Advanced Synthesis of 5-Methoxy-7-Azaindole for Commercial Scale-Up and Procurement
The pharmaceutical industry continuously seeks efficient pathways for constructing nitrogen-containing heterocycles, particularly azaindole derivatives which serve as critical bioisosteres in drug design. Patent CN110128422B introduces a transformative synthesis method for 5-methoxy-7-azaindole, a key building block for various active pharmaceutical ingredients targeting protease inhibition and oncology. This technical disclosure addresses the longstanding economic and operational bottlenecks associated with traditional azaindole synthesis, offering a route that significantly shortens the reaction sequence while utilizing cost-effective starting materials. By shifting the synthetic strategy away from expensive precursors like 5-bromo-7-azaindole, this method enhances the overall feasibility of large-scale manufacturing. The innovation lies in the strategic use of 2-amino-3-methyl-5-bromopyridine as the foundational raw material, which is not only more accessible but also allows for a more direct construction of the target molecular architecture. This report analyzes the technical merits and commercial implications of this patented process for global supply chain stakeholders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
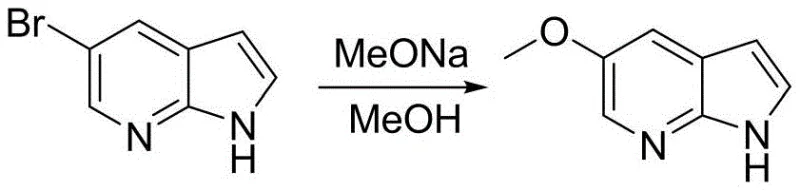
Historically, the preparation of 5-methoxy-7-azaindole has relied heavily on 5-bromo-7-azaindole as a key intermediate, a substance that presents significant supply chain vulnerabilities due to its high production cost and complex synthesis. Conventional routes often involve multi-step sequences including bromination, elimination, reduction, and oxidation, which collectively generate substantial waste streams and require rigorous purification protocols. For instance, prior art methods utilizing 7-azaindole often necessitate the use of excessive bromine and manganese dioxide, leading to serious environmental pollution and high waste treatment costs. Furthermore, routes involving Sonogashira cross-coupling introduce expensive palladium or copper catalysts that are difficult to remove completely, posing risks for residual metal impurities in the final drug substance. These traditional pathways are characterized by low total yields and harsh reaction conditions that complicate industrial scale-up and increase the overall cost of goods sold.
The Novel Approach
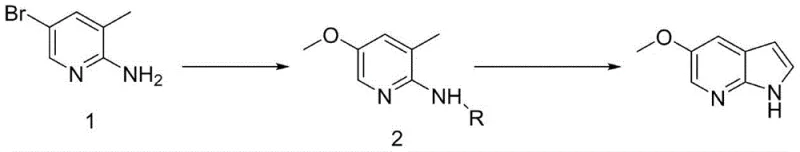
In stark contrast, the method disclosed in CN110128422B streamlines the synthesis into a highly efficient two-step process that bypasses the need for pre-formed azaindole scaffolds. The core innovation involves the direct methoxylation of 2-amino-3-methyl-5-bromopyridine followed by a protected cyclization sequence. This approach effectively avoids the generation of complex byproducts common in oxidation-reduction heavy routes, thereby simplifying downstream processing. The use of sodium methoxide for the initial substitution allows for high conversion rates under relatively mild thermal conditions, while the subsequent cyclization utilizes standard organolithium reagents that are well-understood in process chemistry. By eliminating the need for expensive and hard-to-source 5-bromo-7-azaindole, this novel route drastically reduces raw material expenditure and shortens the lead time for production. The overall yield is markedly improved, and the purity profile is enhanced, making it an ideal candidate for GMP manufacturing environments.
Mechanistic Insights into Copper-Catalyzed Methoxylation and Cyclization
The first critical stage of this synthesis involves the copper-catalyzed nucleophilic substitution of the bromine atom on the pyridine ring with a methoxy group. This reaction utilizes sodium methoxide in a methanol solvent system, facilitated by a copper catalyst such as copper powder or cuprous bromide. The mechanism likely proceeds through a copper-mediated oxidative addition and reductive elimination cycle, which activates the carbon-bromine bond for nucleophilic attack. The choice of catalyst and the molar ratio are crucial for minimizing side reactions such as dehalogenation or over-alkylation. Operating at temperatures between 100°C and 130°C ensures sufficient kinetic energy for the substitution while maintaining the stability of the amino group. This step is pivotal as it installs the essential methoxy functionality early in the sequence, setting the electronic properties of the ring for the subsequent cyclization event without requiring protecting group manipulation at this early stage.
Following the methoxylation, the synthesis proceeds through an amino protection step using di-tert-butyl dicarbonate, followed by a base-mediated cyclization. The protected intermediate is treated with a strong base such as n-butyllithium at low temperatures to deprotonate the methyl group, generating a reactive nucleophile. This anion then attacks N,N-dimethylformamide (DMF) to form an aldehyde intermediate in situ, which subsequently undergoes acid-catalyzed cyclization to close the pyrrole ring. The use of an amino protecting group is strategic, preventing unwanted side reactions at the nitrogen atom during the lithiation step. The final acidic quench not only neutralizes the reaction mixture but also facilitates the removal of the protecting group and the cyclization in a telescoped manner. This mechanistic sequence ensures high regioselectivity and minimizes the formation of isomeric impurities, resulting in a product with purity levels exceeding 98% after simple crystallization.
How to Synthesize 5-Methoxy-7-Azaindole Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the dissolution of 2-amino-3-methyl-5-bromopyridine in methanol, followed by the addition of sodium methoxide and a copper catalyst under heating. After the methoxylation is complete, the intermediate is isolated and subsequently protected with Boc anhydride in the presence of a base like DMAP. The final cyclization step demands strict temperature control during the addition of n-butyllithium to prevent thermal runaway, followed by the addition of DMF and acidic workup. Detailed standard operating procedures regarding stoichiometry, addition rates, and quenching protocols are essential for reproducible results. The following guide outlines the standardized synthesis steps derived from the patent examples for technical reference.
- Prepare 2-amino-3-methyl-5-methoxypyridine via copper-catalyzed reaction of 2-amino-3-methyl-5-bromopyridine with methoxide.
- Protect the amino group using di-tert-butyl dicarbonate to form the intermediate compound 2.
- Execute cyclization using n-butyllithium and DMF followed by acidic quenching to yield 5-methoxy-7-azaindole.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis method offers substantial advantages by fundamentally altering the raw material cost structure. By replacing the expensive and supply-constrained 5-bromo-7-azaindole with the more commoditized 2-amino-3-methyl-5-bromopyridine, manufacturers can achieve significant cost reduction in pharmaceutical intermediate manufacturing. The elimination of multiple redox steps and heavy metal catalysts further reduces the cost of goods by simplifying the bill of materials and lowering waste disposal expenses. This route is designed for robustness, meaning that supply chain disruptions related to specialized reagents are minimized. The reliance on common solvents like methanol, THF, and dichloromethane ensures that material availability remains high even during global supply fluctuations. Consequently, this method supports a more stable and predictable pricing model for long-term supply agreements.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of high-cost starting materials with readily available commodity chemicals. By avoiding the multi-step synthesis of 5-bromo-7-azaindole, the total number of unit operations is reduced, which directly correlates to lower labor and utility costs. Furthermore, the high yield and purity achieved reduce the need for extensive chromatographic purification, allowing for cost-effective crystallization as the primary isolation method. The removal of expensive transition metal catalysts also eliminates the need for costly metal scavenging steps, further enhancing the overall economic efficiency of the process.
- Enhanced Supply Chain Reliability: The use of 2-amino-3-methyl-5-bromopyridine as the starting material significantly reduces lead time for high-purity pharmaceutical intermediates because this chemical is produced by multiple suppliers globally. Unlike specialized azaindole derivatives which may have limited production capacity, the precursors for this route are part of established supply chains. This diversity of supply sources mitigates the risk of single-source dependency and ensures continuity of supply for downstream drug manufacturers. The robustness of the reaction conditions also means that production can be easily transferred between different manufacturing sites without significant re-validation efforts.
- Scalability and Environmental Compliance: This synthesis route is inherently scalable due to its use of standard reaction vessels and common reagents that do not require specialized high-pressure or cryogenic equipment beyond standard industry capabilities. The reduction in waste generation, particularly the avoidance of manganese and excessive bromine waste, aligns with increasingly stringent environmental regulations. This facilitates smoother regulatory approvals and reduces the environmental compliance burden on manufacturing facilities. The process safety profile is improved by avoiding hazardous oxidation steps, making it a safer option for large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They are intended to provide clarity on the feasibility, purity, and operational requirements of the process for potential partners and technical evaluators. Understanding these details is crucial for assessing the fit of this technology within existing manufacturing portfolios.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: This route utilizes cheaper, readily available starting materials like 2-amino-3-methyl-5-bromopyridine instead of expensive 5-bromo-7-azaindole, significantly reducing raw material costs and shortening the synthetic sequence.
Q: How does this method address impurity control in large-scale production?
A: The process avoids complex oxidation-reduction steps and heavy metal catalysts found in prior art, resulting in fewer byproducts and simplifying purification through crystallization.
Q: Is this synthesis method suitable for industrial scale-up?
A: Yes, the reaction conditions are mild, utilizing common solvents like methanol and THF, and the equipment requirements are standard, facilitating easy transition from lab to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Methoxy-7-Azaindole Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent technologies into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this synthesis route are fully realized in practice. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the high-quality standards required for pharmaceutical intermediates. We understand the critical nature of supply chain continuity and are committed to delivering consistent quality that meets the exacting demands of global R&D and production teams.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis specific to your project requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized synthesis can enhance your supply chain efficiency. By partnering with us, you gain access to a reliable supply of high-purity 5-methoxy-7-azaindole produced via this advanced, cost-effective method. Contact us today to discuss how we can support your drug development and commercialization goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
