processing workflow, replacing cumbersome gel chromatography with a more durable and efficient nano-polymer based system that is inherently more suitable for industrial amplification.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of teicoplanin from fermentation broths has relied heavily on macroporous resin adsorption followed by gel chromatography or standard ultrafiltration techniques. While these methods can achieve basic separation, they often fail to precisely control the ratio of the six structural proximates, specifically the A2 and A3 groups, which are critical for biological activity and regulatory compliance. Conventional gel matrices, such as dextran gels, suffer from low physical strength and poor chemical stability, limiting the operating pressure and flow rates during chromatography. This results in prolonged processing times, broad peak elution, and significant band broadening, which ultimately compromises the resolution between closely related impurities. Furthermore, traditional processes often require multiple decolorization and desalting steps that increase solvent consumption and reduce overall yield, creating bottlenecks in cost reduction in pharmaceutical manufacturing for high-volume production facilities.
The Novel Approach
The novel approach detailed in the patent leverages the unique physicochemical properties of nano-polymer microspheres to overcome the inherent limitations of soft gel matrices. These microspheres, typically composed of polystyrene derivatives like PS-30 or PSA-30 types, possess a rigid skeleton structure that withstands high flow rates and high-pressure operations without deformation or channeling. By employing a gradient elution strategy with a polar solvent and water mixture, the method achieves superior resolution, allowing for the fractional collection of teicoplanin desorbed solutions with exact component specifications. This precision ensures that the A2 group sum remains not less than 80.0% and individual components like A2-2 are maintained within the 35.0% to 55.0% range. The integration of nanofiltration for desalting and concentration further streamlines the process, significantly reducing solvent usage and enabling a more environmentally sustainable and economically viable production pathway for commercial scale-up of complex glycopeptides.
Mechanistic Insights into Nano-Polymer Microsphere Chromatography
The core of this purification technology lies in the interaction between the teicoplanin molecules and the high-specific surface area of the nano-polymer microspheres. With a specific surface area reaching as high as 1000 m²/g, these microspheres provide an extensive interface for adsorption and separation, facilitating rapid mass transfer kinetics even under high flow conditions. The rigid polymeric structure prevents the swelling and shrinking issues common in hydrophilic gels, ensuring consistent column performance over hundreds of cycles. During the gradient elution, the varying polarity of the mobile phase—typically involving ethanol or methanol concentrations between 25% and 70%—modulates the retention time of different teicoplanin analogues based on their hydrophobicity and structural nuances. This mechanism allows for the precise isolation of the desired A2 components from the A3 group and other impurities, ensuring the final product meets the rigorous impurity profile standards demanded by global regulatory bodies.
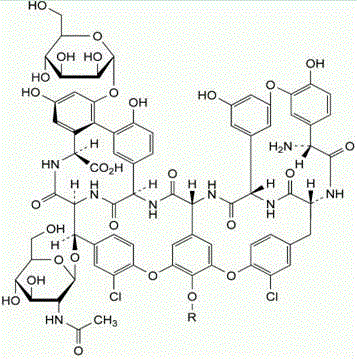
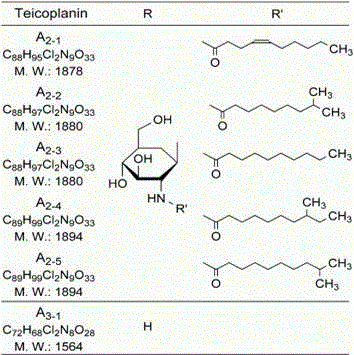
Understanding the structural variability of teicoplanin is essential for appreciating the complexity of its purification. The molecule exists as a mixture of six major components, distinguished primarily by the fatty acyl side chains attached to the amino sugar moiety. As illustrated in the structural diagrams, variations in the R and R' groups define the specific analogues such as A2-1 through A2-5 and A3-1. The biological potency and pharmacokinetic properties are highly dependent on the precise distribution of these components. The nano-polymer chromatography method excels in resolving these subtle structural differences, which traditional methods often blur together. By controlling the elution profile, manufacturers can selectively enrich the most potent fractions while excluding less active or potentially immunogenic variants, thereby enhancing the therapeutic index of the final API.
Impurity control is another critical aspect of the mechanistic design, particularly regarding the removal of fermentation by-products and colored pigments. The process incorporates a dedicated decolorization step using activated carbon or specific ion-exchange resins immediately following chromatography. This step is vital for removing high molecular weight impurities and chromophores that could affect the stability and appearance of the final powder. Subsequent nanofiltration using membranes with a molecular weight cut-off of 100Da to 800Da effectively removes salts and residual solvents without losing the high molecular weight teicoplanin molecules. This multi-stage purification strategy ensures that the final crystalline powder not only meets purity thresholds but also exhibits excellent physical properties, such as flowability and solubility, which are crucial for downstream formulation into injectable dosage forms.
How to Synthesize High-Purity Teicoplanin Efficiently
The synthesis and purification protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to industrial production. The process begins with the dissolution of crude teicoplanin in a buffered aqueous solution, followed by loading onto the nano-polymer column. The detailed standardized synthesis steps involve precise control of flow rates, typically around 1500mL/h for industrial columns, and careful monitoring of eluent fractions via HPLC. The merging of fractions is dynamically adjusted based on real-time analysis to ensure the final blend meets the target component ratios. This level of process control is essential for maintaining batch-to-batch consistency, a key requirement for any reliable antibiotic API supplier serving the global market.
- Dissolve crude teicoplanin in water or buffer salt solution and load onto a chromatographic column packed with nano-polymer microspheres.
- Perform gradient elution using a polar solvent and water mixture to separate components, collecting fractions based on HPLC analysis.
- Decolorize the pooled solution with activated carbon, concentrate via nanofiltration, and crystallize using acetone to obtain fine powder.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nano-polymer chromatography method offers substantial strategic advantages beyond mere technical performance. The robustness of the polymeric stationary phase significantly extends column life compared to traditional gels, reducing the frequency of media replacement and lowering long-term operational expenditures. Furthermore, the use of common organic solvents like ethanol and acetone, rather than exotic or hazardous reagents, simplifies solvent recovery and waste management protocols. This alignment with green chemistry principles not only reduces environmental compliance costs but also mitigates supply chain risks associated with the procurement of specialized chemicals. The streamlined workflow, which combines separation, decolorization, and concentration into a cohesive sequence, minimizes hold times and intermediate storage requirements, thereby enhancing overall production throughput and responsiveness to market demand.
- Cost Reduction in Manufacturing: The elimination of fragile gel matrices and the reduction in solvent consumption directly contribute to significant cost optimization in the manufacturing process. By enabling high-flow rate operations, the cycle time for each batch is drastically reduced, allowing for higher equipment utilization rates without compromising resolution. Additionally, the efficient recovery of solvents like acetone and ethanol through standard distillation processes further lowers the variable costs associated with raw material consumption. These efficiencies compound over large production volumes, resulting in a more competitive cost structure for the final teicoplanin API, which is critical for maintaining margins in the generic pharmaceutical sector.
- Enhanced Supply Chain Reliability: The scalability of the nano-polymer technology ensures that supply can be rapidly ramped up to meet surges in demand without the need for extensive process re-validation. The physical stability of the microspheres means that columns can be shipped and stored with less risk of damage compared to pre-packed gel columns, simplifying logistics. Moreover, the method's ability to consistently produce material that meets European Pharmacopoeia standards reduces the risk of batch rejection and regulatory delays. This reliability is paramount for reducing lead time for high-purity antibiotics, ensuring that downstream formulation partners receive consistent quality material on schedule.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex antibiotics, with parameters that translate linearly from pilot to production scale. The reduced solvent footprint and the use of less hazardous materials simplify the environmental permitting process for manufacturing facilities. Nanofiltration technology further minimizes aqueous waste streams by concentrating the product efficiently before crystallization. These factors collectively enhance the sustainability profile of the manufacturing site, aligning with the increasing corporate social responsibility goals of major pharmaceutical buyers and ensuring long-term operational viability in strict regulatory environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from the specific technical advantages and operational parameters detailed in the patent documentation, providing clarity for stakeholders evaluating this method for their supply chain. Understanding these nuances is critical for making informed decisions about technology transfer and vendor qualification.
Q: How does nano-polymer chromatography improve teicoplanin purity compared to traditional gel filtration?
A: Nano-polymer microspheres offer higher physical strength and chemical stability than dextran gels, allowing for high-flow rate operations without channeling. This results in superior resolution and the ability to strictly control the A2 and A3 component ratios required by the European Pharmacopoeia.
Q: What solvents are utilized in the gradient elution process for this purification method?
A: The process employs a gradient of semi-polar solvents such as methanol, ethanol, or isopropanol mixed with water. The volume concentration of the polar solvent typically ranges from 25% to 70%, optimizing the separation of teicoplanin analogues.
Q: Is this purification method suitable for large-scale commercial manufacturing?
A: Yes, the method is designed for scalability. The use of robust nano-polymer supports and simplified solvent systems (ethanol/acetone) reduces operational complexity and solvent consumption, making it viable for producing medicinal teicoplanin fine powder on a multi-ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Teicoplanin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity antibiotics in the global healthcare landscape. Our technical team has extensively analyzed the advancements presented in patent CN102690333B and integrated similar high-efficiency purification principles into our own CDMO operations. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with rigorous QC labs and advanced chromatographic systems capable of meeting stringent purity specifications, guaranteeing that every batch of teicoplanin we supply adheres to the highest international pharmacopoeial standards.
We invite you to collaborate with us to optimize your antibiotic supply chain. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our advanced manufacturing capabilities can enhance your product's market competitiveness. Contact us today to discuss how we can support your next project with reliable, high-quality teicoplanin intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
