Advanced Manufacturing of Triterpene Derivatives for Commercial Scale-up and High Purity
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex active pharmaceutical ingredients, particularly those targeting cardiovascular conditions. Patent CN1310940C introduces a significant advancement in the preparation of triterpene derivatives, specifically compound (V), which functions as a potent endothelin receptor antagonist. This chemical class is critical for treating circulation system diseases, hypertension, and cerebral circulation imbalances. The disclosed technology represents a paradigm shift from traditional multi-step syntheses towards a more integrated, efficient approach. By leveraging specific lithium reagents in a controlled organic solvent environment, the process achieves high conversion rates while minimizing the environmental footprint associated with heavy metal catalysts. For stakeholders in the fine chemical sector, this patent data underscores a viable route for producing high-purity pharmaceutical intermediates that meet stringent regulatory standards. The technical nuances of this method suggest a strong potential for reliable triterpene derivative supplier partnerships focused on quality and consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oleanolic acid derivatives and related triterpene structures has been plagued by inefficient processing steps that hinder commercial viability. Prior art, including various patent documents and non-patent literature, often describes methods requiring the isolation of unstable intermediates, which significantly increases processing time and material loss. Furthermore, conventional routes frequently rely on environmentally harmful reagents such as titanium trichloride for reduction steps, creating substantial waste disposal challenges and safety hazards in a manufacturing plant. The need for different reagents and solvents in each reaction step complicates the supply chain and increases the overall cost reduction in pharmaceutical intermediates manufacturing. Additionally, traditional methods often struggle with impurity profiles, necessitating complex purification techniques like column chromatography that are difficult to scale. These factors collectively create bottlenecks that limit the ability to achieve commercial scale-up of complex triterpene derivatives efficiently.
The Novel Approach
The methodology outlined in CN1310940C addresses these historical pain points through a streamlined, one-pot reaction strategy that eliminates the need for intermediate isolation. By utilizing lithium reagents such as lithium chloride and lithium hydroxide in solvents like N,N-dimethylformamide, the process facilitates a smooth transition from starting materials to the final product. This approach not only simplifies the operational workflow but also enhances the overall yield by preventing material degradation during transfer and purification stages. The ability to conduct multiple reaction steps in a single vessel reduces solvent consumption and energy usage, aligning with modern green chemistry principles. Moreover, the method allows for the direct use of crude solutions in subsequent steps, which drastically simplifies the production timeline. This innovation provides a clear pathway for reducing lead time for high-purity pharmaceutical intermediates, offering a competitive edge in the global supply market.
Mechanistic Insights into Lithium-Mediated Cyclization and Deprotection
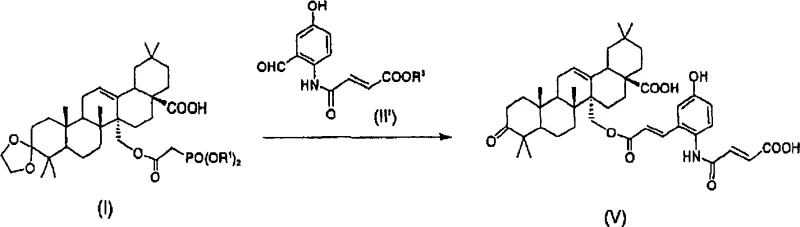
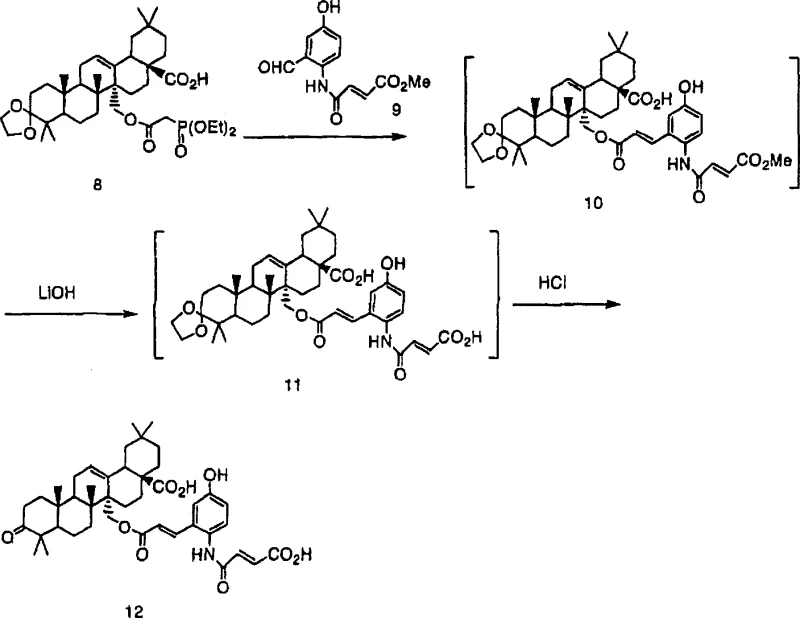
The core of this technological breakthrough lies in the precise manipulation of lithium species to drive the Horner-Emmons reaction and subsequent deprotection without isolating sensitive intermediates. In the first stage, compound (I) reacts with compound (II) in the presence of a lithium reagent to form a solution containing compound (III). The lithium cation likely stabilizes the enolate or transition state, facilitating the coupling reaction under mild conditions ranging from -20°C to 60°C. Following this, the addition of a second lithium reagent, typically lithium hydroxide, promotes the conversion to compound (IV). This step is critical as it prepares the molecule for the final acid-catalyzed deprotection. The use of lithium hydroxide is particularly advantageous due to its solubility profile and reactivity in polar aprotic solvents. The final addition of acid, such as hydrochloric acid, cleaves the protecting groups to yield the target compound (V). This mechanistic sequence ensures that the reaction proceeds with high specificity, minimizing the formation of by-products that could compromise the purity of the final API intermediate.
Impurity control is another vital aspect of this mechanism, particularly concerning the removal of endotoxins which is crucial for injectable formulations. The patent details a specific purification protocol where the final organic solution is washed with purified water. This step exploits the solubility differences between the triterpene derivative and endotoxin contaminants, effectively transferring the impurities into the aqueous phase. Repeating this washing process can reduce endotoxin levels to less than 0.5 EU/ml, meeting rigorous pharmacopeial standards. This mechanism highlights the process's suitability for producing high-purity endothelin receptor antagonist precursors. By integrating purification directly into the workup phase, the method avoids the need for additional chromatographic steps that often introduce variability. This level of control over the impurity profile is essential for R&D directors focusing on the safety and efficacy of the final drug product.
How to Synthesize Triterpene Derivative Efficiently
Implementing this synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize efficiency and yield. The process begins with the dissolution of the starting triterpene material in a suitable organic solvent, followed by the sequential addition of lithium reagents and the coupling partner. It is imperative to maintain the reaction mixture within the specified temperature ranges to prevent side reactions that could lower the overall yield. The patent emphasizes the importance of not isolating the intermediate compounds, as this preserves the integrity of the reactive species and streamlines the workflow. Detailed standardized synthesis steps are essential for reproducibility, especially when scaling from laboratory to pilot plant operations. The following guide outlines the critical operational parameters derived from the patent data to ensure successful implementation.
- Dissolve compound (I) and compound (II) in an organic solvent such as DMF in the presence of a lithium reagent like lithium chloride to form compound (III) solution.
- Add a second lithium reagent, preferably lithium hydroxide, to the reaction mixture to convert compound (III) into compound (IV) without isolation.
- Add an acid such as hydrochloric acid to the solution to deprotect and finalize the formation of compound (V), followed by extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this manufacturing process offers substantial strategic benefits that extend beyond simple technical feasibility. The elimination of hazardous heavy metal catalysts like titanium trichloride significantly reduces the regulatory burden and waste management costs associated with production. This shift not only enhances the environmental compliance profile of the manufacturing site but also mitigates the risk of supply disruptions caused by strict environmental regulations on hazardous chemicals. Furthermore, the use of readily available lithium reagents and common organic solvents ensures a stable supply chain, reducing the dependency on specialized or scarce materials. The one-pot nature of the synthesis reduces the number of unit operations, which translates to lower labor costs and reduced equipment occupancy time. These factors collectively contribute to a more resilient and cost-effective supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of the reaction sequence and the elimination of intermediate isolation steps. By avoiding the need for chromatography and reducing solvent usage, the overall material and operational costs are significantly lowered. The use of lithium reagents, which are generally more cost-effective and easier to handle than traditional transition metal catalysts, further contributes to economic efficiency. Additionally, the high yields reported in the patent examples suggest that raw material utilization is maximized, reducing waste and improving the overall cost structure. This logical deduction of cost savings makes the process highly attractive for large-scale commercial production where margin optimization is critical.
- Enhanced Supply Chain Reliability: The reliance on common chemical reagents such as lithium hydroxide and N,N-dimethylformamide ensures that the supply chain is robust and less susceptible to market volatility. Unlike specialized catalysts that may have limited suppliers, these materials are widely available from multiple global sources, reducing the risk of procurement bottlenecks. The simplified process flow also means that production can be ramped up quickly to meet demand surges without requiring complex retooling or extensive operator training. This reliability is crucial for maintaining continuous supply to downstream pharmaceutical manufacturers who depend on timely delivery of high-quality intermediates to meet their own production schedules.
- Scalability and Environmental Compliance: The design of this synthesis route inherently supports scalability, as it avoids unit operations that are difficult to scale, such as flash chromatography. The crystallization-based purification method is well-suited for large-scale industrial equipment, ensuring consistent product quality across different batch sizes. Moreover, the reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, future-proofing the manufacturing process against potential regulatory changes. This compliance reduces the risk of fines or shutdowns and enhances the corporate sustainability profile, which is becoming a key factor in supplier selection for major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triterpene derivative synthesis process. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific production needs. The information covers aspects ranging from purification efficiency to reagent selection, providing a comprehensive overview of the process capabilities.
Q: How does this process handle endotoxin removal for injectable grades?
A: The patent describes a specific purification step where the final compound is dissolved in an organic solvent like ethyl acetate and washed repeatedly with purified water. This effectively transfers endotoxins to the aqueous layer, reducing content to negligible levels suitable for injectable applications.
Q: What are the advantages of using lithium reagents over traditional titanium reagents?
A: Traditional methods often rely on environmentally harmful titanium trichloride. This novel process utilizes lithium chloride and lithium hydroxide, which are easier to handle, generate less hazardous waste, and allow for a one-pot synthesis that eliminates the need for intermediate isolation steps.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method is designed for commercial scale-up. It uses common organic solvents like DMF and ethyl acetate, operates at moderate temperatures, and avoids complex chromatography purification by utilizing crystallization, making it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triterpene Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into reliable commercial supply chains. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex syntheses like the one described in CN1310940C can be executed with precision and consistency. We are committed to meeting stringent purity specifications through our rigorous QC labs, which are equipped to handle the specific analytical requirements of triterpene derivatives and endothelin receptor antagonists. Our infrastructure is designed to support the unique demands of fine chemical manufacturing, providing a secure foundation for long-term partnerships.
We invite pharmaceutical and chemical companies to collaborate with us to optimize their supply chains for these critical intermediates. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can ensure a stable, high-quality supply of triterpene derivatives that supports your drug development and commercialization goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
