Advanced Manufacturing of Anagrelide Hydrochloride: A Technical Breakthrough for Global Supply Chains
Advanced Manufacturing of Anagrelide Hydrochloride: A Technical Breakthrough for Global Supply Chains
The pharmaceutical industry constantly seeks robust synthetic pathways that balance efficiency, safety, and cost-effectiveness, particularly for critical cardiovascular agents like Anagrelide Hydrochloride. A significant advancement in this domain is detailed in patent CN103254197A, which outlines a novel preparation method addressing the longstanding inefficiencies of traditional synthesis routes. This technical insight report analyzes the strategic implications of this new methodology for R&D directors, procurement managers, and supply chain leaders who require reliable pharmaceutical intermediate suppliers. By shifting the starting material from expensive and problematic precursors to readily available 2,3-dichlorobenzoic acid, this innovation offers a compelling solution for cost reduction in API manufacturing while simultaneously enhancing process safety and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Anagrelide Hydrochloride has been plagued by significant technical and economic hurdles that hinder efficient commercial production. As illustrated in prior art such as US4146718, traditional routes often commence with m-chloroaniline, a raw material that is not only costly but also leads to synthetic pathways characterized by excessive length and complexity. These legacy methods frequently suffer from low overall yields, often hovering around merely 8.3% to 10%, which drastically inflates the cost of goods sold and generates substantial chemical waste. Furthermore, certain established protocols rely on the use of cuprous cyanide, a highly toxic reagent that poses severe safety risks to personnel and creates complex disposal challenges for waste management teams. The accumulation of impurities in these long sequences makes downstream purification arduous, often requiring resource-intensive chromatographic techniques that are difficult to scale.
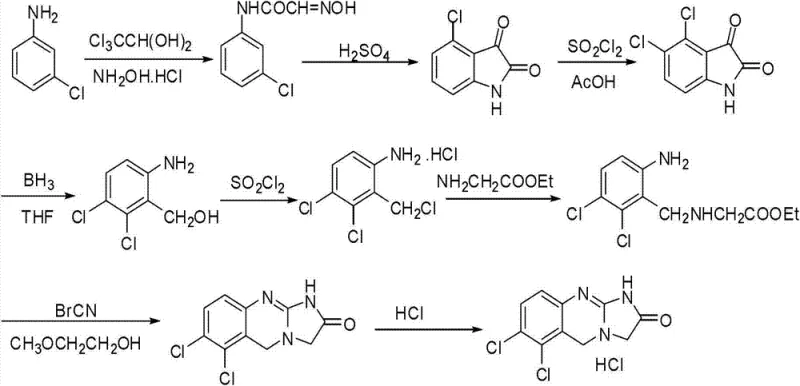
In addition to yield and safety concerns, the reliance on hazardous reagents like cyanogen bromide in early-stage intermediates further complicates the regulatory landscape for manufacturers. The presence of heavy metals or toxic byproducts necessitates stringent testing and additional purification steps to meet pharmacopeial standards, thereby extending lead times and increasing operational overhead. For supply chain heads, these factors translate into volatility; any disruption in the supply of specialized, toxic reagents can halt production entirely. Consequently, the industry has urgently required a paradigm shift towards a greener, more linear synthesis that minimizes the use of dangerous substances while maximizing atom economy and throughput.
The Novel Approach
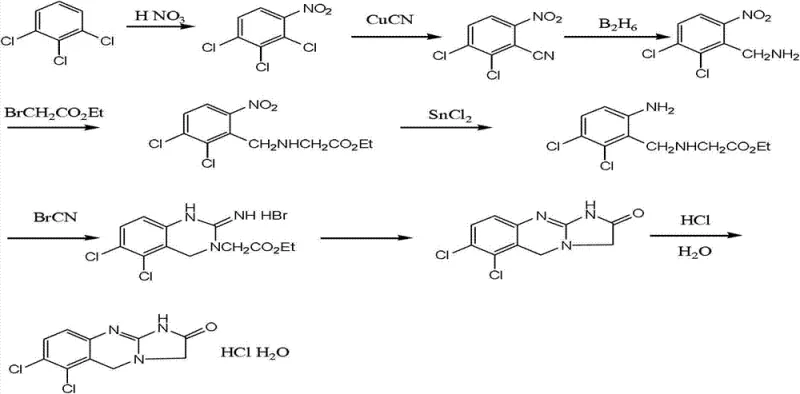
The methodology disclosed in patent CN103254197A represents a decisive break from these inefficient traditions by introducing a streamlined seven-step sequence initiated from 2,3-dichlorobenzoic acid. This starting material is inexpensive and commercially abundant, immediately addressing the raw material cost bottleneck associated with m-chloroaniline. The new route strategically employs a reduction-nitration-chlorination sequence to construct the core scaffold, followed by a precise alkylation with glycine ethyl ester and a final cyclization. Crucially, this approach eliminates the need for cuprous cyanide entirely, replacing it with safer reagents like stannous chloride for reduction and standard chlorinating agents. The result is a process that is not only chemically elegant but also operationally superior, delivering high-purity intermediates through simple recrystallization rather than complex separation technologies.
Mechanistic Insights into the Optimized Synthetic Pathway
At the heart of this improved synthesis lies a sophisticated yet practical application of reduction and cyclization chemistry that ensures high fidelity in bond formation. The initial conversion of 2,3-dichlorobenzoic acid to 2,3-dichlorobenzyl alcohol utilizes a borohydride-based reduction system enhanced by a Lewis acid and iodine in THF, operating under reflux conditions for 12 to 24 hours. This specific combination facilitates a high-yielding transformation (up to 94% in experimental trials) while maintaining excellent selectivity, preventing over-reduction or side reactions that could compromise the aromatic ring. Subsequent nitration at controlled temperatures between 20°C and 30°C ensures the regioselective introduction of the nitro group at the 6-position, a critical structural feature for the final biological activity of the molecule. The precision in these early steps sets the foundation for a clean impurity profile throughout the remainder of the synthesis.
Furthermore, the final cyclization and salt formation steps are engineered to maximize recovery and purity without resorting to extreme conditions. The reaction of the amino-intermediate with cyanogen bromide is carefully managed in an ethanol solvent system, followed by a hydrolysis and acidification sequence that precipitates the crude hydrochloride salt. The mechanism favors the formation of the imidazo-quinazolinone core through an intramolecular nucleophilic attack, driven by the thermal energy of refluxing acetonitrile and hydrochloric acid. By controlling the pH and temperature during the workup—specifically adjusting to pH 8-9 before final acidification—the process effectively segregates organic impurities from the desired product. This mechanistic control allows for the production of high-purity Anagrelide Hydrochloride with a simple impurity spectrum, reducing the burden on quality control laboratories and ensuring batch-to-batch consistency.
How to Synthesize Anagrelide Hydrochloride Efficiently
Implementing this novel synthetic route requires strict adherence to the optimized reaction parameters defined in the patent to ensure reproducibility and safety on a manufacturing scale. The process begins with the preparation of the benzyl alcohol intermediate, followed by sequential functionalization to introduce the necessary nitrogen and chlorine atoms for ring closure. Operators must pay close attention to the stoichiometry of the reducing agents and the temperature profiles during the nitration and chlorination phases to avoid exothermic runaways. While the general workflow is straightforward, the specific details regarding solvent volumes, addition rates, and crystallization conditions are critical for achieving the reported high yields. For a comprehensive, step-by-step technical guide including exact molar ratios and equipment specifications, please refer to the standardized protocol below.
- Reduction of 2,3-dichlorobenzoic acid to 2,3-dichlorobenzyl alcohol using NaBH4, Lewis Acid, and Iodine in THF.
- Nitration followed by chlorination to form 2,3-dichloro-6-nitro-benzyl chloride.
- Alkylation with glycine ethyl ester, reduction of the nitro group, and final cyclization with cyanogen bromide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers transformative benefits for procurement strategies and supply chain resilience in the pharmaceutical sector. By transitioning to a raw material base of 2,3-dichlorobenzoic acid, manufacturers can decouple their production costs from the volatile pricing of specialized aniline derivatives, leading to substantial cost savings in the long term. The elimination of cuprous cyanide not only reduces the direct cost of hazardous waste disposal but also simplifies the regulatory compliance burden, allowing for faster throughput in facilities with strict environmental mandates. This operational simplicity translates directly into enhanced reliability for downstream partners who depend on consistent delivery schedules for their own API manufacturing lines.
- Cost Reduction in Manufacturing: The economic advantage of this route is primarily driven by the substitution of expensive starting materials with commodity chemicals and the drastic improvement in step yields. Unlike legacy methods that struggle to exceed 10% overall yield, this process maintains high efficiency across multiple stages, significantly lowering the cost per kilogram of the final active ingredient. Additionally, the avoidance of expensive chromatographic purification in favor of crystallization reduces solvent consumption and processing time, further driving down the variable costs associated with production.
- Enhanced Supply Chain Reliability: Sourcing 2,3-dichlorobenzoic acid is inherently more stable than sourcing niche intermediates like m-chloroaniline or toxic metal salts, as it is a widely produced bulk chemical. This abundance mitigates the risk of supply disruptions caused by vendor capacity constraints or regulatory shutdowns of hazardous material producers. For supply chain heads, this means a more predictable lead time and the ability to secure larger inventory buffers without incurring prohibitive storage costs or safety risks, ensuring continuous availability for global markets.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial scale-up, utilizing standard unit operations such as reflux, filtration, and crystallization that are easily replicated in multi-purpose reactors. The reduction in toxic waste generation aligns with modern green chemistry principles, making it easier for manufacturing sites to maintain their environmental permits and sustainability certifications. This scalability ensures that the technology can grow with market demand, supporting the commercial scale-up of complex heterocycles without requiring massive capital investment in specialized containment infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the new synthesis route for Anagrelide Hydrochloride safer than conventional methods?
A: Conventional methods often utilize highly toxic cuprous cyanide or expensive m-chloroaniline with low yields. The novel route described in patent CN103254197A replaces these with 2,3-dichlorobenzoic acid and avoids heavy metal catalysts, significantly improving operational safety and environmental compliance.
Q: How does this process impact the purity profile of the final API intermediate?
A: The process utilizes multiple recrystallization steps for intermediates, such as toluene recrystallization for the benzyl alcohol and ethyl acetate for the benzyl chloride. This rigorous purification strategy ensures a simple impurity spectrum and low single impurity content in the final Anagrelide Hydrochloride product.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrialization. It features simple operations, avoids complex chromatographic separations, and uses readily available raw materials. The step yields are consistently high, ranging from 70% to 94% in key steps, making it economically viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Anagrelide Hydrochloride Supplier
As the global demand for cardiovascular therapeutics continues to rise, the ability to produce high-quality intermediates efficiently becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in process chemistry to bring innovative synthetic routes like the one described in CN103254197A to commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We are committed to delivering high-purity Anagrelide Hydrochloride that supports the development of life-saving medications while adhering to the highest standards of safety and quality.
We invite pharmaceutical partners to engage with us to explore how this optimized synthesis can enhance your supply chain efficiency and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced capabilities can support your long-term strategic goals in the cardiovascular therapeutic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
