Advanced Synthesis of Bipolar OLED Host Materials for Commercial Display Manufacturing
Advanced Synthesis of Bipolar OLED Host Materials for Commercial Display Manufacturing
Introduction to High-Efficiency Electroluminescent Compounds
The rapid evolution of display technology has necessitated the development of advanced organic electroluminescent materials that can sustain high efficiency under rigorous operating conditions. Patent CN108752372B introduces a novel compound, specifically 9-phenyl-3,9'-bicarbazole-6-boronic acid, which serves as a critical intermediate for constructing bipolar host materials. This chemical architecture is designed to mitigate the efficiency roll-off commonly observed in phosphorescent OLED devices by balancing hole and electron transport properties. The strategic incorporation of carbazole units provides robust hole-transporting capabilities, while the subsequent coupling with electron-deficient moieties creates a balanced charge injection system. 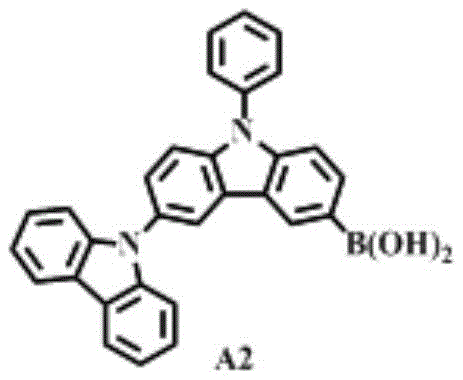 This structural innovation is pivotal for next-generation display panels requiring longer lifespans and lower power consumption. For procurement specialists and R&D directors, understanding the synthesis pathway of this compound is essential for securing a reliable OLED material supplier capable of delivering high-purity organic electroluminescent material at scale.
This structural innovation is pivotal for next-generation display panels requiring longer lifespans and lower power consumption. For procurement specialists and R&D directors, understanding the synthesis pathway of this compound is essential for securing a reliable OLED material supplier capable of delivering high-purity organic electroluminescent material at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for OLED host materials often suffer from complex multi-step procedures that involve expensive transition metal catalysts and harsh reaction conditions which are difficult to control on a large scale. Conventional methods frequently struggle with low overall yields due to the formation of stubborn by-products that are challenging to separate from the desired electroluminescent core. Furthermore, many existing processes rely on purification techniques that are not economically viable for commercial scale-up of complex display & optoelectronic materials, leading to prohibitive costs for mass production. The inability to effectively manage impurity profiles in older synthetic pathways often results in device failure or reduced operational stability, which is unacceptable for high-end commercial applications. Additionally, the use of non-optimized coupling reactions can lead to inconsistent batch-to-batch quality, creating significant supply chain risks for manufacturers who depend on a steady stream of high-purity organic electroluminescent materials. These technical bottlenecks highlight the urgent need for a more streamlined and robust synthetic methodology.
The Novel Approach
The methodology outlined in the patent data presents a breakthrough by utilizing a sequential Ullmann coupling followed by a precise lithiation-borylation sequence to construct the core bicarbazole framework. This novel approach significantly simplifies the synthetic route by reducing the number of isolation steps and employing reagents that are more readily available in the global chemical market. By optimizing the molar ratios of catalysts such as CuI and ligands like phenanthroline, the process achieves superior conversion rates while minimizing the formation of homocoupling by-products. The subsequent Suzuki coupling step allows for modular attachment of various electron-accepting groups, providing flexibility in tuning the electronic properties of the final material without redesigning the entire synthesis. This modularity is crucial for cost reduction in electronic chemical manufacturing as it allows producers to adapt quickly to different device requirements. Ultimately, this streamlined pathway offers a more sustainable and economically attractive alternative to legacy methods, ensuring better scalability and consistency.
Mechanistic Insights into CuI-Catalyzed Ullmann Coupling and Lithiation
The core of this synthesis lies in the initial Ullmann coupling reaction where 3,6-dibromocarbazole reacts with carbazole in the presence of CuI and 1,10-phenanthroline in DMF solvent at elevated temperatures around 165°C. This catalytic system facilitates the formation of the carbon-nitrogen bond between the carbazole units with high selectivity, creating the 9-phenyl-3,9'-bicarbazole backbone which is essential for hole transport. The choice of DMF as a polar aprotic solvent is critical for dissolving the inorganic base K2CO3 and stabilizing the catalytic intermediates throughout the prolonged reaction time of 20 to 28 hours. Following this, the brominated intermediate undergoes a delicate lithiation step using n-BuLi at cryogenic temperatures of -78°C to generate a reactive organolithium species. 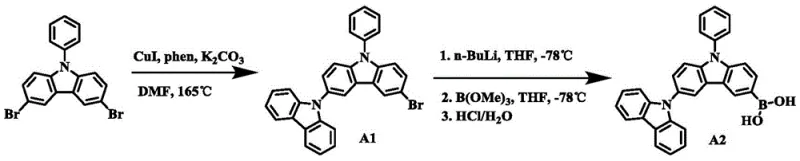 This low-temperature control is paramount to prevent side reactions such as nucleophilic attack on the carbazole ring or decomposition of the organolithium reagent. The subsequent addition of trimethyl borate converts this reactive species into the stable boronic acid derivative, ready for the final cross-coupling stage.
This low-temperature control is paramount to prevent side reactions such as nucleophilic attack on the carbazole ring or decomposition of the organolithium reagent. The subsequent addition of trimethyl borate converts this reactive species into the stable boronic acid derivative, ready for the final cross-coupling stage.
Impurity control is rigorously managed through specific workup procedures including quenching with dilute hydrochloric acid and extraction with dichloromethane to remove inorganic salts and metal residues. The patent specifies the use of column chromatography with precise mobile phase ratios, such as petroleum ether and dichloromethane, to isolate the white solid intermediates with high purity. This attention to purification is vital because trace metal contaminants like copper or palladium can act as quenching sites in the final OLED device, drastically reducing efficiency. The mechanistic pathway ensures that the electron-withdrawing groups are introduced only after the stable carbazole core is fully formed and purified, minimizing the risk of functional group incompatibility. By maintaining strict control over reaction stoichiometry and temperature profiles, the process ensures that the final bipolar host material possesses the necessary electronic properties to suppress triplet-triplet annihilation. This level of mechanistic precision is what distinguishes a laboratory curiosity from a commercially viable product for a reliable OLED material supplier.
How to Synthesize 9-Phenyl-3 9'-Bicarbazole-6-Boronic Acid Efficiently
Executing this synthesis requires strict adherence to the patented reaction conditions to ensure the successful formation of the target boronic acid intermediate. The process begins with the careful weighing of 3,6-dibromocarbazole and carbazole, followed by their dissolution in anhydrous DMF under a nitrogen atmosphere to prevent oxidation of the sensitive reagents. Operators must monitor the temperature closely during the Ullmann coupling phase to maintain the optimal range of 160-165°C, as deviations can lead to incomplete conversion or degradation of the starting materials. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results accurately.
- Perform Ullmann coupling of 3,6-dibromocarbazole with carbazole using CuI and phenanthroline in DMF at 165°C.
- Conduct lithiation of the brominated intermediate with n-BuLi at -78°C followed by borylation with trimethyl borate.
- Execute Suzuki coupling with electron-acceptor intermediates using Pd catalyst to form the final bipolar electroluminescent material.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers substantial benefits for procurement managers and supply chain heads looking to optimize their sourcing strategies for electronic chemicals. The use of relatively common catalysts and solvents reduces the dependency on exotic or highly regulated reagents, thereby stabilizing the raw material supply chain and mitigating the risk of procurement delays. The high yields reported in the patent examples suggest that waste generation is minimized, which translates to lower disposal costs and a reduced environmental footprint for the manufacturing facility. 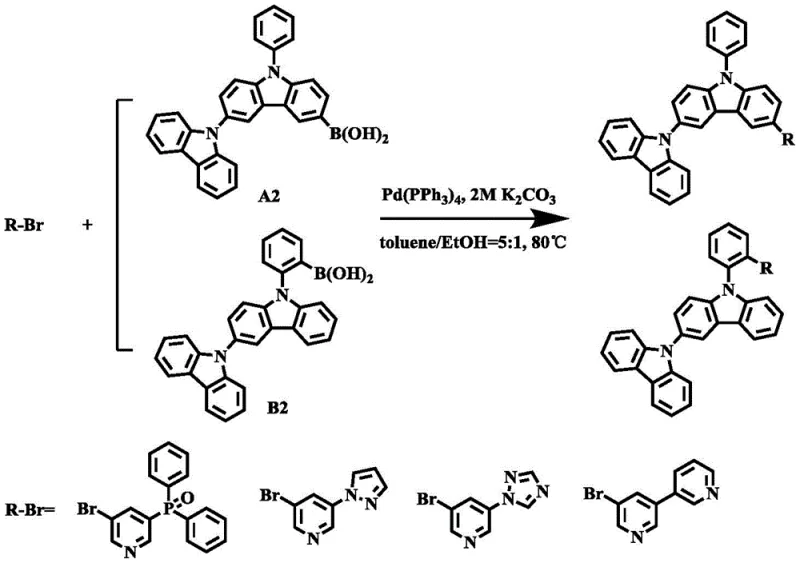 Furthermore, the modular nature of the final Suzuki coupling step allows for the production of a family of related materials using the same core intermediate, enhancing inventory efficiency. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding schedules of the display industry.
Furthermore, the modular nature of the final Suzuki coupling step allows for the production of a family of related materials using the same core intermediate, enhancing inventory efficiency. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding schedules of the display industry.
- Cost Reduction in Manufacturing: The elimination of complex protection-deprotection steps and the use of efficient catalytic systems significantly lower the operational expenditure associated with producing these advanced materials. By avoiding the need for expensive transition metal removal resins typically required for palladium-heavy processes, the overall production cost is drastically simplified. The high conversion efficiency means that less raw material is wasted, leading to substantial cost savings over large production runs. Additionally, the ability to recycle solvents like DMF and THF further enhances the economic viability of the process. This economic efficiency makes the material highly competitive in the market for cost reduction in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 3,6-dibromocarbazole and carbazole ensures that production is not bottlenecked by scarce precursors. The robust nature of the reaction conditions allows for manufacturing in standard chemical plants without requiring specialized high-pressure or cryogenic infrastructure beyond standard low-temperature reactors. This accessibility reduces lead time for high-purity organic electroluminescent materials by enabling multiple qualified suppliers to enter the market. Consequently, buyers can secure long-term contracts with greater confidence in the continuity of supply, which is critical for maintaining uninterrupted display panel production lines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and purification methods that are easily transferable from pilot scale to multi-ton production. The waste streams generated are primarily organic solvents and inorganic salts which can be treated using standard industrial wastewater treatment protocols, ensuring compliance with environmental regulations. The high selectivity of the reactions minimizes the formation of hazardous by-products, simplifying the environmental management burden. This alignment with green chemistry principles supports the growing demand for sustainable manufacturing practices in the specialty chemical sector. It facilitates the commercial scale-up of complex display & optoelectronic materials without compromising on regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bipolar electroluminescent materials. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this material into their product lines.
Q: What is the primary advantage of the bipolar host material described in CN108752372B?
A: The material utilizes a double electron-withdrawing group structure to balance carrier transport, significantly suppressing triplet-triplet annihilation and concentration quenching effects in OLED devices.
Q: How does the synthesis route ensure high purity for electronic applications?
A: The process employs rigorous column chromatography purification steps after each critical reaction stage, including Ullmann coupling and lithiation, to remove metal catalyst residues and by-products.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the reaction conditions utilize standard organic solvents like DMF and THF and common catalysts, making the transition from laboratory scale to commercial tonnage production feasible with proper engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Phenyl-3 9'-Bicarbazole-6-Boronic Acid Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the Ullmann and Suzuki coupling reactions described in the patent to meet stringent purity specifications required for OLED applications. We operate rigorous QC labs equipped with advanced analytical instrumentation to ensure every batch meets the high standards expected by global display manufacturers. Our commitment to quality ensures that the 9-phenyl-3,9'-bicarbazole-6-boronic acid we supply is free from detrimental impurities that could affect device performance.
We invite procurement leaders to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By collaborating with us, you can leverage our expertise to achieve a Customized Cost-Saving Analysis for your specific material requirements. We are dedicated to supporting your innovation with reliable supply and technical excellence, ensuring your display products reach the market with a competitive advantage.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
