Advanced Synthesis of Apixaban Intermediates: A Scalable Commercial Solution
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical anticoagulant therapies, and the synthesis of Apixaban intermediates remains a focal point for process optimization. Patent CN111170930B discloses a groundbreaking preparation method for 5,6-dihydropyridine-2(1H)-ketone derivatives, which serve as pivotal building blocks in the production of Apixaban, a direct Factor Xa inhibitor. This technical disclosure represents a significant departure from traditional synthetic routes that rely on costly and unstable iodo-substances. By leveraging p-phenylenediamine and delta-valerolactone as primary starting materials, the invention establishes a foundation for a more economical and environmentally sustainable supply chain. The strategic shift away from complex coupling reactions towards a streamlined amidation and condensation sequence offers profound implications for commercial manufacturing efficiency. This report analyzes the technical merits and commercial viability of this novel approach for global procurement and R&D stakeholders.
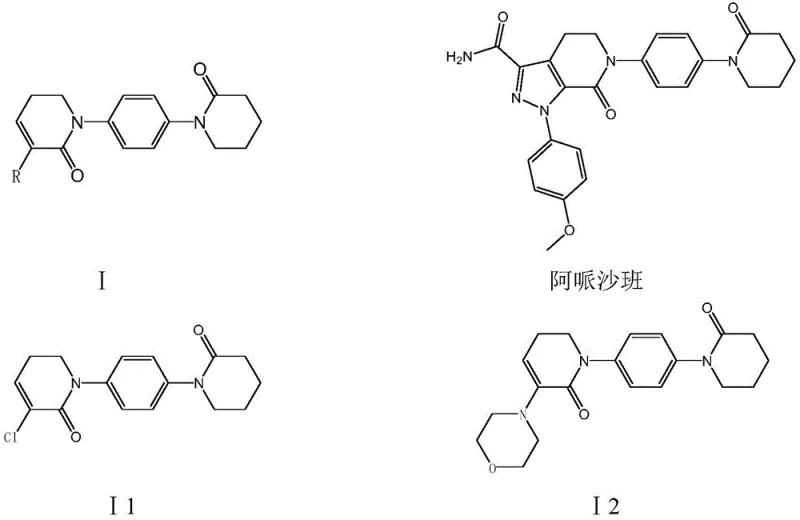
The structural integrity of the final API depends heavily on the purity and consistency of these key intermediates, specifically compounds I1 and I2 as depicted in the patent documentation. The ability to synthesize these complex heterocyclic structures with high selectivity directly influences the downstream purification burden and overall process mass intensity. For R&D directors evaluating technology transfer, the elimination of phosphorus-containing byproducts and heavy metal catalysts in the early stages of synthesis reduces the complexity of impurity profiling. This patent provides a clear roadmap for achieving high-purity pharmaceutical intermediates without compromising on yield or operational safety. The technical specifications outlined herein demonstrate a mature understanding of organic synthesis constraints and offer a viable alternative for companies seeking to diversify their supplier base for cardiovascular medication components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Apixaban intermediates has been plagued by reliance on expensive raw materials and hazardous reagents that complicate industrial scale-up. Prior art routes, such as those described in WO2003049681, utilize piperidine-2-ketone and phosphorus pentachloride for chlorination, a process known to generate substantial amounts of phosphorus-containing wastewater. This not only increases the environmental footprint but also necessitates costly waste treatment protocols that erode profit margins. Furthermore, the use of 1-(4-iodophenyl)-2-piperidone introduces a significant cost burden due to the high market price of iodo-substances and their inherent instability during storage and handling. The multi-step nature of these conventional pathways often involves high-temperature coupling reactions that promote side reactions, leading to lower overall yields and complex impurity profiles that require extensive chromatographic purification. These factors collectively create bottlenecks in the supply chain, making it difficult to ensure consistent delivery volumes for large-scale commercial production.
The Novel Approach
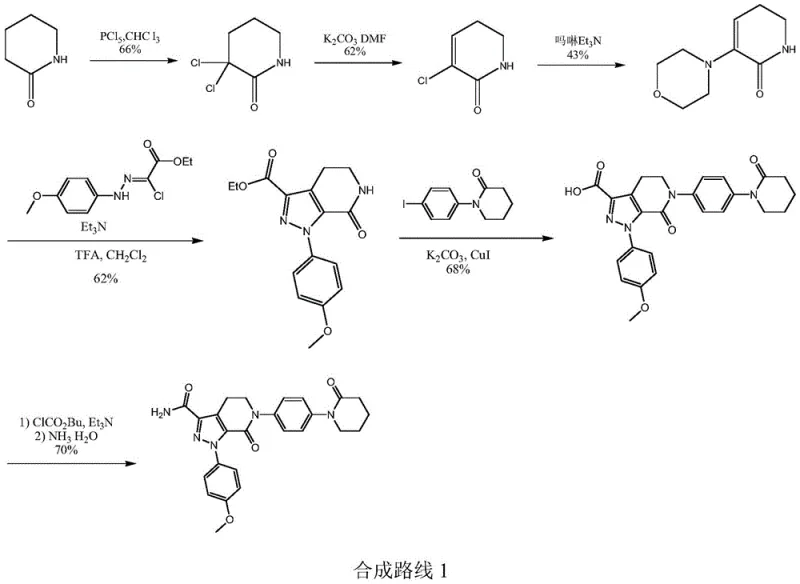
In stark contrast, the method disclosed in CN111170930B introduces a paradigm shift by utilizing p-phenylenediamine and delta-valerolactone, which are cheap and easily obtained industrial chemicals. This novel approach circumvents the need for expensive iodo-intermediates entirely, replacing them with a robust amidation strategy that builds the molecular complexity from simpler precursors. The process is designed to be operationally simple, with reaction conditions that are mild and easy to realize in standard stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. By avoiding the use of phosphorus pentachloride and cuprous bromide-triphenylphosphine catalysts, the new route significantly reduces the generation of hazardous waste and eliminates the need for expensive heavy metal removal steps. This results in a cleaner reaction profile with high reaction selectivity in each step, ensuring that the final product meets stringent purity specifications with minimal downstream processing. The total yield of this optimized pathway is reported to reach substantial levels, demonstrating its superiority over the low-yielding conventional methods that struggle to exceed 30% efficiency in complex sequences.
Mechanistic Insights into Amidation and Cyclization Strategy
The core of this innovative synthesis lies in the precise control of functional group transformations, beginning with the amidation of p-phenylenediamine with delta-valerolactone to form N,N'-bis(5-hydroxy-N-pentanoyl)p-phenylenediamine. This initial step is critical as it sets the stereochemical and structural foundation for the subsequent ring-closing reactions. The hydroxyl groups of the amidation product are then activated through reaction with a halogenated reagent or sulfonyl chloride, facilitating an intramolecular condensation that constructs the piperidine-2-ketone rings. This cyclization is performed under basic conditions in polar aprotic solvents, which promotes the nucleophilic attack required to close the rings while minimizing intermolecular polymerization. The careful selection of solvents such as N,N-dimethylformamide or toluene allows for precise temperature control, ensuring that the reaction proceeds with high conversion rates and minimal formation of oligomeric byproducts. This mechanistic pathway ensures that the two piperidone segments are introduced simultaneously, avoiding the regioselectivity issues often encountered when introducing them sequentially in older methodologies.
Following the formation of the bis-piperidone structure, the process employs a controlled monohalogenation at the ortho-position of the carbonyl group, followed by elimination to generate the crucial carbon-carbon double bond of the dihydropyridine system. This step is meticulously managed to control the dosage of the halogenating reagent, ensuring that the conversion rate of raw materials remains high while preventing over-halogenation which could lead to intractable impurities. The resulting double bond is highly reactive and serves as a handle for the final functionalization, where addition of a halogenating reagent followed by elimination or substitution with morpholine introduces the desired R substituent. This modular approach allows for the flexible synthesis of various derivatives, including the 3-chloro and 3-morpholino variants required for different API analogs. The high selectivity of these elimination and substitution reactions ensures that the final product possesses the exact structural configuration needed for biological activity, thereby reducing the risk of batch failure due to isomeric impurities.
How to Synthesize 5,6-dihydropyridine-2(1H)-one Derivative Efficiently
The standardized synthesis of these high-value intermediates requires strict adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process begins with the preparation of the amidation product, followed by activation and cyclization, and concludes with the functionalization of the dihydropyridine ring. Each step has been optimized for solvent ratios, temperature ranges, and reaction times to maximize yield and purity while minimizing waste generation. Operators must monitor the reaction progress closely, particularly during the halogenation steps, to prevent the formation of side products that could complicate purification. The detailed standardized synthesis steps see the guide below.
- Perform amidation reaction between p-phenylenediamine and delta-valerolactone to obtain N,N'-bis(5-hydroxy-N-pentanoyl)p-phenylenediamine.
- Subject the amidation product to halogenation or sulfonylation followed by condensation to form 1,4-di(piperidine-2-ketone-1-yl)benzene.
- Execute ortho-halogenation and elimination to generate the dihydropyridine core structure.
- Finalize with addition and elimination or substitution reactions to introduce specific R substituents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs, as the process eliminates the dependency on volatile and expensive iodo-substances that are subject to significant market price fluctuations. By switching to commodity chemicals like p-phenylenediamine, manufacturers can secure a more stable and predictable cost base, which is essential for long-term contract negotiations and budget planning. Furthermore, the simplification of the process flow reduces the operational complexity, allowing for faster batch turnover and increased production capacity without the need for capital-intensive equipment upgrades. This efficiency gain translates directly into improved supply chain reliability, ensuring that critical intermediates are available to meet the demands of downstream API manufacturing schedules without unexpected delays.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and iodo-reagents removes significant cost drivers from the bill of materials, leading to substantial cost savings in pharmaceutical intermediate manufacturing. The avoidance of phosphorus pentachloride also reduces the expenditure associated with hazardous waste disposal and environmental compliance, further enhancing the economic viability of the process. Additionally, the high yield and purity achieved in each step minimize the loss of valuable materials, ensuring that the overall process mass intensity is optimized for commercial production. These factors combine to create a highly competitive cost structure that allows suppliers to offer more attractive pricing without compromising on quality or margin.
- Enhanced Supply Chain Reliability: Utilizing cheap and easily obtained raw materials mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. The robustness of the reaction conditions means that production can be maintained consistently across different facilities and geographic locations, providing a diversified supply base for global pharmaceutical companies. The reduced generation of wastewater and hazardous byproducts simplifies the regulatory approval process for new manufacturing sites, accelerating the time to market for new supply sources. This reliability is crucial for maintaining the continuity of API production, especially for life-saving medications where supply interruptions can have severe consequences for patient health.
- Scalability and Environmental Compliance: The mild reaction conditions and simple operational procedures make this process highly scalable from pilot plant to commercial tonnage production. The significant reduction in three wastes, particularly phosphorus-containing wastewater, aligns with increasingly stringent environmental regulations, reducing the risk of regulatory penalties or shutdowns. The green nature of the process also enhances the corporate social responsibility profile of the manufacturer, appealing to partners who prioritize sustainable supply chains. This scalability ensures that the method can meet growing global demand for anticoagulant therapies while maintaining a low environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the patent specifications and are intended to clarify the operational benefits and technical constraints of the method. Understanding these details is essential for stakeholders evaluating the feasibility of technology transfer or supplier qualification. The responses highlight the specific advantages of this route over conventional methods in terms of cost, safety, and environmental impact.
Q: What are the primary cost advantages of this synthesis route?
A: This method avoids expensive iodo-substances and high-temperature coupling reactions required in prior art, utilizing cheap and easily obtained raw materials like p-phenylenediamine.
Q: How does this process impact environmental compliance?
A: The process generates a significantly smaller amount of wastewater compared to conventional routes using phosphorus pentachloride, aligning with stricter environmental regulations.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild and easy to realize, with high reaction selectivity and product yield, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5,6-dihydropyridine-2(1H)-one derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of complex heterocyclic synthesis and is equipped to implement this patented route with stringent purity specifications. We operate rigorous QC labs that ensure every batch of 5,6-dihydropyridine-2(1H)-one derivative meets the highest international standards for pharmaceutical intermediates. Our commitment to quality and consistency makes us an ideal partner for companies seeking to optimize their supply chain for Apixaban and related cardiovascular therapies. We understand the critical nature of these intermediates and are dedicated to providing a seamless supply experience.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific production needs. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this more efficient manufacturing method. Our experts are ready to provide specific COA data and route feasibility assessments to support your decision-making process. By partnering with us, you gain access to a reliable supply of high-purity intermediates that will enhance the stability and profitability of your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
