Scalable Synthesis of Stable Boron-Containing Diradicals for Next-Generation Organic Electronics
Scalable Synthesis of Stable Boron-Containing Diradicals for Next-Generation Organic Electronics
The rapid evolution of organic electronics demands materials that combine exceptional electronic performance with robust chemical stability, a balance that has historically been difficult to achieve with open-shell radical species. Patent CN113773338A introduces a groundbreaking class of boron-containing organic radical compounds that effectively resolve the longstanding challenges of radical instability and synthetic complexity. By integrating electron-deficient boron atoms into a rigid indenofluorene conjugated framework, this technology enables the creation of stable singlet diradicals with tunable electronic properties. These materials exhibit remarkable near-infrared light absorption and bipolar charge transport characteristics, positioning them as prime candidates for advanced applications in organic field-effect transistors and spintronic devices. The strategic incorporation of steric hindrance groups further ensures that these highly reactive species maintain integrity under ambient conditions, overcoming a major bottleneck in the commercialization of radical-based functional materials.
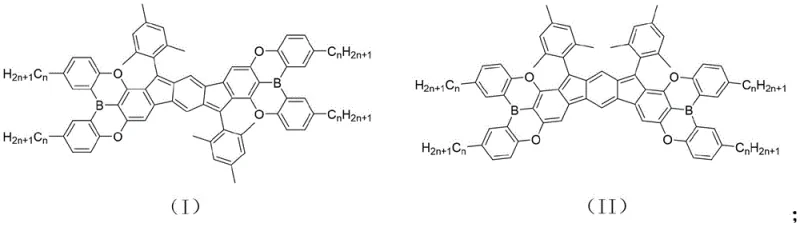
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing stable organic radicals often rely on massive steric bulk to kinetically trap the unpaired electrons, which frequently results in molecules with poor solubility and limited processability. Furthermore, conventional radical precursors often suffer from narrow bandgaps that are difficult to tune without compromising the stability of the open-shell structure. Many existing synthetic routes require harsh conditions or multiple purification steps that degrade the delicate radical character, leading to low yields and inconsistent batch-to-batch quality. The lack of effective strategies to modulate the energy levels of these radicals has restricted their utility in bipolar devices, where balanced hole and electron transport is essential. Consequently, the industry has faced a significant shortage of reliable, solution-processable radical materials capable of operating efficiently in practical electronic environments.
The Novel Approach
The methodology disclosed in the patent revolutionizes this landscape by utilizing a boron-doped indenofluorene scaffold that thermodynamically stabilizes the diradical state through electronic delocalization rather than just steric protection. This novel architecture allows for the precise adjustment of the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) levels by varying the substituents on the boron center. A key innovation is the modular synthesis which permits the attachment of alkyl chains of varying lengths, directly addressing the solubility issues that plague earlier generations of radical materials. By shifting from purely steric stabilization to an electronic stabilization strategy involving boron coordination, the synthesis achieves a superior balance between reactivity and stability. This approach not only simplifies the purification process but also enhances the material's compatibility with standard solution-processing techniques used in large-scale manufacturing.
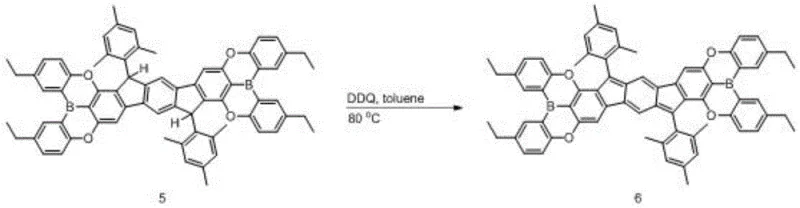
Mechanistic Insights into DDQ-Mediated Oxidative Aromatization
The core of this synthetic breakthrough lies in the final oxidative dehydrogenation step, where a dihydro-precursor is converted into the fully conjugated diradical system using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ). This reaction proceeds through a hydride abstraction mechanism where the electron-deficient quinone removes hydrogen atoms from the benzylic positions of the precursor, driving the formation of the extended pi-conjugated system. The presence of the boron atoms plays a critical role here by lowering the ionization potential of the intermediate, facilitating the removal of electrons and protons under relatively mild thermal conditions. The reaction is typically conducted in non-polar solvents like toluene or xylene at temperatures between 50°C and 100°C, ensuring that the delicate radical structure is not degraded by excessive heat. This controlled oxidation is vital for generating the specific singlet diradical ground state that gives the material its unique magnetic and optical properties.
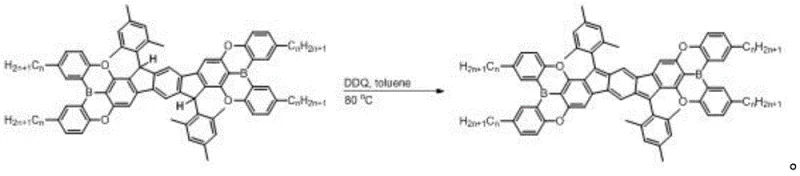
Impurity control in this process is managed through a combination of selective reactivity and rigorous purification protocols. The use of DDQ is advantageous because it is a selective oxidant that targets the specific C-H bonds required for aromatization without attacking the sensitive boron-oxygen bonds or the peripheral alkyl chains. Following the reaction, the crude mixture undergoes neutral alumina column chromatography, which effectively separates the polar quinone byproducts from the neutral radical product. Subsequent recrystallization from dichloromethane and acetonitrile further refines the purity, removing any trace amounts of unreacted precursor or over-oxidized species. This multi-stage purification ensures that the final material meets the stringent purity specifications required for high-performance electronic devices, minimizing trap states that could otherwise hinder charge carrier mobility.
How to Synthesize Boron-Containing Organic Radical Compounds Efficiently
The synthesis of these advanced materials follows a logical, step-wise progression designed to build molecular complexity while maintaining control over stereochemistry and functional group tolerance. The process begins with the construction of the ether-linked backbone, followed by the critical installation of the boron heterocycles which define the electronic character of the molecule. Subsequent coupling reactions extend the conjugation length, setting the stage for the final ring-closing and oxidation steps. Each stage is optimized for high yield and scalability, utilizing standard organometallic reagents and palladium catalysts that are readily available in industrial supply chains. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to replicate this synthesis, please refer to the standardized guide below.
- Perform nucleophilic aromatic substitution between difluorobenzene derivatives and phenols to construct the ether-linked precursor backbone.
- Execute lithiation followed by reaction with boron tribromide and cyclization to form the rigid boron-oxygen heterocyclic core.
- Conduct Suzuki-Miyaura coupling to extend the conjugated system, followed by Grignard addition and BF3-mediated cyclization to form the dihydro-precursor.
- Finalize the synthesis via oxidative dehydrogenation using DDQ in toluene at elevated temperatures to generate the target open-shell diradical species.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this boron-containing radical technology offers substantial advantages over legacy materials, primarily driven by enhanced processability and simplified manufacturing workflows. The ability to tune solubility through alkyl chain modification means that manufacturers can optimize ink formulations for printing processes without resorting to exotic or hazardous solvents. This flexibility reduces the dependency on specialized handling equipment and lowers the overall cost of goods sold by minimizing solvent waste and recovery expenses. Furthermore, the air stability of these compounds in dilute solutions significantly extends their shelf life and reduces the risk of batch rejection due to degradation during storage or transport. These factors collectively contribute to a more resilient and cost-effective supply chain for high-value organic electronic components.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive transition metal catalysts in the final oxidation step, relying instead on organic oxidants that are easier to handle and dispose of. By removing the requirement for rigorous inert atmosphere conditions during the final purification stages, the process reduces energy consumption associated with glovebox operations and nitrogen purging. The high selectivity of the DDQ oxidation minimizes the formation of side products, thereby reducing the load on downstream purification units and increasing the overall throughput of the production line. These efficiencies translate directly into lower operational expenditures and a more competitive pricing structure for the final electronic materials.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including substituted phenols and halogenated benzenes, are commodity chemicals with robust global supply networks, mitigating the risk of raw material shortages. The modular nature of the synthesis allows for the rapid substitution of alkyl chain precursors, enabling manufacturers to adapt quickly to changing market demands or regulatory restrictions on specific solvents. Additionally, the improved solubility of the final products facilitates easier handling and packaging, reducing the likelihood of shipping damages or quality deviations caused by precipitation during transit. This reliability ensures consistent delivery schedules and strengthens the partnership between material suppliers and device fabricators.
- Scalability and Environmental Compliance: The reaction conditions employed, such as moderate heating and the use of common organic solvents like toluene and dioxane, are well-suited for scale-up from laboratory to pilot plant and eventually to commercial tonnage production. The absence of heavy metal residues in the final oxidation step simplifies waste treatment protocols and ensures compliance with increasingly stringent environmental regulations regarding heavy metal discharge. The high atom economy of the coupling reactions further supports green chemistry initiatives by minimizing waste generation. These attributes make the technology highly attractive for large-scale manufacturing facilities aiming to reduce their environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-containing radical technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for R&D teams evaluating the material for specific device architectures and for procurement officers assessing the long-term viability of the supply source.
Q: What makes these boron-containing radicals more stable than traditional organic radicals?
A: The stability is achieved through a dual strategy: introducing electron-deficient boron atoms into the conjugated indenofluorene framework to delocalize spin density, and employing bulky mesityl groups as steric shields to protect the reactive radical centers from environmental degradation.
Q: How does the alkyl chain length affect the processability of these materials?
A: The patent demonstrates that increasing the alkyl chain length (n value) significantly enhances solubility in common organic solvents like dichloromethane and chloroform, which is critical for solution-processing techniques such as spin-coating in device fabrication.
Q: What are the primary electronic applications for these compounds?
A: Due to their narrow bandgap, bipolar charge transport capabilities, and near-infrared absorption properties, these compounds are ideally suited for bipolar organic field-effect transistors (OFETs), organic spintronic devices, and near-infrared dye applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boron-Containing Organic Radical Compound Supplier
As the demand for high-performance organic semiconductors continues to surge, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge materials backed by rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from prototype to mass market is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and electronic properties of every batch. This commitment to excellence guarantees that the boron-containing radicals you receive are perfectly suited for the demanding requirements of next-generation optoelectronic devices.
We invite you to engage with our technical procurement team to discuss how this innovative chemistry can be integrated into your specific product roadmap. By requesting a Customized Cost-Saving Analysis, you can identify specific opportunities to optimize your manufacturing budget without compromising on performance. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique application needs. Let us help you unlock the full potential of stable organic radicals in your commercial products.
