Revolutionizing Statin Side Chain Production: A One-Step Enzymatic Breakthrough for Commercial Scale
The pharmaceutical industry continuously seeks more efficient pathways for constructing complex chiral molecules, particularly the side chains essential for HMG-CoA reductase inhibitors, commonly known as statins. Patent CN112501223B introduces a transformative methodology for preparing 2-((2S,4S)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl)acetonitrile, a critical chiral intermediate in the synthesis of these life-saving medications. This innovation leverages a sophisticated whole-cell catalytic system that integrates aldolase DERA, dehydrogenase ADH, and a nitrile converting enzyme into a single, streamlined operation. By utilizing acetaldehyde and chloroacetaldehyde as primary feedstocks, the process achieves a remarkable convergence of acetalization, oxidation, and cyanation reactions in one pot. This technical advancement addresses long-standing challenges in biopharmaceutical manufacturing, specifically the instability of free enzymes and the complexity of multi-step chemical routes. For global procurement leaders, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-value precursors with improved consistency and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of statin side chains has been plagued by inefficient multi-step protocols that rely heavily on hazardous chemical reagents and unstable biocatalysts. Prior art, such as the routes depicted in existing literature, often necessitates the use of cyanoacetaldehyde or complex protection group strategies that drastically increase raw material costs and process time. Furthermore, free enzyme systems, while selective, are notoriously susceptible to inactivation by high concentrations of aldehyde substrates, leading to inconsistent batch-to-batch performance and lower overall yields. The requirement for separate oxidation steps using stoichiometric oxidants not only escalates production expenses but also generates substantial quantities of chemical waste that require costly treatment. These cumulative inefficiencies create bottlenecks in the supply chain, making it difficult to achieve the cost reduction in API manufacturing that modern generic and branded drug producers demand to maintain competitiveness in a tightening market.
The Novel Approach
In stark contrast, the methodology disclosed in CN112501223B utilizes a robust immobilized whole-cell catalyst to drive the transformation of simple aldehydes directly into the target lactone nitrile structure. This novel approach eliminates the need for isolated intermediate purification and avoids the use of expensive, unstable free enzymes by encapsulating them within a protective hydrogel matrix. The integration of three distinct enzymatic activities within a single microbial host allows for a seamless cascade reaction where the product of one enzyme serves immediately as the substrate for the next. This telescoping of reactions significantly reduces solvent usage and processing time, directly contributing to a more sustainable and economically viable manufacturing model. By overcoming the limitations of enzyme inactivation and complex synthetic sequences, this technology paves the way for the commercial scale-up of complex pharmaceutical intermediates with unprecedented efficiency and reliability.
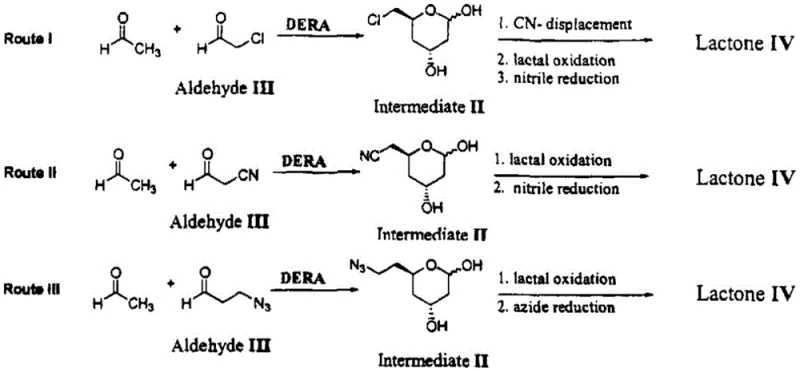
The visual comparison of synthetic routes highlights the dramatic simplification achieved by the new biocatalytic method. While traditional pathways involve multiple discrete chemical transformations and purification stages, the patented process consolidates these into a unified biological system. This reduction in unit operations not only lowers capital expenditure requirements for manufacturing facilities but also minimizes the risk of product loss during transfer between steps. For supply chain managers, this simplification translates into shorter production cycles and a more resilient supply network capable of responding rapidly to market fluctuations. The ability to produce high-purity statin intermediate materials through such a direct route is a key differentiator for manufacturers aiming to optimize their procurement strategies and reduce dependency on fragmented supply chains.
Mechanistic Insights into Whole-Cell Cascade Catalysis
At the heart of this technological breakthrough lies a meticulously engineered metabolic cascade within recombinant Escherichia coli, specifically strain DAC0-003. The process initiates with the aldolase DERA catalyzing the condensation of acetaldehyde and chloroacetaldehyde to form a hemiacetal intermediate, which is subsequently oxidized by the dehydrogenase ADH. Crucially, the system incorporates a cofactor regeneration cycle where NADH is continuously recycled, ensuring that the oxidation step proceeds without the need for expensive external cofactor addition. The final step involves the conversion of the chloro-group to a nitrile functionality via the nitrile converting enzyme, completing the construction of the chiral pyranone ring system. This intricate coordination of enzymatic activities within the cellular environment provides a protective milieu that stabilizes the catalysts against the toxic effects of the aldehyde substrates.
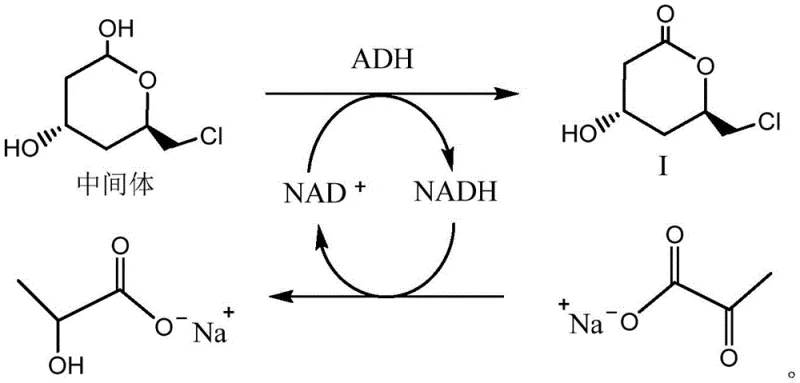
The mechanism of cofactor regeneration is particularly vital for maintaining the economic feasibility of the process on an industrial scale. As illustrated in the reaction scheme, the oxidation of the intermediate alcohol to the ketone consumes NAD+, which is regenerated through the coupled reduction of a co-substrate, thereby sustaining the catalytic cycle. This self-sustaining system eliminates the need for stoichiometric amounts of oxidizing agents, which are often costly and generate hazardous byproducts. Furthermore, the immobilization of the whole cells within a DA-F127 hydrogel matrix enhances the mechanical stability of the biocatalyst, allowing it to withstand the shear forces of industrial stirring and facilitating easy recovery from the reaction mixture. Such mechanistic robustness is essential for reducing lead time for high-purity pharmaceutical intermediates, as it ensures consistent reaction kinetics and minimizes downtime associated with catalyst replacement or regeneration.
How to Synthesize 2-((2S,4S)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl)acetonitrile Efficiently
The practical implementation of this synthesis involves a series of precise biotechnological and chemical engineering steps designed to maximize yield and purity. Initially, the recombinant bacterial strain is cultivated and harvested, followed by immobilization within the hydrogel matrix to create the final whole-cell catalyst. This catalyst is then introduced into a reactor containing the substrate mixture of acetaldehyde, chloroacetaldehyde, and sodium cyanide under controlled pH and temperature conditions. The reaction proceeds at mild temperatures, typically between 30°C and 35°C, which significantly reduces energy consumption compared to traditional high-temperature chemical processes. Detailed standardized synthesis steps see the guide below.
- Preparation of Recombinant E. coli DAC0-003 expressing DERA, ADH, and nitrile converting enzymes via plasmid transformation and fermentation.
- Immobilization of the harvested bacterial cells using DA-F127 hydrogel and UV crosslinking to create a robust whole-cell catalyst.
- Execution of the one-pot reaction at 30-35°C with acetaldehyde, chloroacetaldehyde, and NaCN, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this biocatalytic technology offers substantial strategic advantages beyond mere technical novelty. The simplification of the synthetic route directly correlates with a reduction in the number of raw materials required, thereby mitigating the risk of supply disruptions associated with sourcing multiple specialized reagents. Additionally, the mild reaction conditions and aqueous-based system significantly lower the safety hazards and regulatory burdens associated with handling volatile organic solvents and hazardous oxidants. This shift towards greener chemistry aligns with increasingly stringent environmental regulations, ensuring long-term compliance and reducing the potential for production stoppages due to environmental violations. Consequently, partners can expect a more stable and predictable supply of critical intermediates, essential for maintaining uninterrupted drug manufacturing operations.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps inherently reduces the consumption of solvents, energy, and labor, leading to significant operational cost savings. By avoiding the use of expensive stoichiometric oxidants and protecting groups, the raw material cost profile is drastically improved, allowing for more competitive pricing structures. The high catalytic efficiency and reusability of the immobilized whole-cell system further amortize the cost of the biocatalyst over multiple batches, enhancing the overall economic viability of the process. These factors combine to deliver a manufacturing solution that offers substantial cost savings without compromising on the quality or purity of the final product.
- Enhanced Supply Chain Reliability: The use of commodity chemicals like acetaldehyde and chloroacetaldehyde as starting materials ensures a robust and readily available supply base, reducing dependency on niche suppliers. The stability of the immobilized catalyst allows for consistent production schedules and minimizes the risk of batch failures due to enzyme instability, which is a common issue in traditional biocatalysis. This reliability is crucial for pharmaceutical manufacturers who require guaranteed delivery timelines to meet their own production commitments and regulatory filing deadlines. A stable supply of high-quality intermediates strengthens the entire value chain, from raw material sourcing to final drug product distribution.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium and the absence of hazardous heavy metal catalysts simplify waste treatment processes, significantly reducing the environmental footprint of the manufacturing site. The immobilized catalyst system is inherently scalable, as the hydrogel beads can be easily handled in large-scale reactors using standard filtration and separation equipment. This scalability ensures that the process can be seamlessly transitioned from pilot plant to full commercial production without significant re-engineering or capital investment. Compliance with green chemistry principles not only enhances corporate sustainability profiles but also future-proofs the manufacturing process against evolving environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the consensus on the operational advantages and limitations observed during the development of this technology.
Q: How does this one-step enzymatic method improve upon traditional multi-step chemical synthesis?
A: Traditional methods often require multiple protection and deprotection steps, leading to lower overall yields and higher waste generation. This patent describes a convergent one-step process that combines acetalization, oxidation, and cyanation, significantly simplifying the workflow and reducing solvent consumption.
Q: What are the stability advantages of the immobilized whole-cell catalyst described in CN112501223B?
A: The use of DA-F127 hydrogel immobilization protects the enzymes from denaturation caused by organic substrates like aldehydes. This enhances the operational stability of the catalyst, allowing for repeated use and consistent performance over extended reaction periods compared to free enzymes.
Q: Is this process suitable for large-scale commercial production of statin intermediates?
A: Yes, the mild reaction conditions (30-35°C) and the use of inexpensive raw materials like acetaldehyde and chloroacetaldehyde make it highly scalable. The immobilized catalyst system facilitates easy separation from the reaction mixture, a critical factor for industrial feasibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-((2S,4S)-4-hydroxy-6-oxotetrahydro-2H-pyran-2-yl)acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of pharmaceutical intermediate manufacturing. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN112501223B can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch conforms to the highest international standards. Our infrastructure is designed to support the complex requirements of biocatalytic processes, including specialized fermentation and downstream processing capabilities.
We invite global partners to collaborate with us to optimize their supply chains and achieve significant competitive advantages through the adoption of this advanced synthesis technology. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of how this route can specifically benefit your production economics and timeline. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments tailored to your project requirements. Together, we can drive innovation and efficiency in the production of essential pharmaceutical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
