Advanced Synthesis of Chiral Heterocyclic Tertiary Alcohol Intermediates for Antihypertensive APIs
The pharmaceutical industry's relentless pursuit of more effective cardiovascular treatments has placed significant emphasis on the development of high-quality 1,4-dihydropyridine-3,5-dicarboxylate derivatives, which serve as potent calcium channel blockers. Patent CN108689904B introduces a groundbreaking preparation method for chiral heterocyclic tertiary alcohol intermediates, specifically targeting the synthesis of complex pyrrolidine structures essential for these antihypertensive agents. This technology represents a paradigm shift from traditional resolution-based approaches to a more efficient asymmetric catalytic synthesis, addressing critical bottlenecks in yield and stereochemical control. By leveraging advanced oxidation and cyclization strategies, this method ensures the production of intermediates with exceptional purity and enantiomeric excess, directly impacting the efficacy and safety profile of the final active pharmaceutical ingredients (APIs). For R&D directors and procurement specialists, understanding this technological leap is vital for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent regulatory standards.
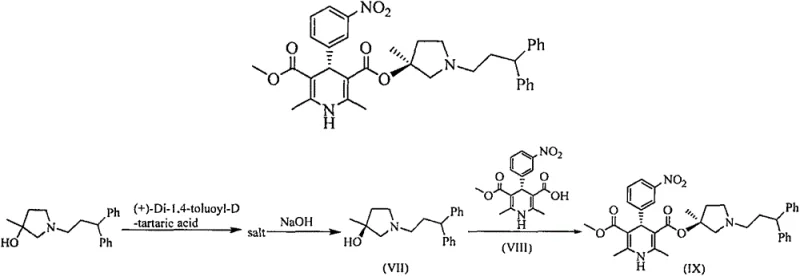
The limitations of conventional methods for producing chiral pyrrolidine intermediates have long plagued the supply chain for cardiovascular drugs, primarily due to the inherent inefficiency of chiral resolution techniques. Historically, obtaining the necessary stereochemistry involved resolving racemic mixtures, a process that theoretically caps the maximum yield at 50% and often results in practical yields as low as 16%, as noted in the background art of the patent. This massive material loss not only drives up the cost of goods sold but also creates significant waste disposal challenges, complicating environmental compliance for manufacturers. Furthermore, the purification conditions for resolved intermediates are often harsh and difficult to control, leading to batch-to-batch variability that can jeopardize the consistency of the final drug product. These factors combined make traditional resolution methods economically unsustainable and operationally risky for large-scale commercial production of high-purity OLED material or pharmaceutical precursors.
In stark contrast, the novel approach detailed in the patent utilizes a sophisticated asymmetric catalytic oxidation followed by a streamlined cyclization sequence to bypass the need for resolution entirely. This new route begins with the asymmetric dihydroxylation of an olefin precursor using a chiral base catalyst system, which establishes the critical stereocenters with an enantiomeric excess (ee) value exceeding 95%. The subsequent steps involve selective hydroxyl activation and intramolecular nucleophilic substitution, which efficiently construct the pyrrolidine ring while preserving the established chirality. By integrating these steps, the overall yield of the key intermediate (Formula VII) is boosted to at least 46%, representing a nearly threefold improvement over the legacy 16% yield benchmark. This drastic improvement in efficiency translates directly into cost reduction in pharmaceutical intermediates manufacturing, making the process viable for industrial expansion and ensuring a stable supply of critical raw materials.
Mechanistic Insights into Asymmetric Catalytic Oxidation and Cyclization
The core of this technological advancement lies in the precise mechanistic execution of the asymmetric catalytic oxidation, which sets the foundation for the molecule's biological activity. The process employs a chiral ligand, such as (DHQ)2PHAL, in conjunction with a catalytic amount of osmium tetroxide generated in situ from potassium osmate, to facilitate the syn-addition of hydroxyl groups across the olefin double bond. This reaction is conducted in a biphasic system of water and tert-butanol at controlled low temperatures, typically around 0°C, to maximize stereocontrol and minimize side reactions. The presence of a co-oxidant like potassium ferricyanide ensures the continuous regeneration of the active osmium species, allowing the reaction to proceed with high turnover numbers and minimal metal waste. This meticulous control over the oxidation step is crucial for achieving the >95% ee required for the subsequent formation of the biologically active dihydropyridine derivatives.
Following the establishment of chirality, the synthesis proceeds through a carefully orchestrated sequence of activation and cyclization to form the heterocyclic core. The primary hydroxyl group of the resulting chiral diol is selectively activated using a sulfonyl chloride, such as methanesulfonyl chloride, under alkaline conditions to create a superior leaving group. This activated intermediate then undergoes nucleophilic substitution with a specific amine derivative, introducing the diphenylpropyl side chain essential for the drug's receptor binding affinity. The final ring closure is achieved through an intramolecular cyclization where the secondary amine attacks the remaining activated position, forming the pyrrolidine ring with the tertiary alcohol intact. This mechanism avoids the use of expensive transition metal catalysts in the later stages, further simplifying the purification process and reducing the risk of heavy metal contamination in the final product.
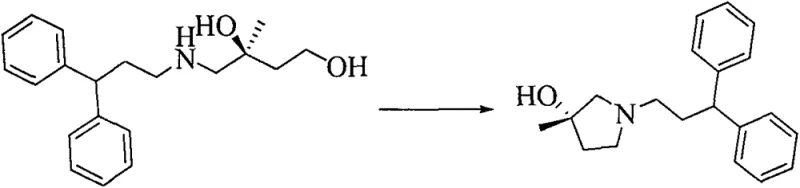
How to Synthesize Chiral Pyrrolidine Tertiary Alcohol Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized reaction parameters outlined in the patent to ensure consistent quality and yield. The process is designed to be robust, utilizing commercially available reagents and standard laboratory equipment, which facilitates easy technology transfer from pilot scale to full commercial production. Operators must pay particular attention to temperature control during the oxidation and activation steps, as deviations can impact the enantiomeric purity and the ratio of byproducts. The detailed standardized synthesis steps provided below offer a comprehensive guide for replicating this high-efficiency route in a GMP-compliant environment.
- Perform asymmetric catalytic oxidation on the olefin precursor using a chiral base catalyst and osmium co-catalyst to generate the chiral diol with high enantiomeric excess.
- Activate the primary hydroxyl group of the diol using a sulfonyl chloride activating agent under alkaline conditions to form the corresponding mesylate intermediate.
- Execute nucleophilic substitution with a diphenylpropyl amine derivative, followed by deprotection and intramolecular cyclization to close the pyrrolidine ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple yield improvements. The shift from a resolution-based process to a direct asymmetric synthesis fundamentally alters the cost structure of the intermediate, eliminating the 50% theoretical loss inherent in resolving racemates. According to the patent data, the production cost per kilogram using this method is only about 40% of the cost associated with traditional chiral resolution, representing a substantial cost savings that can be passed down the supply chain or reinvested in R&D. This economic advantage is compounded by the use of low-toxicity, readily available raw materials, which reduces dependency on scarce or volatile specialty reagents and enhances supply chain reliability.
- Cost Reduction in Manufacturing: The elimination of the chiral resolution step removes the need for expensive resolving agents and the associated recycling processes, which are often capital intensive and energy-consuming. By achieving a yield of at least 46% compared to the historical 16%, the material throughput is significantly increased without a proportional increase in reactor volume or processing time. This efficiency gain allows manufacturers to produce the same quantity of API intermediate with less than half the raw material input, drastically lowering the variable costs per unit. Furthermore, the simplified purification protocol, which relies on extraction and crystallization rather than complex chromatography, reduces solvent consumption and waste treatment expenses.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium carbonate, triethylamine, and common organic solvents ensures that the supply chain is resilient to market fluctuations that often affect specialized chiral catalysts or resolving agents. The robustness of the reaction conditions, which operate at mild temperatures and atmospheric pressure, minimizes the risk of batch failures due to equipment limitations or operational errors. This stability is critical for maintaining continuous production schedules and meeting the just-in-time delivery requirements of global pharmaceutical clients. Additionally, the high stereochemical purity (>95% ee) achieved consistently reduces the need for re-processing or rejection of off-spec batches, further securing the supply flow.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial expansion, with reaction steps that are easily scalable from gram to ton quantities without significant re-optimization. The avoidance of heavy metal catalysts in the final stages and the use of aqueous workups simplify the wastewater treatment process, aiding in compliance with increasingly stringent environmental regulations. The ability to recycle solvents like dichloromethane and ethyl acetate, combined with the high atom economy of the catalytic oxidation, contributes to a greener manufacturing footprint. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific data and embodiments provided in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this new method for the production of chiral heterocyclic intermediates. Understanding these details is essential for stakeholders evaluating the potential for technology licensing or contract manufacturing partnerships.
Q: How does the new catalytic method improve yield compared to traditional chiral resolution?
A: Traditional chiral resolution methods for this intermediate typically achieve yields of only around 16%, whereas the novel asymmetric catalytic synthesis described in patent CN108689904B increases the overall yield to at least 46% with an enantiomeric excess (ee) greater than 95%.
Q: What are the cost implications of switching to this synthetic route?
A: The patent data indicates that the production cost per kilogram using this new method is approximately 40% of the cost associated with traditional chiral resolution, primarily due to the elimination of wasteful resolution steps and the use of readily available raw materials.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild and controllable, utilizing common organic solvents and avoiding extreme temperatures or pressures, which makes the process highly suitable for commercial scale-up and industrial expansion.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Heterocyclic Tertiary Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of next-generation cardiovascular therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral pyrrolidine intermediate meets the highest global standards for safety and efficacy. Our infrastructure is designed to support the complex chemistry required for asymmetric synthesis, providing a secure and compliant environment for your most valuable projects.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your production volume and requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your competitive position in the global pharmaceutical market.
