Advanced Synthesis of High-Purity Ascorbyl Fatty Acid Esters for Global Supply Chains
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for producing high-value antioxidants, specifically ascorbyl fatty acid esters, which serve critical roles in stabilizing formulations across food, cosmetic, and medical sectors. Patent CN101973970B introduces a transformative approach to synthesizing these compounds, addressing long-standing inefficiencies in purification and yield that have plagued conventional manufacturing. This technology leverages a sophisticated liquid-liquid extraction strategy combined with in-situ recrystallization to deliver ascorbyl-6-fatty acid esters with purity exceeding 98 percent and yields consistently over 70 percent. By shifting away from hazardous ether-based extractions and difficult solid-state filtrations, this process offers a reliable food additives supplier pathway that ensures both economic viability and operational safety. The innovation lies not just in the chemical transformation but in the downstream processing engineering, which minimizes solvent consumption and maximizes throughput in a single reaction vessel.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of ascorbyl esters has been hindered by cumbersome post-reaction processing steps that severely impact overall efficiency and product quality. Prior art, such as U.S. Patent 4,151,178, relies heavily on diethyl ether for extraction, a solvent that is highly volatile, flammable, and poses significant safety risks in large-scale operations. Furthermore, these traditional methods often necessitate repetitive scrubbing of the crude product with petroleum ether to remove impurities, a procedure that frequently results in substantial product loss and poor crystal morphology. Another significant drawback found in methods like U.S. Patent 4,705,869 involves the direct addition of reaction mixtures into ice water, creating a viscous sludge that is notoriously difficult to filter and dry. This viscosity leads to emulsification during acid washing, trapping impurities within the product matrix and drastically reducing the final purity, thereby increasing the cost of goods sold due to the need for extensive rework or rejection of batches.
The Novel Approach
In stark contrast, the methodology disclosed in CN101973970B revolutionizes the workflow by maintaining the reaction mixture in a manageable liquid state throughout the critical separation phases. Instead of struggling with viscous solids, the process employs a tailored mixed solvent system comprising aliphatic monoaldehydes or ketones alongside halogenated hydrocarbons. This specific solvent combination exhibits excellent selectivity, dissolving the target ascorbyl ester effectively while remaining immiscible with the aqueous wash layers, thus preventing the emulsification issues that plague older techniques. The ability to perform the entire extraction and subsequent recrystallization within the same reaction kettle eliminates the need for transferring sticky intermediates between vessels, significantly reducing material loss and labor costs. This streamlined approach not only enhances the physical appearance of the final crystals but also ensures that residual fatty acids are efficiently removed during the halohydrocarbon recrystallization step, guaranteeing a high-purity end product suitable for sensitive applications.
Mechanistic Insights into Sulfuric Acid Catalyzed Esterification
The core chemical transformation involves the esterification of ascorbic acid or its alkali metal salts with long-chain fatty acids, catalyzed by concentrated sulfuric acid under controlled thermal conditions. The reaction selectively targets the primary hydroxyl group at the C-6 position of the ascorbic acid molecule, driven by steric and electronic factors that favor this site over the secondary hydroxyls. Operating at temperatures between 20°C and 30°C for a duration of 16 to 24 hours allows for complete conversion while minimizing degradation of the sensitive ascorbic acid backbone. The use of concentrated sulfuric acid serves a dual purpose: it acts as a potent dehydrating agent to drive the equilibrium toward ester formation and as a catalyst to lower the activation energy of the nucleophilic attack by the alcohol on the carboxylic acid carbonyl.
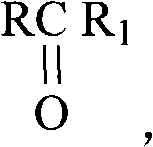
Following the reaction, the mechanistic advantage of the workup procedure becomes evident through the principles of solubility and phase partitioning. The addition of cold water serves to dilute the sulfuric acid and hydrolyze any transient sulfuric esters formed as byproducts, converting them back into water-soluble species that can be easily separated. The subsequent extraction with the aliphatic ketone and halohydrocarbon mixture exploits the differential solubility of the organic ester versus the inorganic salts and residual acids. Finally, the recrystallization step relies on the temperature-dependent solubility profile of the ascorbyl ester in halohydrocarbons; the product is highly soluble at elevated temperatures (near the solvent boiling point) but precipitates out as pure crystals upon cooling, leaving soluble impurities like unreacted fatty acids in the mother liquor. This thermodynamic purification mechanism is far superior to simple precipitation, ensuring the removal of trace contaminants that could otherwise compromise the stability of the final antioxidant.
How to Synthesize Ascorbyl Fatty Acid Esters Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for achieving industrial-grade purity without the need for complex chromatography or multiple isolation steps. The process begins with the careful mixing of fatty acid and sulfuric acid, followed by the addition of ascorbic acid, ensuring that the exothermic nature of the dissolution is managed to prevent localized overheating. After the reaction period, the mixture is cooled, and water is added in a controlled manner to manage the heat of dilution before the critical extraction phase begins. The detailed standardized synthetic steps see the guide below for precise operational parameters regarding solvent ratios and temperature gradients.
- React ascorbic acid or its salt with fatty acid (e.g., palmitic acid) in concentrated sulfuric acid at 20-30°C for 16-24 hours.
- Cool the mixture, add water carefully to control temperature, and extract the product using a mixed solution of aliphatic aldehydes/ketones and halohydrocarbons.
- Wash the organic phase with water to remove acid, evaporate solvents, and recrystallize the crude product in halohydrocarbon to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible strategic advantages regarding cost structure and supply reliability. The elimination of diethyl ether, a solvent with high volatility and strict storage requirements, significantly reduces the infrastructure costs associated with explosion-proof facilities and solvent recovery systems. Furthermore, the ability to conduct the entire process in a single vessel minimizes the capital expenditure required for transfer pumps, filtration units, and additional drying ovens, leading to a drastically simplified production footprint. The reduction in processing steps directly correlates to lower energy consumption, as there is no need for the energy-intensive drying of wet filter cakes that characterizes the older viscous-mass methods. These operational efficiencies allow for a more competitive pricing structure while maintaining healthy margins, making it an attractive option for cost reduction in food additive manufacturing.
- Cost Reduction in Manufacturing: The transition to a liquid-liquid extraction system removes the need for expensive and hazardous ether solvents, replacing them with more manageable halohydrocarbons and ketones that can be efficiently recovered and recycled. By avoiding the formation of viscous solids that are difficult to handle, the process reduces labor hours associated with equipment cleaning and maintenance, as well as minimizing product loss during transfer operations. The high selectivity of the recrystallization step means that fewer batches are rejected due to purity failures, effectively lowering the cost per kilogram of good product and optimizing the overall yield of raw materials.
- Enhanced Supply Chain Reliability: Traditional methods often suffer from batch-to-batch variability due to the unpredictability of filtering viscous mixtures, which can lead to delays and inconsistent delivery schedules. This novel approach offers a robust and reproducible workflow that is less susceptible to operational upsets, ensuring a steady flow of high-purity ascorbyl esters to meet market demand. The use of readily available raw materials such as palmitic acid and standard industrial solvents further secures the supply chain against raw material shortages, providing a stable foundation for long-term procurement contracts.
- Scalability and Environmental Compliance: Scaling up chemical processes often amplifies safety and environmental risks, but this method mitigates those concerns by operating at mild temperatures and avoiding the generation of large volumes of solid waste. The liquid-phase processing facilitates easier containment of emissions and spills, aligning with increasingly stringent environmental regulations regarding volatile organic compound (VOC) management. Additionally, the efficient solvent recovery potential of the halohydrocarbon system reduces the overall environmental footprint of the manufacturing site, supporting corporate sustainability goals without compromising on production volume.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for R&D teams evaluating the feasibility of adopting this technology for their own production lines or for procurement specialists assessing supplier capabilities.
Q: How does this process improve purity compared to traditional ether extraction methods?
A: Traditional methods using diethyl ether often result in poor crystal formation and require repetitive washing with petroleum ether, leading to product loss. This novel process utilizes a specific halohydrocarbon and aliphatic ketone mixture that effectively dissolves the ester while allowing easy phase separation from water, avoiding emulsification and ensuring purity levels exceed 98%.
Q: What are the scalability advantages of the liquid-liquid extraction method?
A: Conventional methods involve filtering viscous mixtures, which is difficult to scale and leads to significant material loss during transfer and drying. By maintaining the process in a liquid state within the same reaction kettle and utilizing liquid-liquid extraction, this method eliminates solid filtration bottlenecks, making it highly suitable for commercial scale-up from 100 kgs to multi-ton production.
Q: Can this synthesis route accommodate different fatty acid chain lengths?
A: Yes, the process is versatile and supports saturated fatty acids ranging from C4 to C20, such as lauric, palmitic, and stearic acid, as well as unsaturated fatty acids. The extraction and recrystallization parameters can be adjusted slightly to optimize yield for specific chain lengths while maintaining the core operational simplicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ascorbyl Fatty Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and scalable production in the global supply of fine chemicals and additives. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of ascorbyl ester meets the highest international standards, including those set by the European Pharmacopoeia. Our commitment to technical excellence allows us to offer customized solutions that align with your specific formulation requirements, whether for pharmaceutical stabilization or food preservation applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-purity intermediates with the reliability and transparency that modern supply chains demand.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
