Advanced Synthetic Route for N-Substituted O-Bromobenzylamide: Commercial Scalability and Purity
The pharmaceutical industry constantly seeks robust synthetic pathways for critical intermediates, and patent CN101024619B presents a transformative approach to producing N-substituted o-bromobenzylamide compounds. These specific chemical structures serve as vital precursors for the synthesis of physiologically active substances, most notably indole-2-one derivatives which are foundational in developing growth factor receptor inhibitors and anti-inflammatory medications. The traditional landscape of synthesizing these intermediates has been fraught with complexity, often involving hazardous reagents and multi-step sequences that hinder efficient commercialization. This new methodology disrupts the status quo by introducing a one-pot reaction system that utilizes 1-bromo-2-(2,2-dibromovinyl)benzene and primary amines under mild alkaline conditions. By leveraging a dimethyl sulfoxide and water mixed solvent system, the process not only simplifies the operational workflow but also aligns with modern green chemistry principles, offering a compelling value proposition for reliable pharmaceutical intermediate supplier networks seeking to optimize their upstream production capabilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
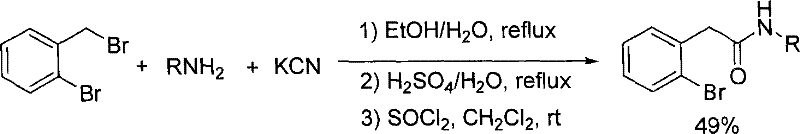
Historically, the preparation of N-substituted o-bromobenzylamide has relied on cumbersome multi-step sequences that introduce significant inefficiencies into the supply chain. As illustrated in prior art, one common pathway involves the reaction of o-bromobenzyl bromide with potassium cyanide, followed by hydrolysis and amidation, a process that is not only labor-intensive but also introduces severe safety hazards due to the use of cyanide salts. Another reported method utilizes o-bromophenylacetonitrile which requires low-temperature conditions and specialized reagents like phosphorus nitrofluoride, leading to operational complexities and increased energy consumption. Furthermore, the cumulative yields of these traditional routes are often suboptimal, with some documented processes achieving only 49% efficiency after three distinct reaction stages, resulting in substantial material waste and inflated production costs. The reliance on chlorinating agents such as thionyl chloride in alternative pathways further exacerbates environmental concerns, generating corrosive waste streams that require expensive treatment protocols before disposal, thereby negatively impacting the overall cost reduction in API manufacturing strategies.
The Novel Approach
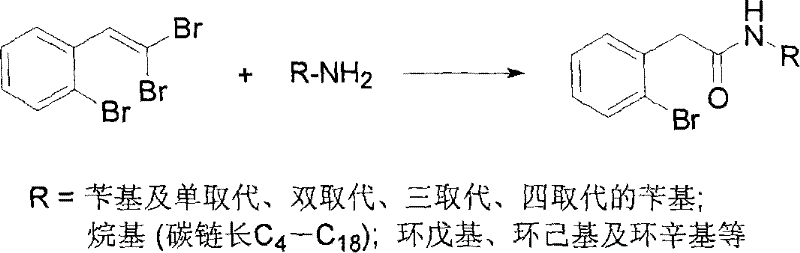
In stark contrast to these legacy methods, the novel approach detailed in the patent data streamlines the synthesis into a single, efficient reaction step that dramatically enhances process viability. By employing 1-bromo-2-(2,2-dibromovinyl)benzene as the starting material, the reaction proceeds through a nucleophilic substitution and hydrolysis mechanism that eliminates the need for isolated intermediate steps. The use of sodium hydroxide as a base in a 3:1 DMSO-water solvent system provides a benign reaction environment that operates effectively at temperatures between 70°C and 100°C, removing the necessity for cryogenic cooling or high-pressure vessels. This simplification allows for a direct conversion to the target amide with reported yields ranging significantly higher than conventional methods, often exceeding 80% in optimized examples. The workup procedure is equally streamlined, involving simple aqueous extraction and standard column chromatography, which facilitates easier purification and reduces the time required for quality control analysis, ultimately supporting the commercial scale-up of complex pharmaceutical intermediates with greater agility and reduced operational overhead.
Mechanistic Insights into NaOH-Catalyzed Hydrolysis and Amidation
The core chemical transformation in this patented method relies on the unique reactivity of the 2,2-dibromovinyl group attached to the benzene ring, which acts as a masked acyl equivalent under alkaline conditions. When heated in the presence of a primary amine and sodium hydroxide, the dibromovinyl moiety undergoes hydrolysis to generate an reactive acyl intermediate in situ, which is immediately trapped by the nucleophilic amine to form the stable amide bond. This tandem hydrolysis-amidation sequence avoids the isolation of unstable acid chloride or nitrile intermediates, thereby minimizing the formation of side products that typically plague multi-step syntheses. The polar aprotic nature of DMSO enhances the solubility of both the organic substrate and the inorganic base, ensuring homogeneous reaction conditions that promote consistent kinetics throughout the reaction vessel. This mechanistic elegance ensures that the reaction proceeds with high atom economy, as the bromine atoms are effectively displaced or converted into benign inorganic salts, reducing the burden on downstream waste management systems and enhancing the overall sustainability profile of the manufacturing process.
From an impurity control perspective, this mechanism offers distinct advantages by avoiding the introduction of extraneous functional groups that are difficult to remove. Traditional methods involving cyanide often leave trace metal residues or cyanide byproducts that require rigorous and costly purification steps to meet pharmaceutical grade specifications. In this new route, the primary byproducts are inorganic bromides and water, which are easily separated during the aqueous workup phase using ethyl acetate extraction. The absence of heavy metal catalysts or toxic reagents means that the final crude product has a cleaner impurity profile, simplifying the subsequent column chromatography purification step. This results in a final solid product with high structural integrity and purity, which is critical for downstream applications in drug synthesis where impurity thresholds are strictly regulated. The ability to consistently produce high-purity pharmaceutical intermediates with minimal purification effort directly translates to improved batch-to-batch reproducibility and reduced risk of regulatory delays during the drug approval process.
How to Synthesize N-Substituted O-Bromobenzylamide Efficiently
To implement this synthesis effectively, operators must adhere to specific molar ratios and temperature controls to maximize yield and minimize side reactions. The process begins by charging a reactor with the dibromovinyl benzene derivative and the chosen primary amine, followed by the addition of the DMSO-water solvent mixture and sodium hydroxide base. It is crucial to maintain the reaction temperature within the 70-100°C window to ensure complete conversion of the starting material, which is monitored via thin-layer chromatography until the disappearance of the reactant spot.
- Mix 1-bromo-2-(2,2-dibromovinyl)benzene with primary amine and sodium hydroxide in a 3: 1 DMSO/Water solvent system.
- Heat the reaction mixture to 70-100°C and monitor via TLC until the starting material is fully consumed.
- Quench with water, extract with ethyl acetate, wash, dry over magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond simple chemical efficiency. The elimination of hazardous reagents like potassium cyanide and thionyl chloride significantly reduces the regulatory burden and safety compliance costs associated with chemical handling and storage. This shift allows facilities to operate with lower insurance premiums and reduced need for specialized containment infrastructure, leading to substantial cost savings in facility operations. Furthermore, the use of readily available raw materials such as sodium hydroxide and common solvents ensures a stable supply chain that is less susceptible to market volatility compared to specialized catalysts or reagents. The simplified workup procedure also reduces the consumption of utilities such as cooling water and energy, contributing to a lower carbon footprint and aligning with corporate sustainability goals.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction eliminates the need for multiple isolation and purification steps, which drastically reduces labor hours and solvent consumption per kilogram of product. By avoiding the use of expensive transition metal catalysts or specialized low-temperature equipment, the capital expenditure required for production is significantly lowered. The high yield achieved in a single step means that less raw material is wasted, directly improving the material cost efficiency of the final intermediate. Additionally, the simplified waste stream reduces the costs associated with hazardous waste disposal and environmental treatment, further enhancing the overall economic viability of the process for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like sodium hydroxide and DMSO ensures that raw material sourcing is robust and less prone to disruption compared to processes requiring niche reagents. The mild reaction conditions reduce the risk of equipment failure or batch loss due to thermal runaway or pressure issues, ensuring consistent production schedules. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts that can delay drug development timelines. The ability to scale this process from gram to ton scale without significant re-engineering provides flexibility to meet fluctuating market demands efficiently.
- Scalability and Environmental Compliance: The aqueous workup and absence of toxic byproducts make this process highly scalable with minimal environmental impact. The waste generated is primarily saline and organic solvent, which can be treated using standard industrial wastewater protocols, avoiding the need for specialized incineration or chemical neutralization. This compliance with environmental regulations facilitates faster permitting for new production lines and reduces the risk of regulatory fines. The green chemistry profile of this method also enhances the marketability of the final product to eco-conscious pharmaceutical partners who prioritize sustainable supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary safety advantages of this new synthetic method compared to traditional routes?
A: Unlike conventional methods that rely on highly toxic potassium cyanide or hazardous thionyl chloride, this patent utilizes sodium hydroxide and a DMSO/water solvent system, drastically reducing occupational health risks and environmental waste disposal costs.
Q: How does the yield of this novel approach compare to existing multi-step processes?
A: The patented method achieves yields ranging from 64% to 83% in a single reaction step, whereas traditional multi-step pathways often suffer from cumulative yield losses, with some reported routes yielding as low as 49% after three distinct stages.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the use of common reagents like sodium hydroxide and easily removable solvents like ethyl acetate, combined with mild reaction temperatures of 70-100°C, makes this protocol highly adaptable for ton-scale manufacturing without requiring specialized cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted O-Bromobenzylamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed patent CN101024619B and validated its potential for industrial application, confirming its suitability for producing high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of N-substituted o-bromobenzylamide meets the highest international standards for pharmaceutical use.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced technology for your specific drug development projects. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates how this specific synthetic route can optimize your bill of materials and reduce overall production timelines. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your unique molecular requirements. Let us help you secure a stable, cost-effective, and compliant supply of this vital intermediate for your next generation of therapeutic agents.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
