Advanced Synthesis of Substituted Phenols for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for critical building blocks, particularly substituted phenols which serve as pivotal intermediates in the synthesis of complex active pharmaceutical ingredients. Patent CN102086147A discloses a highly efficient preparation method for substituted phenols that addresses longstanding challenges in yield and operational complexity. This technology leverages a strategic diazotization followed by a hydrolysis or reduction sequence to convert nitroaniline derivatives directly into the corresponding phenolic structures. By integrating this methodology into existing manufacturing frameworks, producers can achieve substantial improvements in process efficiency and product quality. The significance of this innovation lies in its ability to bypass traditional multi-step syntheses that often suffer from low atom economy and difficult purification requirements. For R&D directors and procurement specialists, understanding the nuances of this patent provides a competitive edge in sourcing high-quality intermediates for drug development pipelines ranging from antiviral agents to kinase inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for generating substituted phenols frequently involve harsh reaction conditions and the use of expensive transition metal catalysts that complicate the downstream processing. Conventional methods often require multiple protection and deprotection steps to manage the reactivity of the phenolic hydroxyl group, leading to increased waste generation and prolonged production cycles. Furthermore, the reliance on precious metal catalysts introduces the risk of heavy metal contamination, necessitating rigorous and costly purification protocols to meet stringent regulatory standards for pharmaceutical intermediates. These legacy processes often struggle with scalability, as exothermic reactions and sensitive intermediates can pose significant safety hazards when transitioning from laboratory benchtop to industrial reactors. The cumulative effect of these limitations is a higher cost of goods sold and a less reliable supply chain, which can delay critical drug development timelines and erode profit margins for manufacturing partners.
The Novel Approach
The methodology outlined in the patent data presents a transformative alternative by utilizing a direct substitution strategy that streamlines the synthetic sequence into fewer operational steps. This novel approach capitalizes on the reactivity of diazonium salts generated in situ from readily available o-nitroaniline precursors, eliminating the need for exotic reagents or complex catalytic systems. By conducting the reaction in a strong acid solution, the process ensures high conversion rates while maintaining control over the reaction kinetics to minimize side product formation. The versatility of this method is evident in its compatibility with various substituents, including halogens and alkyl groups, allowing for the synthesis of a diverse library of phenolic compounds without modifying the core protocol. This simplification not only reduces the operational burden on manufacturing teams but also enhances the overall safety profile of the process by avoiding unstable intermediates that are common in older synthetic routes.
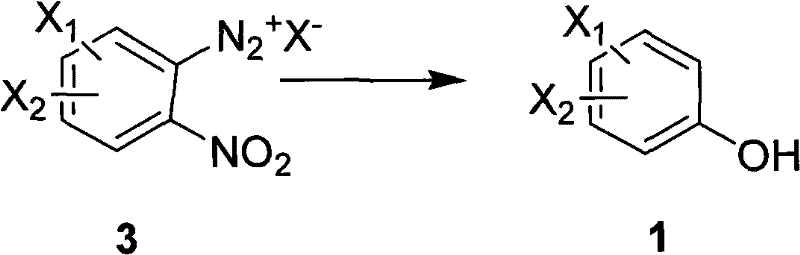
Mechanistic Insights into Diazotization and Hydrolysis
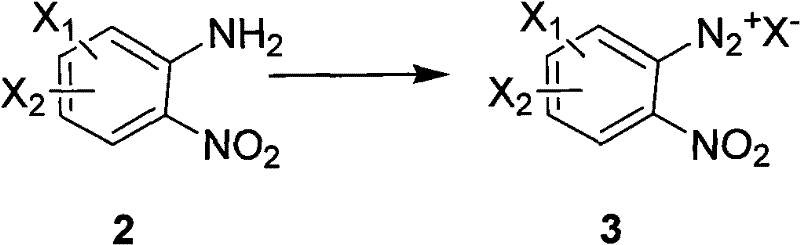
The core of this synthetic innovation relies on a precise diazotization mechanism followed by a controlled hydrolysis or reduction step to install the hydroxyl functionality. Initially, the o-nitroaniline precursor undergoes diazotization in the presence of an inorganic nitrite and a strong acid, forming a reactive diazonium salt intermediate that is stabilized by the acidic medium. This step is critical as it activates the aromatic ring for nucleophilic substitution, setting the stage for the introduction of the oxygen atom. The subsequent transformation involves the displacement of the diazo group, which can be achieved through thermal hydrolysis in water or via reduction using agents like hypophosphorous acid or alcohols. The choice of reducing agent and reaction temperature allows for fine-tuning the process to accommodate different substrate sensitivities, ensuring that sensitive functional groups elsewhere on the molecule remain intact during the transformation.
Impurity control is inherently built into this mechanistic pathway due to the high selectivity of the diazonium intermediate towards the desired substitution. Unlike radical-based halogenation or oxidation methods that can lead to over-substitution or ring degradation, this ionic mechanism proceeds with high regioselectivity, preserving the substitution pattern of the starting material. The use of strong acids not only facilitates the formation of the diazonium salt but also suppresses the formation of azo-coupling byproducts that often plague phenol synthesis. Furthermore, the workup procedure involving extraction and crystallization is highly effective at removing inorganic salts and unreacted starting materials, resulting in a final product with exceptional purity profiles. This level of chemical fidelity is essential for pharmaceutical applications where impurity thresholds are strictly regulated, ensuring that the intermediate meets the rigorous quality standards required for subsequent coupling reactions in API synthesis.
How to Synthesize Substituted Phenol Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent stoichiometry to maximize yield and safety. The process begins with the dissolution of the nitroaniline starting material in a suitable solvent system, followed by the slow addition of nitrite to manage the exotherm of diazotization. Once the diazonium salt is formed, the reaction mixture is warmed to facilitate the substitution, with the specific temperature profile depending on the chosen reducing agent. Detailed standard operating procedures for scaling this reaction from laboratory to production scale are critical for maintaining consistency and safety. The following guide outlines the standardized synthesis steps derived from the patent data to ensure reproducible results.
- Dissolve the o-nitroaniline precursor in a strong acid solution such as hydrochloric or sulfuric acid to prepare for diazotization.
- Add an inorganic nitrite reagent at low temperatures to form the diazonium salt intermediate in situ.
- Heat the mixture with a reducing agent or water to facilitate the substitution of the diazo group with a hydroxyl group, yielding the target phenol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of procurement managers and supply chain directors. The reliance on commodity chemicals such as mineral acids and sodium nitrite significantly reduces raw material costs compared to processes requiring specialized catalysts or organometallic reagents. This shift to readily available inputs enhances supply chain resilience, mitigating the risk of disruptions caused by the scarcity of exotic reagents. Additionally, the simplified workup procedure reduces the consumption of solvents and energy, contributing to a lower environmental footprint and reduced waste disposal costs. These factors combine to create a more cost-effective manufacturing model that can withstand market volatility while maintaining high margins.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes the need for costly metal scavenging steps, which are often a significant portion of the processing budget in traditional phenol synthesis. By utilizing simple acid-base chemistry and common reducing agents, the overall cost of goods is drastically lowered without compromising on product quality. This economic efficiency allows for more competitive pricing strategies in the global market for pharmaceutical intermediates. Furthermore, the high yields achieved in this process mean that less raw material is wasted, maximizing the value extracted from every kilogram of input.
- Enhanced Supply Chain Reliability: The use of stable and commercially available starting materials ensures a consistent supply of intermediates, reducing the lead time associated with sourcing specialized reagents. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of large pharmaceutical clients. The robustness of the reaction conditions also means that the process is less susceptible to variations in raw material quality, further stabilizing the supply chain. Consequently, manufacturers can offer more reliable delivery commitments, strengthening their partnerships with downstream API producers.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction parameters that can be easily adjusted for large-scale reactors without significant re-engineering. The reduced generation of hazardous waste and the avoidance of heavy metals simplify compliance with environmental regulations, lowering the regulatory burden on manufacturing facilities. This alignment with green chemistry principles not only reduces operational risks but also enhances the corporate sustainability profile of the manufacturer. As the industry moves towards more sustainable practices, this technology positions suppliers as forward-thinking partners capable of meeting future regulatory demands.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this substituted phenol synthesis method. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation. They are intended to provide clarity on the feasibility and advantages of adopting this route for industrial production. Understanding these details is essential for making informed decisions about process integration and supplier selection.
Q: What are the primary advantages of this phenol synthesis method over traditional routes?
A: This method significantly simplifies the process by avoiding multi-step sequences and expensive catalysts. It utilizes readily available raw materials and achieves high yields with minimal side reactions, ensuring a cleaner impurity profile crucial for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability. The reaction conditions are manageable, and the workup involves standard extraction and crystallization techniques, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Q: How does this method impact the purity of the final substituted phenol?
A: The method demonstrates excellent control over side reactions, consistently delivering products with HPLC purity exceeding 99%. This high level of purity reduces the burden on downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of pharmaceutical development and commercialization. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical manufacturing settings. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of substituted phenol meets the exacting standards required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of chemical manufacturing, delivering products that facilitate your drug development timelines.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced synthetic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact us to request specific COA data and route feasibility assessments that demonstrate how our capabilities align with your project goals. By partnering with us, you gain access to a reliable source of high-purity intermediates backed by deep technical expertise and a commitment to long-term supply stability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
