Advanced Grignard-Based Synthesis of Higenamine Hydrochloride for Commercial Scale-Up
Introduction to Advanced Higenamine Hydrochloride Manufacturing
The pharmaceutical landscape for cardiovascular therapeutics demands intermediates of exceptional purity and consistent supply reliability. Patent CN103554022A introduces a transformative synthetic methodology for Higenamine hydrochloride (CAS: 11041-94-4), a critical active pharmaceutical ingredient known for its efficacy in treating cardiac insufficiency and chronic arrhythmia. This innovative approach fundamentally restructures the production pathway by leveraging a highly efficient Grignard reaction coupled with a streamlined demethylation process. By addressing the inherent limitations of traditional multi-step syntheses, this technology offers a robust solution for cost reduction in API manufacturing while simultaneously elevating product quality standards. The strategic elimination of hazardous reagents and the consolidation of reaction steps represent a significant leap forward in green chemistry principles applied to fine chemical synthesis.
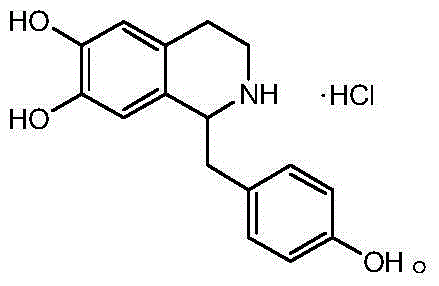
For global procurement teams and R&D directors, the implications of this patent extend beyond mere chemical novelty; it represents a tangible opportunity to optimize supply chains for high-purity pharmaceutical intermediates. The structural integrity of Higenamine hydrochloride, characterized by its specific hydroxylation pattern on the isoquinoline ring, is preserved and enhanced through this gentle yet effective synthetic route. As a reliable pharmaceutical intermediates supplier, understanding the nuances of such patented methodologies allows us to offer clients not just a commodity, but a value-added partnership grounded in technical superiority and regulatory compliance. The following analysis dissects the mechanistic advantages and commercial viability of this process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Higenamine hydrochloride has been plagued by inefficiencies and safety hazards that hinder large-scale adoption. Prior art, such as the method disclosed in CN1539823A, relies on a cumbersome four-step sequence involving condensation, cyclization, hydrogenation, and demethoxylation. A critical bottleneck in these legacy processes is the reliance on aggressive and dangerous reagents like phosphorus oxychloride (POCl3) for cyclization and strong reducing agents for subsequent steps. These conditions not only pose severe occupational health and safety risks but also generate complex waste streams that are costly to treat and dispose of in an environmentally compliant manner. Furthermore, the multiplicity of reaction steps inherently accumulates yield losses at each stage; documented total recoveries for these conventional routes often stagnate around a mere 37%, rendering them economically unviable for competitive commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in CN103554022A achieves the target molecule through a concise two-step strategy that dramatically simplifies the operational workflow. The first step utilizes a Grignard reaction to construct the carbon-carbon bond between the benzyl moiety and the isoquinoline core, bypassing the need for harsh cyclization agents. The second step involves a direct acidic hydrolysis to remove protecting groups, followed by simple salification. This reduction in step count directly correlates to a reduction in processing time, solvent consumption, and labor costs. By avoiding the use of toxic phosphorus compounds and unstable hydrides, the new route aligns with modern sustainability goals while delivering a product with significantly improved purity profiles. This streamlined architecture is essential for reducing lead time for high-purity pharmaceutical intermediates in a fast-paced market.
Mechanistic Insights into Grignard Addition and Acidic Hydrolysis
The core of this synthetic innovation lies in the precise execution of the Grignard reaction under controlled low-temperature conditions. The process initiates with the activation of magnesium chips using iodine and a halohydrocarbon initiator in an anhydrous solvent such as tetrahydrofuran. This activation ensures the rapid formation of the organomagnesium species from 4-methoxybenzyl bromide. Subsequently, this nucleophilic Grignard reagent attacks the electrophilic C1 position of 6,7-dimethoxy-3,4-dihydroisoquinoline. Maintaining the reaction temperature between -70°C and -60°C is critical to suppress side reactions and ensure regioselectivity, leading to the formation of the tetrahydroisoquinoline intermediate with high fidelity. The careful control of stoichiometry, specifically maintaining a molar ratio of the isoquinoline to the Grignard reagent between 1:1.2 and 1:2.0, further optimizes conversion efficiency and minimizes the formation of unreacted starting materials or over-addition byproducts.
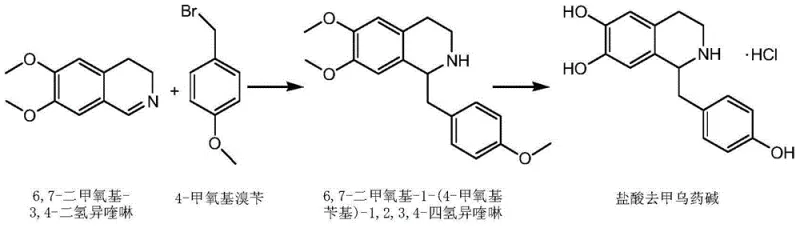
Following the construction of the carbon skeleton, the demethylation step employs acidic hydrolysis, typically using hydrobromic acid or formic acid at elevated temperatures (120-140°C). This step cleaves the methyl ether protecting groups to reveal the crucial phenolic hydroxyl groups required for biological activity. Unlike oxidative demethylation methods that can degrade the sensitive isoquinoline ring, this acidic approach is remarkably selective and mild regarding the core structure. The subsequent neutralization with a mild base like ammonia or sodium bicarbonate allows for the precipitation of the free base, which is then converted to the stable hydrochloride salt. This sequence effectively manages impurity profiles, as evidenced by HPLC analysis showing major peak purities exceeding 98%, thereby meeting the rigorous quality expectations of reliable pharmaceutical intermediates supplier networks globally.
How to Synthesize Higenamine Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to anhydrous conditions during the Grignard formation and precise temperature control during the addition phases. The process begins with the preparation of the Grignard reagent in a dry reactor, followed by the slow addition of the dihydroisoquinoline substrate to manage exothermicity. After aqueous workup and extraction, the crude intermediate is subjected to reflux in acidic media to effect deprotection. The final isolation involves pH adjustment and crystallization from aqueous hydrochloric acid. For detailed operational parameters, stoichiometry, and specific workup protocols, please refer to the standardized guide below.
- Perform a Grignard reaction by reacting 4-methoxybenzyl bromide with magnesium chips in anhydrous solvent, followed by addition to 6,7-dimethoxy-3,4-dihydroisoquinoline at low temperature.
- Execute acidic hydrolysis using hydrobromic acid or similar acids at elevated temperatures to remove methoxy protective groups.
- Neutralize the resulting salt with a base to isolate the free base, followed by salification with hydrochloric acid to obtain the final Higenamine hydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthetic route offers profound advantages in terms of cost structure and supply security. The elimination of hazardous reagents like phosphorus oxychloride removes the need for specialized corrosion-resistant equipment and expensive waste neutralization protocols, leading to substantial cost savings in capital expenditure and operational overhead. Furthermore, the simplified two-step process reduces the overall cycle time per batch, allowing manufacturing facilities to increase throughput without expanding their physical footprint. This efficiency is critical for maintaining competitive pricing in the global pharmaceutical intermediates market, where margin compression is a constant challenge for buyers.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis drastically reduces raw material consumption and solvent usage compared to the four-step legacy methods. By consolidating the synthesis into fewer unit operations, manufacturers can significantly lower energy consumption and labor costs associated with multiple isolation and purification stages. The avoidance of expensive and dangerous catalysts further contributes to a leaner cost of goods sold (COGS), enabling more aggressive pricing strategies for downstream API producers seeking cost reduction in API manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically 4-methoxybenzyl bromide and 6,7-dimethoxy-3,4-dihydroisoquinoline, are commercially available and chemically stable, reducing the risk of supply disruptions associated with custom-synthesized precursors. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality. This reliability is paramount for reducing lead time for high-purity pharmaceutical intermediates and securing long-term supply agreements with major pharmaceutical companies.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind. The use of standard solvents like tetrahydrofuran and ethyl acetate simplifies solvent recovery and recycling, aligning with green chemistry initiatives. Moreover, the absence of heavy metal catalysts or toxic phosphorus waste simplifies the environmental impact assessment and regulatory filing process, accelerating the time-to-market for new drug applications relying on this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Higenamine hydrochloride using this advanced methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on yield, purity, and process safety.
Q: What are the primary safety advantages of this new synthetic route compared to prior art?
A: Unlike conventional methods that utilize hazardous reagents such as phosphorus oxychloride (POCl3) or lithium aluminum hydride, this novel Grignard-based approach operates under milder conditions without dangerous chemicals, significantly enhancing operational safety and reducing waste treatment complexity.
Q: How does the new method improve yield and purity for industrial applications?
A: By condensing the synthesis into two main steps rather than four, the process minimizes material loss during isolation and purification. Experimental data indicates a substantial improvement in total yield compared to the 37% typical of older methods, with product purity reaching levels as high as 98.64%.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the process is specifically designed for industrial scalability. The use of common solvents like tetrahydrofuran and straightforward workup procedures, such as filtration and crystallization, facilitates easy scale-up from laboratory to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Higenamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity levels demonstrated in patent CN103554022A can be reliably replicated at an industrial scale. We maintain stringent purity specifications across all batches, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay against global pharmacopoeia standards.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this optimized synthetic route can enhance your supply chain resilience. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments tailored to your project needs, ensuring a seamless integration of high-quality Higenamine hydrochloride into your pharmaceutical pipeline.
