Advanced Synthesis of 4-Hydroxybenzophenone Hydrazone Schiff Base for Oncology and Optical Applications
The pharmaceutical and fine chemical industries are constantly seeking advanced molecular scaffolds that bridge the gap between diagnostic imaging and therapeutic efficacy. Patent CN109824572B introduces a groundbreaking development in this sector with the synthesis of 4-((Z)-(((E)-(1-methyl-1H-indol-3-yl)methylene)hydrazono)(phenyl)methyl)phenol, a sophisticated Schiff base derivative. This compound represents a significant leap forward in the design of multifunctional organic materials, combining potent fluorescence properties with demonstrated antitumor activity against major cancer cell lines. The patent outlines a remarkably efficient synthetic route that bypasses the traditional complexities associated with heterocyclic functionalization, offering a streamlined pathway from readily available precursors to high-value end products. For R&D directors and procurement specialists alike, this technology signals a shift towards more sustainable and cost-effective manufacturing paradigms for complex organic intermediates. By leveraging a simple condensation reaction, the process minimizes waste generation and energy consumption while delivering a product with exceptional thermal stability and crystalline purity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex Schiff bases incorporating indole and benzophenone moieties has been plagued by inefficient multi-step protocols that require stringent anhydrous conditions and expensive transition metal catalysts. Conventional routes often involve the protection and deprotection of sensitive functional groups, leading to substantial material loss and extended production timelines that strain supply chain logistics. Furthermore, the purification of these intermediates frequently necessitates column chromatography or multiple recrystallization cycles, which not only escalates operational costs but also introduces risks of solvent contamination that can compromise downstream biological assays. The reliance on harsh reaction conditions, such as high temperatures or strong acidic environments, often results in the degradation of the indole ring system, generating difficult-to-remove impurities that lower the overall yield and optical quality of the final fluorescent material. These legacy methods create significant bottlenecks for manufacturers aiming to scale production for clinical or commercial optical applications.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN109824572B revolutionizes the production landscape by employing a direct, one-step condensation strategy that eliminates the need for complex catalytic systems or protective group chemistry. This innovative approach allows for the reaction to proceed efficiently in common organic solvents like acetonitrile or ethanol, and remarkably, it can even be executed via a solvent-free solid-phase grinding technique. This flexibility dramatically reduces the environmental footprint of the manufacturing process by cutting down on volatile organic compound (VOC) emissions and solvent disposal costs. The reaction conditions are mild, typically requiring only ambient temperature or gentle reflux, which preserves the integrity of the sensitive hydrazone linkage and ensures the formation of the desired Z/E isomeric configuration with high selectivity. By simplifying the workflow to a single mixing and stirring operation followed by straightforward filtration, this novel method offers a robust solution for the rapid deployment of high-purity fluorescent intermediates in both research and industrial settings.
Mechanistic Insights into Schiff Base Condensation and Crystallization
The core chemical transformation driving this synthesis is a nucleophilic addition-elimination reaction between the hydrazine moiety of 4-hydroxybenzophenone hydrazone and the carbonyl group of 1-methyl-3-indole-carbaldehyde. This condensation proceeds through the formation of a tetrahedral intermediate, which subsequently undergoes dehydration to establish the characteristic carbon-nitrogen double bond (C=N) of the Schiff base. The electronic interplay between the electron-rich indole ring and the conjugated benzophenone system stabilizes the resulting hydrazone linkage, facilitating the spontaneous organization of molecules into a highly ordered crystalline lattice. The structural rigidity imparted by this extended pi-conjugation system is directly responsible for the compound's intense fluorescence emission in the 350.0-470.0nm range, making it an ideal candidate for optoelectronic applications. Understanding this mechanism is crucial for process chemists aiming to optimize reaction kinetics and maximize the yield of the thermodynamically stable isomer.
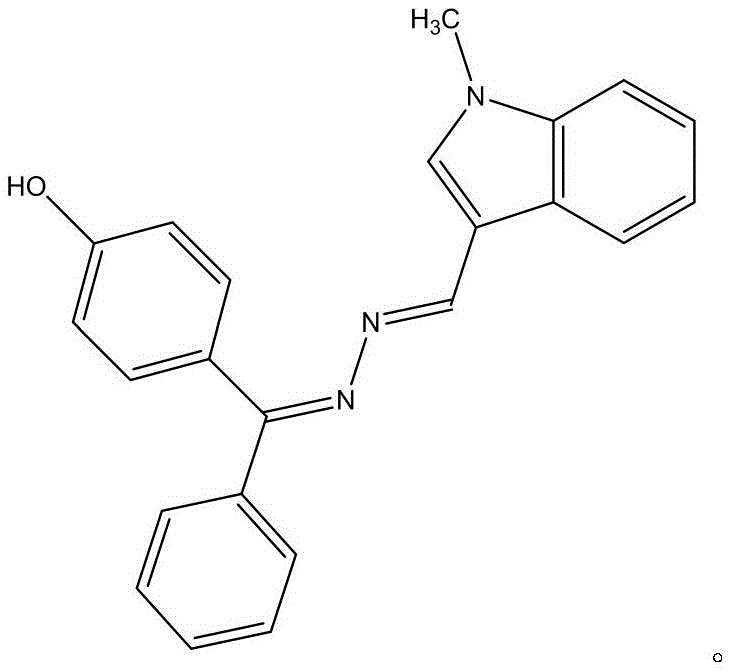
Impurity control in this synthesis is inherently managed by the high selectivity of the condensation reaction and the distinct solubility profile of the target product. The patent specifies that the resulting crystals belong to the monoclinic system with space group P21/n, exhibiting a sharp melting point of 195.1-196.1°C, which serves as a critical quality attribute for batch consistency. Any unreacted starting materials or side products typically remain in the mother liquor during the natural volatilization or recrystallization steps, ensuring that the precipitated yellow-green flakes possess the high purity required for biological testing. The absence of heavy metal catalysts in the reaction mixture further simplifies the impurity profile, removing the need for expensive scavenging resins or complex aqueous workups to meet regulatory limits for residual metals. This inherent purity by design significantly lowers the barrier for entry into regulated pharmaceutical supply chains where trace impurities can halt entire production batches.
How to Synthesize 4-Hydroxybenzophenone Hydrazone Schiff Base Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing operational simplicity and reproducibility. The process begins with the dissolution of the hydrazone precursor in a selected solvent, followed by the controlled addition of the indole aldehyde to initiate the condensation. Detailed standard operating procedures regarding stoichiometry, agitation speeds, and crystallization times are essential for maintaining the specific crystal habit and particle size distribution required for downstream processing. The following guide outlines the critical parameters derived from the patent examples to ensure successful replication of this high-value intermediate.
- Dissolve 4-hydroxybenzophenone hydrazone in a suitable organic solvent such as acetonitrile, ethanol, or dichloromethane, or prepare for a solvent-free solid-phase reaction.
- Add 1-methyl-3-indole-carbaldehyde to the solution maintaining a molar ratio between reactants of 1: 4 to 4:1, ensuring precise stoichiometric control for optimal yield.
- Stir or grind the mixture at room temperature or under reflux conditions for 2 to 10 hours, followed by filtration and recrystallization to isolate yellow-green flaky crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this synthesis route offers profound advantages in terms of cost structure and supply chain resilience. The elimination of exotic reagents and the ability to utilize commodity solvents or solvent-free conditions drastically reduces the raw material expenditure per kilogram of finished product. For procurement managers, this translates into a more predictable cost model that is less susceptible to fluctuations in the prices of specialized catalysts or reagents. The simplified workflow also means that production can be outsourced to a broader range of contract manufacturing organizations (CMOs) that may not possess specialized high-pressure or cryogenic capabilities, thereby expanding the potential supplier base and mitigating single-source risks. This flexibility is paramount for maintaining continuity of supply in the volatile global chemical market.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the reduction in unit operations and the avoidance of costly purification technologies. By consolidating the synthesis into a single step, manufacturers save significantly on labor, energy, and equipment depreciation costs associated with multi-stage reactors. The option to perform the reaction under solvent-free conditions further removes the substantial expense of solvent purchase, recovery, and hazardous waste disposal, leading to a leaner and more environmentally compliant production cost structure. Additionally, the high yield and purity achieved without chromatographic purification mean that material throughput is maximized, effectively lowering the cost of goods sold (COGS) for this high-performance fluorescent intermediate.
- Enhanced Supply Chain Reliability: The reliance on commercially abundant starting materials such as 4-hydroxybenzophenone hydrazone and 1-methyl-3-indole-carbaldehyde ensures a stable and secure supply chain foundation. These precursors are widely produced for various industrial applications, reducing the likelihood of shortages that often plague niche fine chemical intermediates. The robustness of the reaction conditions, which tolerate ambient temperatures and standard atmospheric pressure, allows for manufacturing in diverse geographic locations without the need for highly specialized infrastructure. This decentralization capability strengthens the overall supply network, ensuring that critical volumes of this anticancer and fluorescent agent can be delivered consistently to meet the demands of research and clinical trials.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is straightforward due to the absence of exothermic hazards or sensitive catalytic cycles that often complicate scale-up. The solid-phase or solution-based methods can be easily adapted to large-scale stirred tank reactors or continuous flow milling equipment, facilitating rapid capacity expansion as market demand grows. Furthermore, the green chemistry attributes of the process, particularly the potential for solvent-free operation and the generation of minimal waste streams, align perfectly with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturers and enhances the sustainability profile of the final product, a key differentiator for eco-conscious pharmaceutical and electronic material buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel Schiff base compound. These answers are derived directly from the experimental data and structural analysis provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details is vital for integrating this material into existing development pipelines.
Q: What are the key physical properties of this Schiff base compound?
A: The compound presents as yellow-green flaky crystals with a sharp melting point range of 195.1-196.1°C, indicating high structural integrity and purity suitable for sensitive analytical applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes a simple one-step condensation that can be performed in common solvents or via solvent-free solid-phase grinding, drastically simplifying scale-up and reducing equipment complexity.
Q: What are the primary biological and optical applications?
A: This material exhibits strong fluorescence emission between 350.0-470.0nm and demonstrates inhibitory activity against A549 lung cancer, 4T1 breast cancer, and Hep G2 liver cancer cells.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxybenzophenone Hydrazone Schiff Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in patent CN109824572B for the next generation of oncology therapeutics and optical sensors. As a premier CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific crystallization and purification requirements of this compound, guaranteeing stringent purity specifications that meet the rigorous demands of pharmaceutical grade intermediates. With our rigorous QC labs and commitment to process optimization, we deliver consistent quality that supports your critical R&D milestones and commercial launches.
We invite you to collaborate with us to leverage this advanced synthesis route for your specific application needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized manufacturing processes can enhance your project's economic viability. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your supply chain for high-purity fluorescent and anticancer intermediates.
