Advanced Purification Technology for High-Purity Fidaxomicin Commercial Production
The pharmaceutical landscape for treating Clostridium difficile infections has been significantly advanced by the development of Fidaxomicin, a narrow-spectrum macrocyclic antibiotic. As detailed in patent CN103275152A, a groundbreaking preparation method has been established to isolate and purify this critical active pharmaceutical ingredient (API) directly from fermented mycelia with unprecedented efficiency. This technology represents a paradigm shift from traditional, labor-intensive extraction protocols to a streamlined, resin-based adsorption and polymer chromatography workflow. By integrating macroporous resin decolorization with high-resolution polymer microsphere separation, the process effectively eliminates interference from fermentation secondary metabolites while maintaining the structural integrity of the sensitive 18-membered ring system. 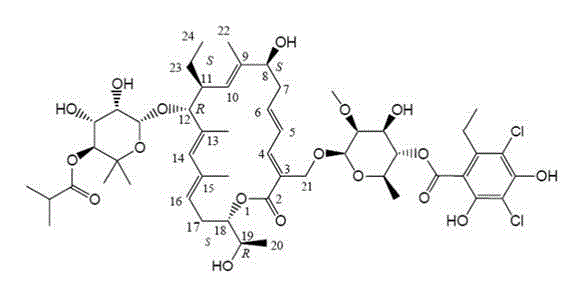 For procurement and R&D leaders, this innovation offers a reliable fidaxomicin supplier pathway that balances high purity requirements with industrial feasibility. The method ensures that the final product not only meets the rigorous quality specifications demanded by global regulatory bodies but also optimizes the overall yield, addressing the chronic supply constraints often associated with complex fermentation-derived antibiotics.
For procurement and R&D leaders, this innovation offers a reliable fidaxomicin supplier pathway that balances high purity requirements with industrial feasibility. The method ensures that the final product not only meets the rigorous quality specifications demanded by global regulatory bodies but also optimizes the overall yield, addressing the chronic supply constraints often associated with complex fermentation-derived antibiotics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of fidaxomicin has been plagued by significant technical bottlenecks that hinder cost reduction in antibiotic manufacturing. Prior art, such as the methods disclosed in CN200880009132, relied heavily on mixing macroporous resin directly with the fermentation broth, creating a nightmare for downstream processing where separating the resin from the mycelium proved exceptionally difficult and environmentally taxing. Furthermore, these legacy processes frequently employed elution systems containing acetic acid, which poses severe risks to chromatographic media; the acidic environment degrades the bonding phase of reversed-phase silica columns, drastically reducing their lifespan and utilization rate. The high boiling point of acetic acid (118°C) also complicates solvent recovery, as water and methanol evaporate readily while acetic acid remains, altering the solvent ratio and preventing effective reuse. Additionally, alternative approaches like those in CN201110104051 utilized repetitive chromatography on silica gel and gel columns, resulting in overly complicated workflows with low throughput and poor suitability for large-batch production.
The Novel Approach
The methodology outlined in CN103275152A introduces a sophisticated yet operationally simple strategy that circumvents these historical inefficiencies. Instead of co-processing resin with mycelium, this novel approach initiates with a dedicated solid-liquid extraction of the filtered mycelium using polar organic solvents, ensuring a cleaner starting material for subsequent purification stages. The process employs a dual-resin system where macroporous decolorizing resins (such as LX98 or D301) first remove pigments and bulk impurities, followed by specific adsorption on macroporous adsorptive resins (like HZ816 or D312). Crucially, the final polishing step utilizes polymer microsphere columns rather than traditional silica, allowing for gradient elution with safe, conventional solvents like methanol and ethanol. This eliminates the need for corrosive acids and enables the production of high-purity fidaxomicin fine powder with a purity greater than 95%, all while maintaining a total recovery rate that significantly outperforms previous iterations.
Mechanistic Insights into Macroporous Resin Adsorption and Polymer Chromatography
The core of this purification success lies in the precise manipulation of polarity and adsorption kinetics inherent to the fidaxomicin molecule. The initial extraction leverages the solubility profile of the macrocyclic lactone in polar organic solvents such as ethanol or methanol, typically at concentrations between 80% and 95%, to efficiently pull the API from the cellular matrix of the Actinoplanes bacterial strain. Once in solution, the liquid is diluted to reduce solvent strength, facilitating the selective adsorption of impurities onto the decolorizing resin. This step is critical for removing fermentation byproducts that share similar hydrophobic characteristics but differ in polarity or molecular size. The subsequent adsorption onto specific macroporous resins capitalizes on the hydrogen bonding and van der Waals forces between the resin matrix and the fidaxomicin molecule, effectively concentrating the API while washing away non-binding contaminants.
Impurity control is further refined during the gradient elution and final polymer chromatography stages. By utilizing a stepwise increase in alcohol concentration (e.g., from 40% to 80%), the process selectively desorbs fidaxomicin from the resin while leaving behind more tightly bound or more loosely bound impurities. The final injection into polymer microsphere columns (such as PS30-300) provides a high-resolution separation mechanism based on subtle differences in hydrophobicity and steric hindrance. Unlike silica-based media, these polymer microspheres are chemically inert across a wide pH range, ensuring that the acid-sensitive ester linkages within the fidaxomicin structure remain intact. This mechanistic precision ensures that the final crystalline product is free from the secondary metabolites that often compromise the efficacy and safety profile of fermentation-derived antibiotics, delivering a consistent and high-quality API suitable for formulation.
How to Synthesize Fidaxomicin Efficiently
Implementing this purification protocol requires strict adherence to the optimized parameters regarding solvent ratios, flow rates, and resin loading capacities to ensure reproducibility at scale. The process begins with the fermentation of the Actinoplanes strain, followed by filtration to isolate the mycelium, which is then subjected to solvent extraction. The resulting extract undergoes a sequential treatment through decolorization and adsorption columns before the final chromatographic polishing. For R&D teams looking to replicate or adapt this workflow, the detailed standardized synthesis steps provided below outline the specific conditions for resin selection, gradient elution profiles, and solvent recovery necessary to achieve the reported purity benchmarks.
- Extract fidaxomicin from filtered fermentation mycelium using polar organic solvents like methanol or ethanol, followed by solid-liquid separation.
- Pass the diluted extract through macroporous decolorizing resin (e.g., LX98) to remove pigments, then adsorb onto macroporous adsorptive resin (e.g., HZ816).
- Elute the adsorbed resin with gradient alcohol solutions, concentrate the eluate, and perform final polishing using polymer microsphere columns to obtain fine powder.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers substantial strategic benefits for organizations managing the supply of complex antibiotic intermediates. By transitioning away from corrosive acid-based elution systems and fragile silica columns, manufacturers can drastically simplify their operational infrastructure and reduce the frequency of equipment replacement. The reliance on conventional, low-toxicity solvents such as methanol, ethanol, and ethyl acetate ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with volatile specialty chemical markets. Furthermore, the elimination of acetic acid not only protects capital equipment but also streamlines the solvent recovery process, as the azeotropic issues associated with acid-water mixtures are completely avoided. This leads to a more sustainable manufacturing cycle with reduced waste generation and lower energy consumption during distillation.
- Cost Reduction in Manufacturing: The removal of acetic acid from the elution system serves as a primary driver for cost optimization, as it prevents the degradation of expensive chromatography columns and extends their operational lifecycle significantly. Additionally, the use of standard alcohols allows for highly efficient solvent recycling loops, minimizing the volume of fresh solvent required per batch and reducing hazardous waste disposal costs. The simplified workflow, which avoids repetitive multi-column chromatography, also reduces labor hours and processing time, contributing to a leaner production model that enhances overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: By utilizing widely available macroporous resins and polymer microspheres rather than specialized, single-source reagents, the supply chain becomes more resilient to disruptions. The robustness of the polymer columns against pH variations means that minor fluctuations in process conditions are less likely to result in batch failures, ensuring a consistent output of high-purity material. This reliability is crucial for maintaining continuous supply to downstream formulation partners, particularly in the context of essential antibiotics where inventory shortages can have significant clinical implications.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, avoiding the bottlenecks of preparative thin-layer chromatography which is limited to small batch sizes. The use of non-halogenated, low-toxicity solvents aligns with increasingly stringent environmental regulations regarding VOC emissions and wastewater treatment. This green chemistry approach not only facilitates easier regulatory approval in key markets but also future-proofs the manufacturing site against tightening environmental compliance standards, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity fidaxomicin preparation method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this technology resolves specific pain points in antibiotic purification. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing the quality credentials of potential suppliers.
Q: How does this method improve upon traditional acetic acid-based purification?
A: Traditional methods often utilize acetic acid in elution systems, which can degrade reversed-phase silica columns due to its corrosive nature and high boiling point (118°C), making solvent recovery difficult. This novel protocol eliminates acetic acid entirely, using conventional alcohols instead, which preserves column integrity and simplifies solvent recycling.
Q: What purity levels can be achieved with this polymer microsphere technique?
A: By utilizing specific polymer microsphere columns (such as PS30-300) for the final chromatographic separation, the process effectively removes fermentation secondary metabolites. This results in a final fidaxomicin fine powder with a purity exceeding 95%, meeting stringent pharmaceutical standards.
Q: Is this purification process suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability. It avoids complex multi-column repetitive chromatography and expensive preparative thin-layer chromatography. Instead, it relies on robust macroporous resins and standard solvent systems (methanol/ethanol/ethyl acetate), which are cost-effective and easy to handle in large-volume reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fidaxomicin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable supply of high-quality antibiotics for the global healthcare market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of multinational pharmaceutical partners. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch of fidaxomicin meets the >95% purity threshold essential for therapeutic efficacy. Our capability to implement complex resin-based purification technologies allows us to deliver a product that is not only chemically pure but also free from the residual solvents and impurities that can complicate downstream drug formulation.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall cost of goods. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our fidaxomicin supply aligns perfectly with your development timelines and quality expectations.
