Advanced Catalytic Strategy for Scalable 3-Alkoxy-4-Quinolinone Pharmaceutical Intermediates
Advanced Catalytic Strategy for Scalable 3-Alkoxy-4-Quinolinone Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes for heterocyclic scaffolds that possess potent biological activities, particularly in the realms of oncology and antiviral therapy. Patent CN111423370B introduces a groundbreaking preparation method for 3-alkoxy-4-quinolinone derivatives, a core skeleton found in numerous natural products such as Waltherione C and Leiokinene A. This technology addresses the critical need for efficient access to these pharmacophores, which exhibit significant anti-tumor, anti-HIV, and antifungal properties. By leveraging a novel catalytic cyclization strategy, this invention transforms the synthesis landscape from cumbersome multi-step processes into a streamlined, one-pot operation. For R&D directors and procurement specialists, this represents a pivotal shift towards more sustainable and cost-effective manufacturing of high-value pharmaceutical intermediates. The ability to generate diverse structural analogues through simple substrate variation offers unparalleled flexibility for drug discovery programs aiming to optimize potency and metabolic stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 4-quinolinone core has been fraught with synthetic challenges that hinder large-scale production and rapid analogue generation. Traditional methodologies often rely on harsh reaction conditions, involving highly acidic media and elevated temperatures that can degrade sensitive functional groups and lead to poor atom economy. Furthermore, classical routes frequently necessitate multiple synthetic steps, requiring the isolation and purification of unstable intermediates, which drastically increases processing time and waste generation. A significant drawback in prior art is the frequent formation of regioisomeric mixtures, creating a nightmare for downstream purification and compromising the overall yield of the desired active pharmaceutical ingredient (API) precursor. These inefficiencies not only inflate the cost of goods sold (COGS) but also introduce supply chain vulnerabilities due to the reliance on specialized reagents and complex operational protocols that are difficult to scale safely in a GMP environment.
The Novel Approach
In stark contrast, the methodology disclosed in CN111423370B utilizes a transition metal-catalyzed annulation of 2-azidoaryl alkynyl ketones with alcohol compounds to directly furnish the target 3-alkoxy-4-quinolinone structure. This innovative approach operates under remarkably mild conditions, typically ranging from 0°C to 80°C, and often proceeds efficiently at room temperature, thereby minimizing energy consumption and thermal stress on the molecules. The reaction exhibits exceptional atom economy and high selectivity, effectively suppressing the formation of unwanted regioisomers and simplifying the purification process to standard column chromatography or crystallization. As illustrated in the general reaction scheme below, the transformation is direct and versatile, accommodating a wide array of substituents on the aromatic ring and the alkyne terminus.
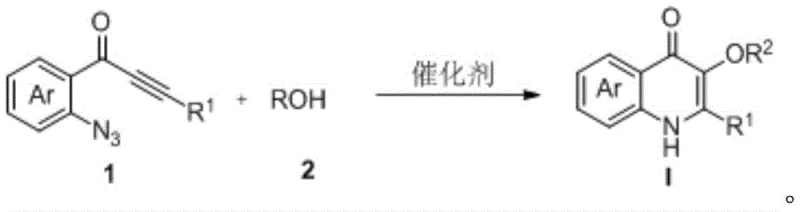
This streamlined process eliminates the need for pre-functionalized halogenated intermediates often required in cross-coupling strategies, thereby reducing the number of unit operations and the associated solvent usage. For supply chain managers, this translates to a significantly simplified logistics profile, as the starting materials—alkynes and common alcohols—are commercially abundant and stable. The robustness of this catalytic system ensures consistent batch-to-batch quality, a critical parameter for maintaining regulatory compliance in the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Gold-Catalyzed Cyclization
The success of this synthetic route hinges on the precise activation of the alkyne moiety by soft Lewis acid catalysts, such as gold(I) or gold(III) complexes, although silver and copper salts have also proven effective. The mechanism likely initiates with the coordination of the metal catalyst to the triple bond of the alkynyl ketone, increasing its electrophilicity and facilitating a nucleophilic attack by the nitrogen of the adjacent azide group or potentially an intramolecular cyclization followed by alcohol trapping. This activation lowers the energy barrier for the ring-closing step, allowing the reaction to proceed rapidly under ambient conditions. The subsequent rearrangement and elimination of nitrogen gas drive the reaction forward, resulting in the formation of the stable quinolinone core with the alkoxy group installed at the 3-position with high regiocontrol. Understanding this mechanistic pathway is vital for process chemists aiming to further optimize catalyst loading or explore alternative ligand systems to enhance turnover numbers.
From an impurity control perspective, the high selectivity of this catalytic cycle is paramount. The specific electronic and steric environment created by the catalyst-substrate complex ensures that the alcohol nucleophile attacks the correct position on the intermediate vinyl-metal species, preventing the formation of constitutional isomers that are notoriously difficult to separate. The patent data demonstrates that even with sterically demanding substrates, such as those bearing naphthyl groups or bulky alkyl chains, the reaction maintains high fidelity. The structural diversity achievable through this method is extensive, as evidenced by the successful synthesis of various derivatives shown below, where R1 can be methyl, phenyl, or naphthyl, and R2 can range from simple methyl to complex chiral terpenes.
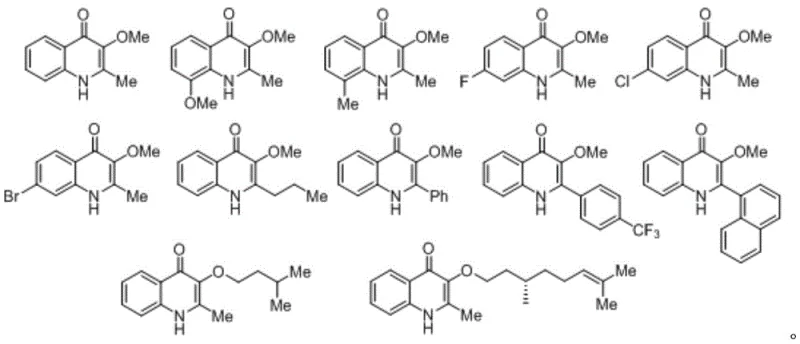
This level of control over the impurity profile is essential for meeting the stringent specifications required for clinical trial materials. By minimizing side reactions, the process reduces the burden on analytical teams and ensures that the final product meets the rigorous purity standards demanded by global health authorities. The ability to tolerate various functional groups, including halogens and electron-donating methoxy groups on the aromatic ring, further underscores the versatility of this mechanism for generating focused libraries for structure-activity relationship (SAR) studies.
How to Synthesize 3-Alkoxy-4-Quinolinone Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The procedure typically involves mixing the 2-azidoaryl alkynyl ketone substrate with an excess of the chosen alcohol, which serves as both the reactant and the solvent, in the presence of a catalytic amount of the metal salt. The reaction progress is monitored via TLC or HPLC, and upon completion, the workup is straightforward, involving solvent removal and purification. This simplicity makes it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates, as it avoids the need for specialized high-pressure equipment or cryogenic conditions. Detailed standardized operating procedures for specific analogues can be derived directly from the experimental examples provided in the patent documentation.
- Charge a reaction vessel with gold chloride catalyst (AuCl, 0.01 mmol), the alkyne substrate 1a (0.2 mmol), and methanol (1.0 mL) as both reactant and solvent.
- Stir the reaction mixture at room temperature for approximately 8 hours to allow for complete cyclization and alkoxylation.
- Concentrate the mixture under reduced pressure to remove solvent and purify the crude residue via column chromatography using methanol/dichloromethane to obtain the pure yellow solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain; by utilizing readily available alkynes and commodity alcohols, manufacturers can mitigate the risks associated with sourcing exotic or custom-synthesized building blocks. This reliance on bulk chemicals ensures a stable supply base and protects against market volatility, which is crucial for maintaining continuous production schedules for long-term commercial contracts. Furthermore, the reduction in synthetic steps directly correlates to a reduction in overall manufacturing lead time, allowing companies to respond more agilely to market demands or urgent clinical supply needs without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive reagents and the minimization of waste disposal costs. By avoiding harsh acidic conditions and multi-step sequences, the process significantly lowers the consumption of solvents and energy, leading to substantial operational expenditure savings. The use of low-loading catalysts, particularly when recoverable or inexpensive metals like copper are employed, further drives down the raw material costs per kilogram of product. Additionally, the high yields reported in the patent examples mean that less starting material is required to produce the same amount of API intermediate, optimizing the overall material balance and improving the gross margin potential for the final drug product.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply chain resilience by reducing the likelihood of batch failures due to sensitive operational parameters. Since the reaction tolerates a wide range of temperatures and does not require strictly anhydrous or oxygen-free environments in many cases, it is more forgiving in large-scale reactors where perfect control is harder to maintain than in the lab. This reliability ensures consistent delivery timelines to downstream customers, fostering stronger partnerships and trust. The ability to source diverse alcohol partners also allows for rapid adaptation if a specific supply line is disrupted, as alternative alcohols can often be substituted with minimal process re-optimization.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method aligns perfectly with green chemistry principles. The high atom economy means that a greater proportion of the reactant mass ends up in the final product, reducing the volume of hazardous waste generated. The avoidance of strong mineral acids and the use of milder catalysts lower the corrosivity of the process streams, extending equipment life and reducing maintenance costs. These factors collectively simplify the regulatory approval process for new manufacturing sites and support corporate sustainability goals, making the technology attractive for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What catalysts are suitable for this quinolinone synthesis?
A: The patent discloses a range of effective catalysts including gold salts (AuCl, AuCl3), silver salts (AgSbF6), copper salts (Cu(OTf)2), and even organic acids like TsOH, providing flexibility for cost optimization.
Q: Can this method accommodate diverse alcohol substrates?
A: Yes, the protocol demonstrates high tolerance for various alcohols, ranging from simple methanol to complex chiral alcohols like (S)-(-)-beta-citronellol, enabling the synthesis of diverse ether derivatives.
Q: What are the typical reaction conditions required?
A: The reaction proceeds under mild conditions, typically between 0°C and 80°C, often at room temperature, which significantly reduces energy consumption compared to traditional high-temperature cyclizations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Alkoxy-4-Quinolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic cyclization technology for the development of next-generation anticancer and antiviral therapeutics. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology from bench-scale discovery to full commercial production. Our facilities are equipped to handle diverse synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, supported by our state-of-the-art rigorous QC labs which ensure every batch is fully characterized and compliant with international standards.
We invite pharmaceutical innovators and procurement leaders to collaborate with us to leverage this efficient synthesis route for your pipeline projects. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules, ensuring a seamless path from concept to commercial reality.
