Advanced Synthesis of Fluorinated Indole Derivatives for High-Purity Pharmaceutical Applications
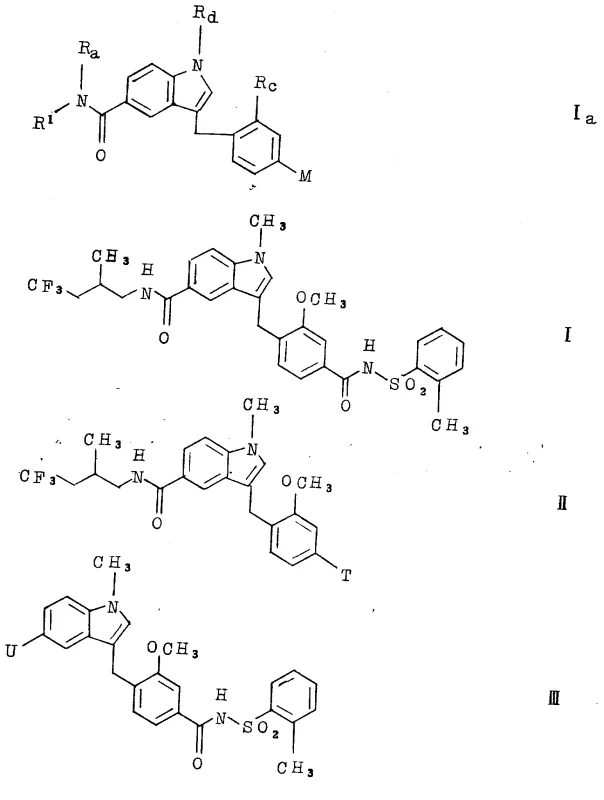
The pharmaceutical landscape for respiratory and inflammatory diseases continues to evolve, driven by the need for more potent and selective leukotriene antagonists. Patent CN1025786C introduces a novel class of carbamoyl derivatives, specifically 5-(2-methyl-4,4,4-trifluoro butyl formamyl) indole derivatives, which demonstrate significant pharmacological activity in antagonizing arachidonic acid metabolites. These compounds are particularly valuable for treating conditions such as asthma, allergic rhinitis, and inflammatory disorders where leukotrienes play a critical pathogenic role. The structural innovation lies in the specific fluorinated side chain attached to the indole core, which enhances metabolic stability and receptor binding affinity compared to earlier generations of antagonists. For R&D directors and procurement specialists, understanding the synthesis and supply chain implications of this patent is crucial for developing next-generation anti-inflammatory therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for leukotriene antagonists often rely on complex multi-step sequences that suffer from low overall yields and difficult purification processes. Conventional methods frequently utilize non-selective alkylation reactions that produce racemic mixtures, necessitating expensive and time-consuming chiral resolution steps to isolate the active enantiomer. Furthermore, older pathways often involve harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents, which pose significant safety risks and environmental compliance challenges during commercial scale-up. The presence of multiple functional groups in these molecules also leads to the formation of closely related impurities that are difficult to separate, potentially compromising the purity profile required for pharmaceutical grade intermediates. These limitations result in higher manufacturing costs and longer lead times, creating bottlenecks for supply chain managers aiming to secure reliable sources of high-quality API intermediates.
The Novel Approach
The methodology described in CN1025786C offers a streamlined and more efficient pathway by focusing on the precise construction of the fluorinated side chain prior to coupling with the indole core. This approach utilizes specific chiral auxiliary reagents, such as (4R,5S)-4-methyl-5-phenyl-2-oxazolidone, to induce high stereoselectivity during the formation of the 2-methyl-4,4,4-trifluoro butylamine moiety. By establishing the chiral center early in the synthesis, the need for downstream resolution is significantly reduced, thereby improving the overall process mass intensity. The use of mild coupling agents like 1-(3-dimethylaminopropyl)-3-ethyl carbodiimide (EDC) allows for amide bond formation under ambient or near-ambient temperatures, preserving the integrity of sensitive functional groups. This novel strategy not only enhances the chemical purity of the final product but also simplifies the workflow, making it more amenable to large-scale production while maintaining strict control over the impurity profile.
Mechanistic Insights into Fluorinated Indole Amide Coupling
The core chemical transformation in this patent involves the nucleophilic attack of the fluorinated amine on the activated carboxylic acid derivative of the indole scaffold. The reaction mechanism typically proceeds through the formation of an O-acylisourea intermediate when using carbodiimide coupling reagents, which is then attacked by the amine nitrogen to form the stable amide bond. The presence of the trifluoromethyl group in the side chain exerts a strong electron-withdrawing effect, which can influence the nucleophilicity of the amine and requires careful optimization of reaction conditions to ensure complete conversion. The patent details the use of organic bases like 4-dimethylaminopyridine (DMAP) to catalyze this acylation, facilitating the reaction in solvents such as methylene dichloride or tetrahydrofuran. Understanding this mechanism is vital for process chemists to troubleshoot potential issues related to incomplete reactions or the formation of N-acylurea byproducts, ensuring robust process performance.
Impurity control is another critical aspect of this synthesis, particularly regarding the enantiomeric excess (ee) of the final product. The patent specifies that the (R)-enantiomer is preferred for its superior leukotriene antagonistic properties, with typical ee values exceeding 95% or even 99% in optimized examples. The synthesis achieves this high optical purity by starting with enantiomerically pure precursors or by employing chiral resolution techniques during the intermediate stages. Analytical methods such as 19F NMR using chiral shift reagents are employed to monitor the stereochemical integrity throughout the process. This rigorous control ensures that the final pharmaceutical intermediate meets the stringent specifications required for clinical applications, minimizing the risk of biological activity variation caused by the presence of the inactive (S)-enantiomer.
How to Synthesize 5-(2-methyl-4,4,4-trifluoro butyl formamyl) Indole Derivatives Efficiently
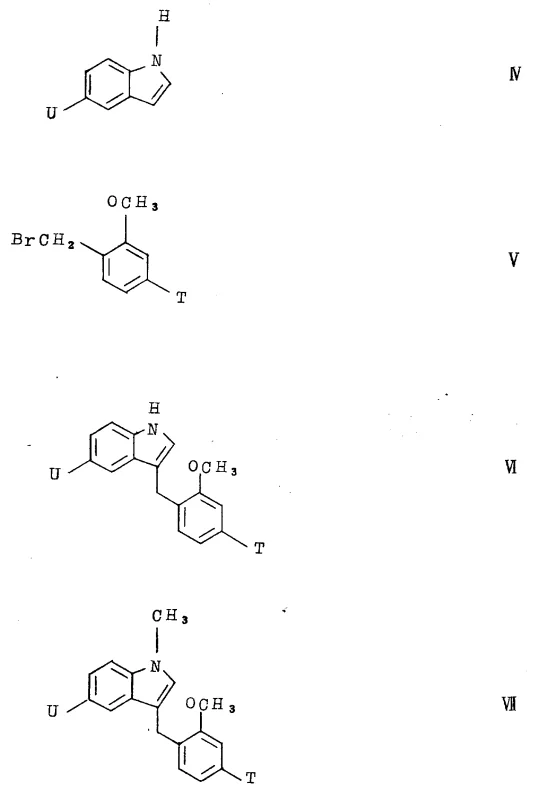
The synthesis of these high-value intermediates requires a systematic approach that integrates the preparation of the indole acid core with the chiral fluorinated amine side chain. The process begins with the alkylation of indole-5-carboxylic acid esters, followed by the introduction of the specific benzamide moiety. Detailed standard operating procedures for each reaction step, including precise stoichiometry, temperature profiles, and work-up protocols, are essential for reproducibility. The following guide outlines the critical stages of this synthesis, providing a roadmap for technical teams to implement this patented technology in a pilot or commercial setting. Adhering to these standardized steps ensures consistent quality and yield, which are paramount for maintaining supply chain reliability.
- Preparation of the indole-carboxylic acid core via alkylation of indole-5-carboxylic acid esters with alpha-brominated toluic acid esters.
- Synthesis of the chiral amine side chain, specifically 2-methyl-4,4,4-trifluoro butylamine, using chiral auxiliary reagents or resolution.
- Final amide coupling between the indole acid and the fluorinated amine using dehydrating agents like EDC or acid chlorides.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for anti-inflammatory intermediates. The streamlined process reduces the number of unit operations required, which directly translates to lower operational expenditures and reduced consumption of solvents and reagents. By eliminating the need for complex chiral resolution at the final stage, manufacturers can achieve significant cost savings in terms of both material usage and processing time. Additionally, the use of commercially available starting materials for the indole core enhances supply chain security, reducing the risk of disruptions caused by scarce raw materials. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The synthetic route described eliminates the need for expensive transition metal catalysts often found in cross-coupling reactions, thereby removing the costly and technically challenging step of heavy metal scavenging from the downstream process. This simplification significantly lowers the cost of goods sold (COGS) by reducing the consumption of specialized reagents and minimizing waste disposal costs associated with metal contaminants. Furthermore, the high yield and selectivity of the amide coupling step reduce the volume of mother liquor requiring treatment, leading to substantial savings in environmental compliance and waste management. These efficiency gains allow for a more competitive pricing structure without compromising on the quality or purity of the final intermediate.
- Enhanced Supply Chain Reliability: The reliance on robust and well-established chemical transformations, such as ester hydrolysis and carbodiimide-mediated coupling, ensures that the manufacturing process is less susceptible to technical failures or batch-to-batch variability. The intermediates involved, such as indole-5-carboxylic acid derivatives and fluorinated amines, are derived from widely available chemical feedstocks, which mitigates the risk of supply shortages. This stability is crucial for long-term supply agreements, providing procurement teams with the confidence that production timelines will be met consistently. The ability to source these materials from multiple qualified suppliers further strengthens the supply chain against geopolitical or logistical disruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that can be safely translated from laboratory scale to multi-ton commercial production. The avoidance of extremely low temperatures or high-pressure reactions simplifies the engineering requirements for production facilities, making it easier to scale up without significant capital investment in specialized equipment. Moreover, the reduced use of hazardous solvents and the generation of less toxic byproducts align with modern green chemistry principles, facilitating easier regulatory approval and environmental permitting. This compliance advantage is increasingly important for pharmaceutical companies aiming to meet stringent sustainability goals and regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbamoyl derivatives. These answers are derived from the detailed experimental data and claims presented in the patent documentation, providing clarity on the feasibility and advantages of this technology. Understanding these aspects helps stakeholders make informed decisions regarding the integration of this intermediate into their drug development pipelines. The information covers synthesis specifics, purity standards, and potential applications in therapeutic areas.
Q: What is the primary therapeutic application of these carbamoyl derivatives?
A: These compounds act as leukotriene antagonists, useful in treating allergic diseases, inflammation, asthma, and traumatic shock by blocking arachidonic acid metabolites.
Q: How is enantiomeric purity achieved in the synthesis?
A: High enantiomeric excess (ee) is achieved by using optically pure starting materials like (4R,5S)-4-methyl-5-phenyl-2-oxazolidone or through resolution of racemic mixtures.
Q: What are the key challenges in scaling this fluorinated synthesis?
A: Key challenges include handling fluorinated reagents safely, maintaining strict temperature controls during lithiation steps, and ensuring complete removal of heavy metal catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbamoyl Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing for complex pharmaceutical intermediates, possessing the technical expertise to bring patented routes like CN1025786C to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from R&D to market supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and quality of every batch. Our commitment to technical excellence ensures that the fluorinated indole derivatives you receive meet the highest standards required for global pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for leukotriene antagonist intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can support your drug development goals efficiently and reliably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
