Advanced Synthesis of Apixaban Intermediates for Commercial Scale-Up and Cost Reduction
Advanced Synthesis of Apixaban Intermediates for Commercial Scale-Up and Cost Reduction
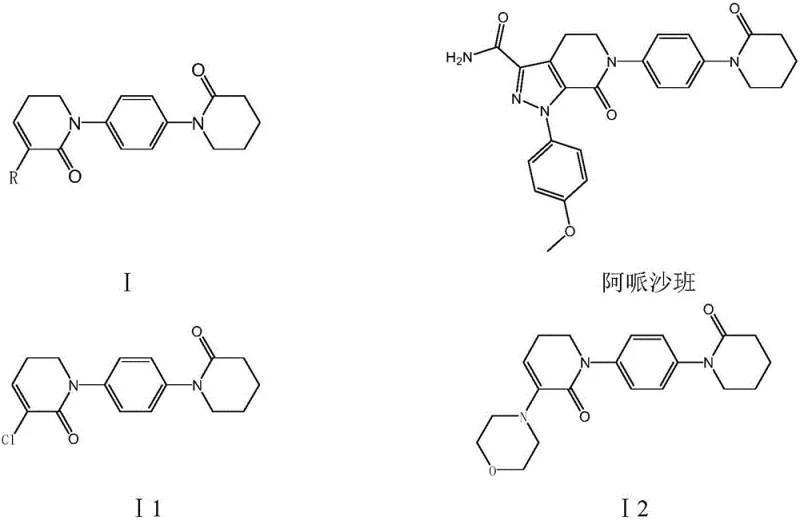
The pharmaceutical industry continuously seeks robust and scalable pathways for the production of critical anticoagulant medications, with Apixaban standing out as a premier direct factor Xa inhibitor. Patent CN111170992A introduces a groundbreaking preparation method for 5,6-dihydropyridine-2(1H)-ketone derivatives, which serve as the pivotal building blocks for this life-saving drug. This technical disclosure addresses the longstanding challenges associated with the synthesis of intermediates I1 and I2, offering a route that prioritizes both economic efficiency and environmental sustainability. By leveraging p-acetamino aniline and delta-valerolactone as primary raw materials, the process circumvents the reliance on costly and unstable iodo-substances that have historically plagued the supply chain. The strategic design of this synthesis ensures high reaction selectivity and exceptional product purity, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming to secure long-term production stability. The following analysis delves into the mechanistic superiority and commercial viability of this novel approach.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
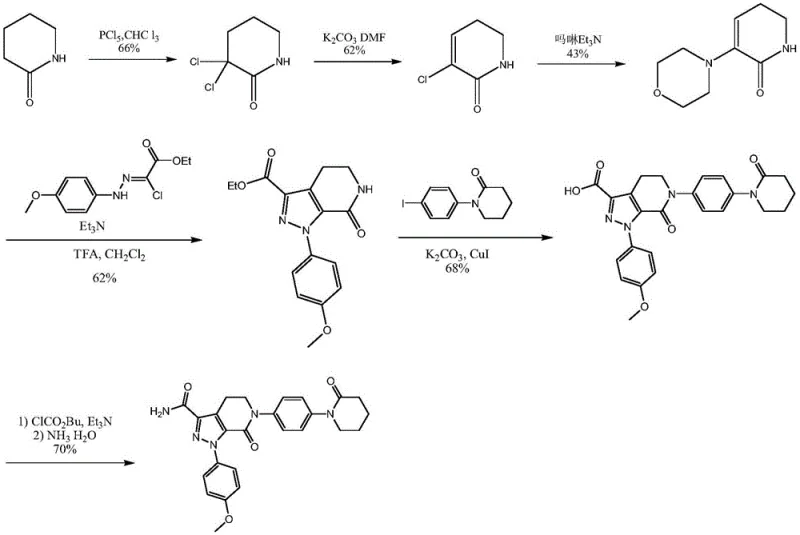
Historically, the manufacturing of Apixaban intermediates has been constrained by synthetic routes that are both economically burdensome and environmentally hazardous. Prior art documents, such as WO2003049681 and WO2004083177, describe pathways that rely heavily on 1-(4-iodophenyl)-2-piperidone, a raw material characterized by its exorbitant price and poor stability during storage and transport. Furthermore, the chlorination steps in these conventional methods often utilize phosphorus pentachloride, a reagent known for generating substantial amounts of phosphorus-containing wastewater that requires complex and costly treatment protocols. The total yield in these legacy routes is frequently reported to be as low as 5.2 percent, rendering them inefficient for large-scale commercial application. Additionally, the use of cuprous bromide-triphenylphosphine catalysts necessitates high reaction temperatures, which not only increases energy consumption but also promotes the formation of side products that compromise the purity of the final intermediate. These factors collectively create significant bottlenecks for cost reduction in API manufacturing, forcing procurement teams to manage volatile pricing and inconsistent supply availability.
The Novel Approach
In stark contrast to the deficiencies of the prior art, the method disclosed in patent CN111170992A presents a streamlined and highly efficient synthetic strategy. This novel approach initiates with p-acetamino aniline, a commodity chemical that is cheap and easily available, thereby fundamentally altering the cost structure of the production process. The route employs a series of amidation, halogenation, and condensation reactions that proceed under mild conditions, significantly reducing the energy footprint and operational risks associated with high-temperature processing. By avoiding the use of expensive iodo-materials and hazardous phosphorus reagents, the process inherently lowers the generation of three wastes, aligning with modern green chemistry principles. The reaction selectivity is meticulously controlled at each step, ensuring that functional group transformations occur with precision, which directly translates to a total yield that can reach 72 percent. This substantial improvement in efficiency not only enhances the economic feasibility of the project but also ensures a more reliable supply chain for high-purity pharmaceutical intermediates, meeting the rigorous demands of global regulatory bodies.
Mechanistic Insights into Amidation and Cyclization Reactions
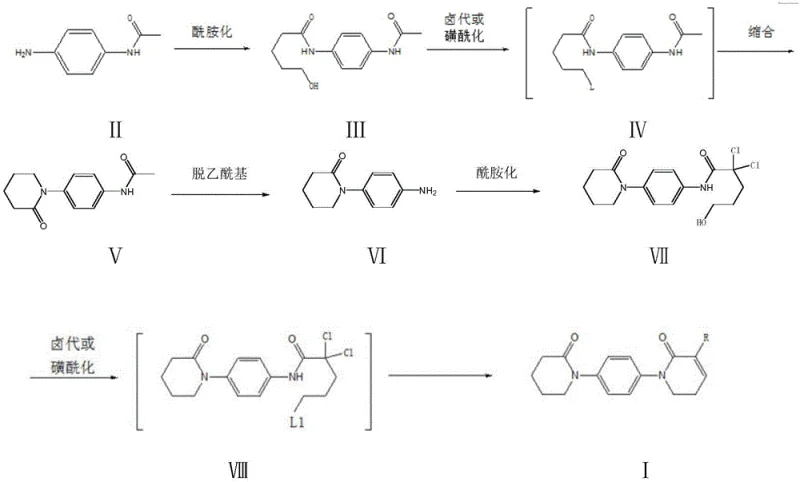
The core of this technological breakthrough lies in the precise orchestration of amidation and cyclization reactions that construct the 5,6-dihydropyridine-2(1H)-ketone scaffold. The process begins with the amidation of p-acetamino aniline with delta-valerolactone, a reaction that is highly specific and occurs exclusively between the amino group and the ester functionality of the lactone. This selectivity is crucial as it prevents unwanted side reactions that could lead to complex impurity profiles, thereby simplifying the downstream purification process. Following this, the hydroxyl group generated in the first step serves as the sole active site for subsequent halogenation or sulfonylation, demonstrating a high degree of chemoselectivity. The intramolecular condensation that follows effectively closes the piperidin-2-one ring, a key structural motif required for the biological activity of the final API. Each transformation is optimized to proceed with minimal byproduct formation, ensuring that the reaction mixture remains clean and manageable. This level of control over the reaction mechanism is essential for maintaining the stringent purity specifications required for pharmaceutical ingredients, as it minimizes the risk of genotoxic impurities or difficult-to-remove side products.
Furthermore, the impurity control mechanism embedded within this synthesis route is robust, relying on the stability of the intermediates and the mildness of the reaction conditions. The deacetylation step, for instance, is carried out in an alcoholic solvent under acidic conditions that are gentle enough to preserve the integrity of the sensitive ketone and amine functionalities while efficiently removing the protecting group. The subsequent amidation with 2,2-dichloro-delta-valerolactone is designed to introduce the necessary side chain without compromising the existing ring structure. The final condensation-elimination or substitution reaction in the presence of morpholine is conducted at temperatures that prevent thermal degradation, ensuring that the final product retains its structural fidelity. This systematic approach to impurity management means that the crude product often meets high purity standards even before recrystallization, reducing the need for extensive chromatographic purification. For R&D directors, this translates to a more predictable and reproducible process that can be validated with greater ease, facilitating faster technology transfer from the laboratory to the commercial manufacturing plant.
How to Synthesize 5,6-dihydropyridine-2(1H)-ketone Derivatives Efficiently
The synthesis of these critical intermediates is achieved through a multi-step sequence that balances chemical efficiency with operational simplicity. The process begins with the preparation of the hydroxy-pentanoyl intermediate, followed by cyclization to form the piperidinone ring, and concludes with the introduction of the specific substituents required for Apixaban activity. Detailed standard operating procedures for each reaction step, including solvent ratios, temperature profiles, and work-up methods, are essential for replicating the high yields reported in the patent. The following guide outlines the critical stages of this synthesis, providing a framework for technical teams to evaluate the feasibility of implementation within their existing infrastructure. Adhering to these standardized steps ensures that the quality and consistency of the output meet the rigorous demands of the pharmaceutical industry.
- Perform amidation reaction between p-acetamino aniline and delta-valerolactone to obtain the hydroxy-pentanoyl intermediate.
- Execute halogenation or sulfonylation followed by condensation to form the piperidin-2-one ring structure.
- Complete the synthesis via deacetylation, second amidation, and final condensation-elimination to yield the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound advantages for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring continuity. The elimination of expensive iodo-substituted raw materials represents a significant shift in the cost structure, allowing for substantial cost savings that can be passed down through the supply chain or reinvested into further process optimization. The use of cheap and easily available starting materials like p-acetamino aniline reduces the risk of supply disruptions caused by the scarcity of specialized reagents, thereby enhancing supply chain reliability. Moreover, the mild reaction conditions and reduced waste generation lower the operational costs associated with energy consumption and environmental compliance, making the process economically attractive for long-term production. These factors collectively contribute to a more resilient supply network that can withstand market fluctuations and regulatory changes, providing a competitive edge in the global marketplace.
- Cost Reduction in Manufacturing: The replacement of high-cost iodo-materials with commodity chemicals like p-acetamino aniline drastically lowers the raw material expenditure, which is a primary driver of overall manufacturing costs. By avoiding the use of phosphorus pentachloride and expensive metal catalysts, the process eliminates the need for costly waste treatment and catalyst recovery systems, further reducing the operational overhead. The high yield and selectivity of the reaction steps minimize the loss of valuable materials, ensuring that a greater proportion of the input is converted into saleable product. This efficiency translates directly into a lower cost of goods sold, enabling more competitive pricing strategies without compromising profit margins. Additionally, the simplified work-up procedures reduce the consumption of solvents and utilities, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable raw materials mitigates the risk of supply chain disruptions that are often associated with specialized or imported reagents. The robustness of the synthesis route, characterized by mild conditions and stable intermediates, ensures consistent production output even in the face of minor operational variations. This stability is crucial for maintaining uninterrupted supply to downstream API manufacturers, who depend on timely deliveries to meet their own production schedules. The reduced complexity of the process also means that it can be easily scaled up or transferred between different manufacturing sites without significant re-engineering, providing flexibility in supply chain management. Consequently, partners can rely on a steady flow of high-quality intermediates, fostering stronger long-term relationships and trust within the supply network.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reaction vessels and equipment that are common in fine chemical manufacturing facilities. The reduced generation of hazardous waste, particularly phosphorus-containing effluents, simplifies compliance with increasingly stringent environmental regulations, reducing the risk of fines or production shutdowns. The green nature of the synthesis, with its lower energy requirements and safer reagents, aligns with corporate sustainability goals, enhancing the brand reputation of the manufacturer. This environmental compatibility ensures that the production can be sustained over the long term without facing regulatory hurdles, securing the future viability of the supply chain. Furthermore, the ease of scale-up allows for rapid response to increased market demand, ensuring that supply can be expanded quickly to capture new opportunities.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial aspects of this synthesis method, based on the detailed data provided in the patent documentation. These answers are designed to clarify the specific advantages and operational details for stakeholders evaluating the technology. Understanding these key points is essential for making informed decisions about the adoption and implementation of this route in a commercial setting. The information provided here reflects the current state of the art as described in the intellectual property disclosure.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This method avoids expensive iodo-substituted raw materials and hazardous phosphorus pentachloride, utilizing cheap and easily available starting materials like p-acetamino aniline. It significantly reduces wastewater generation and eliminates the need for high-temperature coupling reactions, resulting in higher selectivity and purity suitable for industrial production.
Q: How does this process impact the overall yield and purity of the intermediate?
A: The patent data indicates a total yield reaching approximately 72 percent, with individual step yields often exceeding 90 percent. The process ensures high reaction selectivity at each functional group transformation, leading to liquid phase purity levels consistently above 99 percent, which minimizes downstream purification burdens.
Q: Is this method scalable for commercial API manufacturing?
A: Yes, the reaction conditions are mild and easy to realize, with solvents and reagents that are standard in fine chemical manufacturing. The reduced generation of three wastes and the stability of intermediates make it highly suitable for large-scale commercial production without the safety risks associated with prior art routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-dihydropyridine-2(1H)-ketone Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at navigating the complexities of synthetic organic chemistry, ensuring that routes like the one described in patent CN111170992A are optimized for maximum efficiency and safety. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5,6-dihydropyridine-2(1H)-ketone derivative meets the highest industry standards. Our commitment to quality and reliability makes us the preferred partner for pharmaceutical companies seeking a stable and high-performance supply chain for their critical intermediates.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply strategy. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your organization. We are ready to provide specific COA data and route feasibility assessments to support your decision-making process. By partnering with us, you gain access to a wealth of technical expertise and a robust manufacturing infrastructure designed to support your growth and innovation in the pharmaceutical sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
