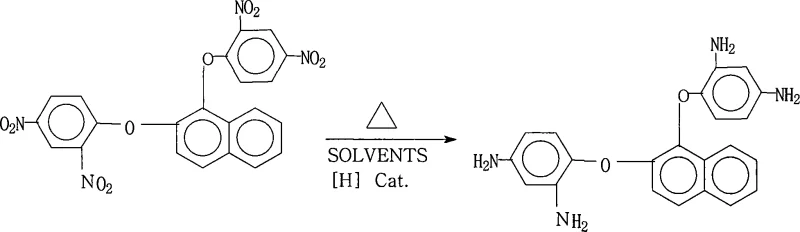
Advanced Catalytic Reduction for High-Purity 1,2-Bis(2,4-Diaminophenoxy)Naphthalene Production
The rapid evolution of the flexible electronics sector has placed unprecedented demands on the thermal and mechanical stability of insulating materials, specifically within the realm of Flexible Copper Clad Laminates (FCCL). Patent CN101245031A introduces a transformative preparation method for 1,2-bis(2,4-diaminophenoxy)naphthalene, a critical aromatic diamine monomer essential for synthesizing high-performance polyimide resins. This patented technology addresses the longstanding challenges in producing high-purity aromatic polyvalent primary amines by utilizing a sophisticated palladium-on-carbon (Pd/C) catalyzed reduction system. Unlike traditional nitro-reduction pathways that often suffer from incomplete conversion or difficult purification steps, this novel approach leverages hydrazine hydrate as a hydrogen donor in a saturated fatty alcohol solvent matrix. The result is a robust, scalable process capable of delivering product purity exceeding 99% with exceptional yield consistency, positioning it as a cornerstone technology for manufacturers seeking a reliable polyimide intermediate supplier.
For R&D directors and process engineers, the significance of this patent lies in its ability to streamline the supply chain for advanced electronic materials. The synthesis targets the precise molecular architecture required for next-generation polyimides that exhibit low dielectric constants and superior thermal resistance. By optimizing the reduction conditions to a moderate temperature range of 70°C to 85°C, the process minimizes thermal degradation of the sensitive naphthalene backbone while ensuring complete reduction of the nitro groups. This balance is crucial for maintaining the integrity of the final polymer, directly impacting the performance of end-use applications in aerospace, automotive, and microelectronics where material failure is not an option.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic diamines like 1,2-bis(2,4-diaminophenoxy)naphthalene has been plagued by inefficiencies inherent in classical reduction techniques. Traditional methods often rely on iron-acid reductions or high-pressure catalytic hydrogenation, both of which present significant operational drawbacks. Iron-acid reductions generate massive quantities of iron oxide sludge, creating severe environmental disposal issues and complicating the downstream purification process. Furthermore, these methods frequently result in products contaminated with residual metals, which can act as catalysts for polymer degradation or adversely affect the electrical properties of the resulting polyimide films. High-pressure hydrogenation, while cleaner, requires specialized, expensive reactor infrastructure and poses substantial safety risks related to hydrogen handling, making it less accessible for mid-scale production facilities.
The Novel Approach
The methodology outlined in CN101245031A represents a paradigm shift by employing a transfer hydrogenation strategy using hydrazine hydrate and a heterogeneous Pd/C catalyst. This approach effectively bypasses the need for high-pressure gas equipment, thereby drastically simplifying the reactor requirements and enhancing operational safety. The use of saturated fatty alcohols, such as 2-methoxyethanol or ethylene glycol, serves a dual purpose: they act as effective solvents for the bulky dinitro precursor and facilitate the smooth progression of the reduction reaction. A key innovation in this process is the implementation of a hot filtration step immediately following the reaction. This critical unit operation removes the spent catalyst and any insoluble impurities before the product crystallizes, ensuring that the final isolation yields a crystal lattice of exceptional purity without the need for energy-intensive recrystallization cycles.
Mechanistic Insights into Pd/C Catalyzed Hydrazine Reduction
The core of this synthetic breakthrough relies on the efficient catalytic decomposition of hydrazine on the surface of the palladium nanoparticles supported on carbon. In this mechanism, hydrazine acts as a source of nascent hydrogen, which is activated by the Pd surface and subsequently transferred to the nitro groups of the 1,2-bis(2,4-dinitrophenoxy)naphthalene substrate. The reaction proceeds through a series of intermediate stages, likely involving nitroso and hydroxylamine species, before finally arriving at the amine state. The choice of Pd/C with a palladium mass percentage ranging from 1% to 15% is critical; it provides sufficient active sites for rapid hydrogen generation while minimizing the leaching of precious metal into the product stream. The solvent system plays a pivotal role in stabilizing these intermediates and ensuring the solubility of the starting material, which is often a limiting factor in heterogeneous reactions involving large, planar aromatic systems.
From an impurity control perspective, the mechanism inherently favors high selectivity. The mild reaction conditions (70°C to 85°C) prevent the occurrence of side reactions such as dehalogenation (if halogens were present) or the reduction of the ether linkages connecting the naphthalene and phenyl rings. The hot filtration step is mechanistically sound because it exploits the solubility difference between the desired diamine and the catalyst/insoluble byproducts at elevated temperatures. By removing the catalyst while the solution is hot, the process prevents the re-adsorption of the product onto the catalyst surface or the catalysis of any potential degradation pathways during the cooling phase. This results in a product profile that meets the stringent specifications required for high-purity electronic chemical manufacturing, with reported purities consistently reaching 99.1% to 99.6% across various experimental embodiments.
How to Synthesize 1,2-Bis(2,4-Diaminophenoxy)Naphthalene Efficiently
The synthesis protocol detailed in the patent offers a clear pathway for laboratory and pilot-scale production, emphasizing precise stoichiometric control and thermal management. To achieve optimal results, practitioners must adhere to the specified molar ratios, particularly maintaining the 1,2-bis(2,4-dinitrophenoxy)naphthalene to hydrazine hydrate ratio between 1:20 and 1:50 to drive the reaction to completion. The detailed standardized synthesis steps below outline the precise addition rates and temperature profiles necessary to replicate the high yields reported in the patent data.
- Charge the reactor with 1,2-bis(2,4-dinitrophenoxy)naphthalene, Pd/C catalyst (1%-15% Pd loading), and a saturated fatty alcohol solvent system such as 2-methoxyethanol or ethylene glycol.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60%-85% concentration) while maintaining a molar ratio of substrate to hydrazine between 1: 20 and 1:50.
- Maintain reaction temperature between 70°C and 85°C for 1 to 5 hours, then perform hot filtration to remove the catalyst before cooling the mother liquor to precipitate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages beyond mere technical performance. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for production, allowing for more flexible manufacturing setups that can be deployed closer to key markets. Furthermore, the simplicity of the workup procedure—consisting primarily of hot filtration and water-induced crystallization—reduces the overall cycle time per batch. This efficiency gain allows for higher throughput without proportional increases in labor or utility costs, directly addressing the industry-wide pressure for cost reduction in electronic chemical manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the removal of expensive and hazardous unit operations. By substituting high-pressure hydrogen gas with liquid hydrazine hydrate, the facility avoids the capital expenditure associated with explosion-proof hydrogen infrastructure and the ongoing costs of safety compliance. Additionally, the catalyst, being heterogeneous, can potentially be recovered and regenerated, further lowering the raw material cost per kilogram of product. The high yield reported in the patent examples, often exceeding 90%, ensures that raw material utilization is maximized, minimizing waste disposal costs and improving the overall gross margin for the manufacturer.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as hydrazine hydrate, ethanol, and ethylene glycol ensures a robust and resilient supply chain. Unlike specialized reagents that may be subject to geopolitical shortages or long lead times, these solvents and reducing agents are produced globally at massive scales. This availability mitigates the risk of production stoppages due to raw material scarcity. Moreover, the process tolerance for various saturated fatty alcohols provides procurement teams with the flexibility to switch solvents based on regional availability and price fluctuations without compromising the reaction outcome, thereby securing continuous supply continuity.
- Scalability and Environmental Compliance: As regulatory frameworks regarding industrial emissions tighten, the environmental profile of a chemical process becomes a critical asset. This method generates significantly less hazardous waste compared to metal-acid reductions, as the primary byproduct is nitrogen gas and water. The solvent system, composed largely of alcohols and glycols, is amenable to distillation and recycling, aligning with green chemistry principles. This reduced environmental footprint simplifies the permitting process for new production lines and reduces the long-term liability associated with waste treatment, making it an ideal candidate for commercial scale-up of complex polyimide intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What is the primary advantage of using hydrazine hydrate over catalytic hydrogenation for this diamine?
A: The hydrazine hydrate method described in patent CN101245031A eliminates the need for high-pressure hydrogen gas equipment, significantly reducing capital expenditure and safety risks associated with high-pressure reactors while maintaining yields above 90%.
Q: How does this process ensure the high purity required for FCCL applications?
A: The process utilizes a hot filtration step immediately after reaction completion to remove the Pd/C catalyst and insoluble byproducts before crystallization, ensuring the final 1,2-bis(2,4-diaminophenoxy)naphthalene crystals achieve purity levels exceeding 99% without additional recrystallization.
Q: Can this synthesis route be scaled for metric ton production?
A: Yes, the patent explicitly states the method is suitable for industrialized production due to its simple operation, mild temperature range (70°C-85°C), and the use of common saturated fatty alcohol solvents that are easily recoverable and scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Bis(2,4-Diaminophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the Pd/C catalyzed reduction process are fully realized in a GMP-compliant environment. We understand that for applications in FCCL and high-performance composites, batch-to-batch consistency is paramount. Therefore, our rigorous QC labs enforce stringent purity specifications, utilizing advanced analytical techniques to verify that every shipment of 1,2-bis(2,4-diaminophenoxy)naphthalene meets the exacting standards required by the global electronics industry.
We invite procurement leaders and R&D innovators to collaborate with us to optimize their material sourcing strategies. By leveraging our expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your next generation of polyimide materials is built on a foundation of quality and reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
