Advanced Benfotiamine Manufacturing: Scalable High-Purity Synthesis for Global Supply Chains
The pharmaceutical and nutritional industries are constantly seeking robust manufacturing pathways for high-value derivatives like benfotiamine, a lipophilic prodrug of vitamin B1 known for superior bioavailability. Patent CN103772432A introduces a transformative production method that leverages polyphosphoric acid as a phosphorylating agent, addressing critical inefficiencies found in legacy synthetic routes. This innovation enables the direct reaction with vitamin B1 followed by high-temperature hydrolysis and a sophisticated extraction process using trioctylamine to remove phosphoric acid residues. The resulting crude phosphate monoester is processed without intermediate purification, reacting directly with benzoyl chloride under controlled pH conditions to yield a white solid with exceptional purity. This technical breakthrough represents a significant leap forward for a reliable pharmaceutical intermediate supplier, offering a pathway that is not only chemically efficient but also inherently designed for industrial scalability and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benfotiamine has been plagued by cumbersome and energy-intensive processes that rely on pyrophosphoric acid or phosphorus pentoxide as key reagents. Traditional methods often necessitate heating phosphoric acid to extreme temperatures around 270°C to generate the active pyrophosphoric species, which places immense thermal stress on production equipment and escalates energy costs significantly. Furthermore, these legacy routes frequently require the reaction mixture to stand at room temperature for approximately one week to facilitate crystallization, creating a severe bottleneck in production throughput and extending lead times unnecessarily. The reliance on alkali metal salts for neutralization in older processes generates substantial quantities of solid phosphate by-products that are extremely difficult to separate, leading to product entrapment and significant yield losses. Additionally, the use of highly corrosive reagents like phosphorus oxychloride introduces safety hazards and complicates hydrolysis operations, making the entire workflow unsuitable for modern, large-scale commercial manufacturing environments.
The Novel Approach
The novel approach detailed in the patent data circumvents these historical challenges by utilizing polyphosphoric acid, which reacts efficiently with vitamin B1 at moderate temperatures between 80°C and 130°C without the need for pre-heating to form pyrophosphoric acid. This method streamlines the workflow by eliminating the week-long standing period, replacing it with a controlled hydrolysis step that completes within hours, thereby drastically accelerating the production cycle. The integration of a trioctylamine extraction step allows for the precise removal of excess phosphoric acid to concentrations below 0.1%, avoiding the need for ion exchange columns that often limit output and increase operational complexity. By precipitating the crude vitamin B1 phosphate monoester directly and proceeding to benzoylation without intermediate purification, the process minimizes unit operations and reduces the potential for material loss. This streamlined methodology ensures high product purity and yield while simplifying the overall operational requirements, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Polyphosphoric Acid-Catalyzed Phosphorylation
The core of this synthetic innovation lies in the mechanistic efficiency of using polyphosphoric acid as a phosphorylating agent, which facilitates the formation of the vitamin B1 phosphate monoester under relatively mild thermal conditions. Unlike pyrophosphoric acid, which requires rigorous dehydration conditions to form, polyphosphoric acid provides a ready source of phosphate groups that react with the hydroxyl functionality of vitamin B1 to form the ester linkage. The subsequent hydrolysis step at 80-120°C ensures the cleavage of any pyrophosphate bonds that may have formed, selectively yielding the desired monoester species while maintaining the integrity of the thiazole ring structure. This selectivity is crucial for maintaining the biological activity of the final benfotiamine product, as harsh conditions could otherwise lead to degradation of the sensitive vitamin B1 backbone. The reaction kinetics are optimized by the weight ratio of vitamin B1 to polyphosphoric acid, typically maintained between 1:3 and 1:5, ensuring complete conversion while minimizing side reactions.
Following the phosphorylation and hydrolysis, the purification mechanism relies on a liquid-liquid extraction strategy using trioctylamine dissolved in an organic solvent to sequester residual phosphoric acid from the aqueous phase. This extraction process is highly specific, reducing the phosphoric acid concentration to negligible levels without the need for solid-phase adsorbents that can trap the product. The crude monoester is then precipitated by adding a water-miscible organic solvent, allowing for the isolation of the intermediate without rigorous recrystallization steps that often compromise yield. The final benzoylation step is conducted under alkaline conditions (pH 8.0-14.0) at low temperatures (0-5°C) to control the reactivity of benzoyl chloride and prevent over-acylation or hydrolysis of the ester bond. The final product is precipitated by adjusting the pH to 3.5-4.0, ensuring the isolation of benfotiamine as a high-purity white solid suitable for downstream pharmaceutical applications.
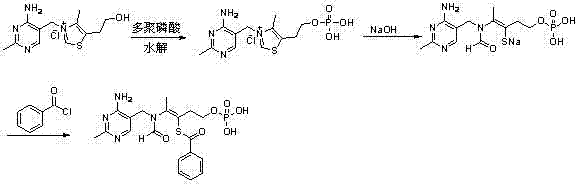
How to Synthesize Benfotiamine Efficiently
The synthesis of benfotiamine via this patented route involves a sequence of carefully controlled reaction and isolation steps designed to maximize yield and purity while minimizing waste. The process begins with the phosphorylation of vitamin B1 using polyphosphoric acid, followed by hydrolysis and extraction to isolate the crude phosphate monoester. This intermediate is then subjected to benzoylation under alkaline conditions to form the final lipophilic derivative. The detailed standardized synthesis steps see the guide below, which outlines the specific temperature ranges, reaction times, and pH adjustments required to replicate the high success rates reported in the patent data. Adhering to these parameters is essential for achieving the reported purity levels of over 98.5% and yields exceeding 77%.
- React Vitamin B1 with polyphosphoric acid at 80-130°C for 3-8 hours, followed by hydrolysis at 80-120°C.
- Extract phosphoric acid using a trioctylamine and organic solvent mixture to reduce phosphate concentration below 0.1%.
- Precipitate crude monoester, adjust pH to 8.0-14.0, react with benzoyl chloride, and finalize precipitation at pH 3.5-4.0.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers substantial strategic advantages regarding cost stability and operational reliability. The elimination of high-temperature pyrophosphoric acid generation removes a significant energy burden from the manufacturing process, leading to reduced utility costs and lower wear on reactor vessels. The ability to recover and reuse solvents from the extraction and precipitation steps further contributes to substantial cost savings by minimizing raw material consumption and waste disposal fees. Moreover, the conversion of solid by-products into sellable chemical fertilizers transforms a waste stream into a potential revenue source, enhancing the overall economic viability of the production line. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive ion exchange columns and reduces the consumption of high-purity reagents by utilizing polyphosphoric acid directly. By avoiding the generation of difficult-to-separate solid phosphate salts, the method minimizes product loss during purification, leading to higher overall yields and lower cost per kilogram of active ingredient. The simplified workflow reduces labor hours and equipment maintenance requirements, contributing to a more lean and efficient manufacturing operation that drives down the total cost of ownership for the production facility.
- Enhanced Supply Chain Reliability: The use of readily available reagents like polyphosphoric acid and trioctylamine ensures that the supply chain is not dependent on specialized or hard-to-source chemicals that could cause production delays. The shortened reaction and crystallization times significantly reduce the manufacturing cycle, allowing for faster turnaround on orders and improved responsiveness to market demand. This agility is critical for maintaining continuous supply to downstream pharmaceutical manufacturers who require just-in-time delivery of high-quality intermediates to meet their own production schedules.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing standard unit operations like extraction and precipitation that are easily transferred from pilot to commercial scale. The ability to recycle solvents and repurpose solid waste as fertilizer aligns with stringent environmental regulations, reducing the regulatory burden and potential fines associated with hazardous waste disposal. This environmental stewardship not only protects the company from compliance risks but also enhances its reputation as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the benfotiamine production method, based on the specific advantages and operational details outlined in the patent documentation. These answers provide clarity on why this route is superior to conventional methods and how it benefits both technical teams and business stakeholders. Understanding these details is crucial for making informed decisions about sourcing and manufacturing partnerships.
Q: Why is polyphosphoric acid preferred over pyrophosphoric acid for benfotiamine synthesis?
A: Polyphosphoric acid eliminates the need for high-temperature heating to 270°C required to form pyrophosphoric acid, significantly reducing energy consumption and equipment stress while simplifying the operational workflow.
Q: How does the trioctylamine extraction method improve product purity?
A: Using trioctylamine effectively removes excess phosphoric acid from the system without requiring ion exchange columns, preventing product loss and avoiding the generation of difficult-to-separate solid phosphate salts.
Q: What are the environmental benefits of this production method?
A: The process allows for solvent recovery and reuse, and the solid by-products separated during filtration can be repurposed as chemical fertilizers, drastically reducing solid waste discharge and environmental pollution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benfotiamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, leveraging advanced synthetic routes like the polyphosphoric acid method to deliver exceptional value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet the rigorous demands of the pharmaceutical industry with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of benfotiamine meets the highest standards of quality and safety required for human consumption. Our commitment to technical excellence allows us to navigate complex chemical landscapes and deliver solutions that drive your business forward.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can optimize your supply chain and reduce your overall production costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of partnering with us for your benfotiamine requirements. We encourage you to reach out for specific COA data and route feasibility assessments to verify our capacity to support your long-term strategic goals. Let us collaborate to build a more efficient and reliable supply network for your critical pharmaceutical intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
